Babesiosis
| Babesiosis | |
|---|---|
| Other names | Babesiasis |
Infectious disease | |
| Symptoms | fever, chills, headache, fatigue[1] |
| Risk factors | Removed spleen, weakened immune system, doing outdoor activities[1] |
| Differential diagnosis | Malaria |
Babesiosis or piroplasmosis is a
Signs and symptoms
Half of all children and a quarter of previously healthy adults with Babesia infection are asymptomatic. When people do develop symptoms, the most common are fever and hemolytic anemia, symptoms that are similar to those of malaria.[5] People with symptoms usually become ill 1 to 4 weeks after the bite, or 1 to 9 weeks after transfusion of contaminated blood products. A person infected with babesiosis gradually develops malaise and fatigue, followed by a fever. Hemolytic anemia, in which red blood cells are destroyed and removed from the blood, also develops. Chills, sweats, and thrombocytopenia are also common symptoms. Symptoms may last from several days to several months.[citation needed]
Less common symptoms and physical exam findings of mild-to-moderate babesiosis:[5]
- Headache
- Muscle pain
- Anorexia
- Nonproductive cough (mucus is not coughed up)
- Arthralgias(noninflammatory joint pain, unlike arthritis, which is inflammatory)
- Nausea
- Vomiting
- Sore throat
- Abdominal pain
- Pink eye/Conjunctivitis
- Photophobia (abnormal intolerance to visual perception of light)
- Weight loss
- Emotional lability
- Depression
- Hyperesthesia (more sensitive to stimuli)
- Enlarged spleen
- Pharyngeal erythema
- Enlarged liver
- Jaundice (yellowing of the skin and of the sclera)
- Retinopathy with splinter hemorrhages
- Retinal infarcts
- Neutropenia[7]
In more severe cases, symptoms similar to malaria occur, with fevers up to 40.5 °C (105 °F), shaking chills, and severe anemia (hemolytic anemia). Organ failure may follow, including adult respiratory distress syndrome. Sepsis in people who have had a splenectomy can occur rapidly, consistent with overwhelming post-splenectomy infection. Severe cases are also more likely to occur in the very young, very old, and persons with immunodeficiency, such as HIV/AIDS patients.[citation needed]
A reported increase in human babesiosis diagnoses in the 2000s is thought to be caused by more widespread testing and higher numbers of people with immunodeficiencies coming in contact with ticks, the disease vector.
Cause
Babesia species are in the phylum Apicomplexa, which also has the protozoan parasites that cause malaria, toxoplasmosis, and cryptosporidiosis.[5] Four clades of Babesia species infect humans. The main species in each clade are:[citation needed]
- B. microti (<3 µm)
- B. duncani
- B. divergens (cattle parasite seen mostly in Europe) and B. venatorum (roe deer parasite, formerly called EU1), most closely related to the large Babesia clade
- Large Babesia (>3 µm) mostly infects ungulates, but also includes K01 strain (an isolated case observed in South Korea, see isolated cases)
Pathophysiology
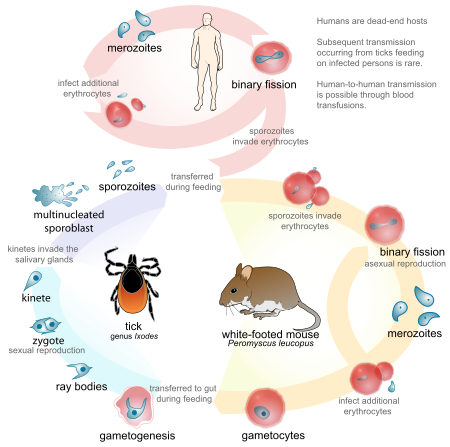
Babesia parasites reproduce in
Unlike the Plasmodium parasites that cause malaria, Babesia species lack an exoerythrocytic phase, so the liver is usually not affected.[citation needed]
In nonhuman animals, Babesia canis rossi, Babesia bigemina, and Babesia bovis cause particularly severe forms of the disease, including a severe haemolytic anaemia, with positive erythrocyte-in-saline-agglutination test indicating an immune-mediated component to the haemolysis. Common sequelae include haemoglobinuria "red-water", disseminated intravascular coagulation, and "cerebral babesiosis" caused by sludging of erythrocytes in cerebral capillaries.[citation needed]
In bovine species, the organism causes hemolytic anemia, so an infected animal shows pale mucous membranes initially. As the levels of bilirubin (a byproduct of red blood cell lysis) continue to increase, the visible mucous membranes become yellow in color (icterus) due to the failure of the liver to metabolize the excess bilirubin. Hemoglobinuria is seen due to excretion of red-blood-cell lysis byproducts via the kidneys. Fever of 40.5 °C (105 °F) develops due to release of inflammatory byproducts.[citation needed]
Diagnosis
Only specialized laboratories can adequately diagnose Babesia infection in humans, so Babesia infections are considered highly under-reported. It develops in patients who live in or travel to an endemic area or receive a contaminated
So-called "Maltese cross formations" on the blood film are diagnostic (
A polymerase chain reaction (PCR) test has been developed for the detection of Babesia from the peripheral blood.[13] PCR may be at least as sensitive and specific as blood-film examination in diagnosing babesiosis, though it is also significantly more expensive.[14] Most often, PCR testing is used in conjunction with blood film examination and possibly serologic testing.[10]
Other laboratory findings include decreased numbers of red blood cells and
In animals, babesiosis is suspected by observation of clinical signs (hemoglobinuria and anemia) in animals in endemic areas. Diagnosis is confirmed by observation of merozoites on thin film blood smear examined at maximum magnification under oil using Romonovski stains (methylene blue and eosin). This is a routine part of the veterinary examination of dogs and ruminants in regions where babesiosis is endemic.[citation needed]
Babesia canis and B. bigemina are "large Babesia species" that form paired merozoites in the erythrocytes, commonly described as resembling "two pears hanging together", rather than the "Maltese cross" of the "small Babesia species". Their merozoites are around twice the size of small ones.[citation needed]
Cerebral babesiosis is suspected in vivo when neurological signs (often severe) are seen in cattle that are positive for B. bovis on blood smear, but this has yet to be proven scientifically. Outspoken red discoloration of the grey matter post mortem further strengthens suspicion of cerebral babesiosis. Diagnosis is confirmed post mortem by observation of Babesia-infected erythrocytes sludged in the cerebral cortical capillaries in a brain smear.[citation needed]
Treatment
Treatment of asymptomatic carriers should be considered if parasites are still detected after 3 months. In mild-to-moderate babesiosis, the treatment of choice is a combination of
Imidocarb is a drug used for treatment of babesiosis in dogs.[16] Extracts of the poisonous,
Epidemiology
Babesiosis is a
In the United States, the majority of babesiosis cases are caused by B. microti, and occur in the Northeast and northern Midwest from May through October. The Centers for Disease Control and Prevention now requires state health departments to report infections using Form OMB No. 0920-0728.[25] In 2014, Rhode Island had an incidence of 16.3 reported infections per 100,000 people.[26]
In Europe, B. divergens is the primary cause of infectious babesiosis and is transmitted by I. ricinus.[5]
Babesiosis has emerged in Lower Hudson Valley, New York, since 2001.[27]
In Australia, one locally-acquired case of B. microti has been reported, which was fatal.[28] A subsequent investigation found no additional evidence of human Babesiosis in over 7000 patient samples, leading the authors to conclude that Babesiosis was rare in Australia.[29] A similar disease in cattle, commonly known as tick fever, is spread by Babesia bovis and B. bigemina in the introduced cattle tick Rhipicephalus microplus. This disease is found in eastern and northern Australia.[30]
Isolated cases
A table of isolated cases of babesiosis, which may be underestimated given how widely distributed the tick vectors are in temperate latitudes.[5]
| Location | Species |
|---|---|
| Pacific Coast (northern California to Washington) | B. duncani |
| Kentucky, Missouri, and Washington | B. divergens |
| Austria, Germany, Italy | B. venatorum |
Canary islands |
B. microti |
| Africa (Egypt, Mozambique, South Africa) | Uncharacterized spp. |
| Asia (Taiwan, Japan) | B. microti |
| South Korea | Babesia KO1[31] |
| Australia | B. microti[29] |
| South America (Brazil, Colombia) | Uncharacterized spp. |
History
The disease is named for the genus of the causative organism,
In 1957, the first human case was documented in a
Other animals
Veterinary treatment of babesiosis does not normally use antibiotics. In livestock and animals,
References
- ^ a b "CDC - Babesiosis". Centers for Disease Control and Prevention. Archived from the original on 2021-03-08. Retrieved 2021-02-25.
- ISBN 978-0-7637-2962-2. Archivedfrom the original on 2013-11-13. Retrieved 2016-10-14.
- ^ Prevention CC. "CDC - Babesiosis". www.cdc.gov. Archived from the original on 2015-10-25. Retrieved 2015-10-25.
- ^ Prevention CC. "CDC2 - Babesiosis" (PDF). www.cdc.gov. Archived (PDF) from the original on 2018-07-27. Retrieved 2018-09-20.
- ^ (PDF) from the original on 2021-11-12.
- ^ PMID 18440005.
- from the original on 2019-12-18. Retrieved 2015-08-19.
- ISBN 978-1-84076-578-6. Archivedfrom the original on 26 June 2014. Retrieved 14 October 2016.
- ^ PMID 18434647.
- ^ (PDF) from the original on 2020-07-26. Retrieved 2019-09-20.
- PMID 12804380.
- PMID 8133112.
- PMID 1500517.
- PMID 8897184.
- PMID 26232090.
- ^ "Imizol | Overview". Merck Animal Health. Archived from the original on 7 August 2015. Retrieved 15 August 2015.
- ^ Duke JA. ""Boophone disticha" (Liliaceae)". Dr. Duke's Phytochemical and Ethnobotanical Databases. Archived from the original on January 15, 2018. Retrieved November 10, 2011.
- S2CID 74852997.
- S2CID 807441.
- ISBN 978-0-87488-596-5. Archivedfrom the original on 2020-07-26. Retrieved 2016-10-14.
- ^ Belluck P (September 6, 2009). "Tick-Borne Illnesses Have Nantucket Considering Some Deer-Based Solutions". The New York Times. Archived from the original on November 15, 2011. Retrieved September 6, 2009.
- S2CID 46733758.
- from the original on 2022-07-21. Retrieved 2012-05-26.
- ^ "Babesiosis FAQs". Centers for Disease Control and Prevention (CDC. May 5, 2009. Archived from the original on November 28, 2010. Retrieved September 6, 2009.
- ^ "Parasites - Babesiosis: National Surveillance". Centers for Disease Control and Prevention. Archived from the original on December 16, 2017. Retrieved 19 June 2018.
- ^ Centers for Disease Control and Prevention. "Data & Statistics". Center. Archived from the original on 19 June 2018. Retrieved 19 June 2018.
- PMID 21529393.
Related news articles: Laurie Tarkan (June 20, 2011). "Once Rare, Infection by Tick Bites Spreads". The New York Times. Archived from the original on November 25, 2016. Retrieved February 24, 2017. - S2CID 33068508.
- ^ S2CID 148570372.
- ^ Queensland Government, Department of Primary Industries and Fisheries. "Tick fever (bovine babesiosis)". Archived from the original on 5 April 2011. Retrieved 21 February 2012.
- PMID 17392446.
- ^ "babesia" Archived 2022-04-24 at the Wayback Machine at Merriam-Webster online.
- Who Named It?
- ^ "Definition of Piroplasma". lexic.us. Archived from the original on November 11, 2011. Retrieved November 9, 2011.
External links
- Center for Global Health (2019-06-25). "Babesiosis". Parasites and Health, DPDx—Laboratory Identification of Parasites of Public Health Concern. Centers for Disease Control & Prevention. Archived from the original on 2013-03-07. Retrieved 2003-10-07. Public domain source from which the first version of this article was derived.
- Krause PJ, Auwaerter PG, Bannuru RR, Branda JA, Falck-Ytter YT, Lantos PM, et al. (27 January 2021). "Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA): 2020 Guideline on Diagnosis and Management of Babesiosis". Clinical Infectious Diseases. 72 (2): e49–e64. PMID 33252652.
- Homer MJ, Aguilar-Delfin I, Telford SR, Krause PJ, Persing DH (July 2000). "Babesiosis". Clin. Microbiol. Rev. 13 (3): 451–69. PMID 10885987.
- "Babesiosis: Overview"—The Merck Veterinary Manual
- Current status of Equine piroplasmosis worldwide at OIE. WAHID Interface—OIE World Animal Health Information Database
- Disease card—OIE
