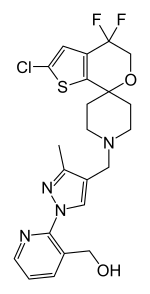
BTRX-246040
 | |
| Clinical data | |
|---|---|
| Other names | BTRX-246040 |
| Routes of administration | By mouth |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
BTRX-246040, also known as LY-2940094, is a potent and selective nociceptin receptor antagonist which is under development by BlackThorn Therapeutics and Eli Lilly for the treatment of major depressive disorder (MDD).[1][2][3] It has demonstrated proof-of-concept clinical efficacy for depression.[4] As of 2017, it is in phase II clinical trials for the treatment of MDD.[1][2][3] It was also under investigation for the treatment of alcoholism, and similarly reached phase II clinical studies for this indication, but development was discontinued.[1]
See also
References
External links
- "BTRX-246040 (LY-2940094)". AdisInsight. Springer Nature Switzerland AG.
