Azulene
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Azulene[1] | |||
| Systematic IUPAC name
Bicyclo[5.3.0]decapentaene | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.005.449 | ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C10H8 | |||
| Molar mass | 128.174 g·mol−1 | ||
| Melting point | 99 to 100 °C (210 to 212 °F; 372 to 373 K) | ||
| Boiling point | 242 °C (468 °F; 515 K) | ||
| -98.5·10−6 cm3/mol
g/L[2] | |||
| Thermochemistry | |||
Std enthalpy of (ΔcH⦵298)combustion |
−1266.5 kcal/mol[3] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Azulene is an aromatic organic compound and an isomer of naphthalene. Naphthalene is colourless, whereas azulene is dark blue. The compound is named after its colour, as "azul" is Spanish for blue. Two terpenoids, vetivazulene (4,8-dimethyl-2-isopropylazulene) and guaiazulene (1,4-dimethyl-7-isopropylazulene), that feature the azulene skeleton are found in nature as constituents of pigments in mushrooms, guaiac wood oil, and some marine invertebrates.
Azulene has a long history, dating back to the 15th century as the azure-blue
Structure and bonding


Azulene is usually viewed as resulting from fusion of
Its

The dipolar nature of the ground state is reflected in its deep colour, which is unusual for small unsaturated aromatic compounds.[7] Another notable feature of azulene is that it violates Kasha's rule by exhibiting fluorescence from an upper-excited state (S2 → S0).[8]
Organic synthesis
Synthetic routes to azulene have long been of interest because of its unusual structure.[9] In 1939 the first method was reported by St. Pfau and Plattner [10] starting from indane and ethyl diazoacetate.
An efficient one-pot route entails annulation of cyclopentadiene with unsaturated C5-synthons.[11] The alternative approach from cycloheptatriene has long been known, one illustrative method being shown below.[12][13]
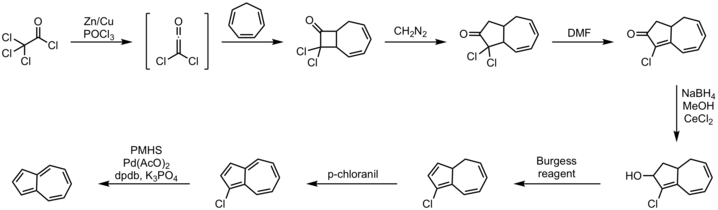
Procedure:
- cycloheptatriene 2+2 cycloaddition with dichloro ketene
- diazomethane insertion reaction
- dehydrohalogenation reaction with DMF
- Luche reduction to alcohol with sodium borohydride
- elimination reaction with Burgess reagent
- p-chloranil
- DPDB ligand
Organometallic complexes
In organometallic chemistry, azulene serves as a ligand for low-valent metal centers. Illustrative complexes are (azulene)Mo2(CO)6 and (azulene)Fe2(CO)5.[14]
Derivatives
1-Hydroxyazulene is an unstable green oil and it does not show
In naphth[a]azulene, a naphthalene ring is condensed at the 1,2-positions of azulene. In one such system[17] deviation from planarity is found, similar to that of tetrahelicene.
Guaiazulene (1,4-dimethyl-7-isopropylazulene) is an alkylated derivative of azulene with an almost identical intensely blue colour. It is commercially available to the cosmetics industry where it functions as a skin conditioning agent.
References
- ISBN 978-0-85404-182-4.
- PMID 8975804.
- .
- S2CID 21207966.
- doi:10.26021/9162.
- .
- .
- .
- .
- .
- .
- PMID 16013070.
- .
- ISBN 9780470166123.
- .
- ^ .
- PMID 16238325.
External links
- MSDS Website