Barbier reaction
| Barbier reaction | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Named after | Philippe Barbier | ||||||||||
| Reaction type | Coupling reaction | ||||||||||
| Reaction | |||||||||||
| |||||||||||
| Identifiers | |||||||||||
| RSC ontology ID | RXNO:0000084 | ||||||||||
carbonyl group and a metal. The reaction can be performed using magnesium, aluminium, zinc, indium, tin, samarium, barium or their salts. The reaction product is a primary, secondary or tertiary alcohol. The reaction is similar to the Grignard reaction but the crucial difference is that the organometallic species in the Barbier reaction is generated in situ, whereas a Grignard reagent is prepared separately before addition of the carbonyl compound.[1] Unlike many Grignard reagents, the organometallic species generated in a Barbier reaction are unstable and thus cannot be stored or sold commercially. Barbier reactions are nucleophilic addition reactions that involve relatively inexpensive, water insensitive metals (e.g zinc powder) or metal compounds. For this reason, it is possible in many cases to run the reaction in water, making the procedure part of green chemistry. In contrast, Grignard reagents and organolithium reagents are highly moisture sensitive and must be used under an inert atmosphere without the presence of water. The Barbier reaction is named after Philippe Barbier, who was Victor Grignard
's teacher.
Scope
Examples of Barbier reactions are the reaction of propargylic bromide with butanal with zinc metal (The attached reference details that the reaction goes to completion after the addition of saturated aqueous ammonium chloride):[2]
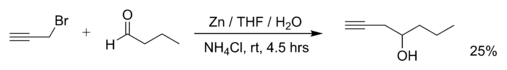
With a substituted
allene
product is favouredthe intramolecular Barbier reaction with samarium(II) iodide:[3]
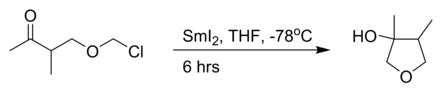
the reaction of an

The Barbier reaction is accompanied by an allylic rearrangement to a terminal alkene
The reaction of 3-Bromocyclohexene with benzaldehyde and zinc powder in water:[5]
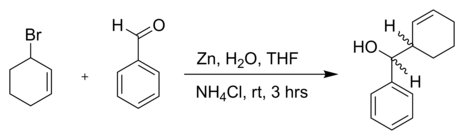
The observed
threo
= 83 : 17Asymmetric variants
The synthesis of (+)-aspicillin, starts first with a hydroboration, then transmetallation to zinc which can then do an addition into the aldehyde substituent.[6]

See also
- Nozaki–Hiyama–Kishi reaction
- Indium mediated allylation
External links
- Barbier reaction @ University of Connecticut Website
References
- ^ Barbier, P. (1899). "Synthèse du diéthylhepténol". Compt. Rend. 128: 110.
- PMC 6236518.
- ^ Tore Skjæret & Tore Benneche (2001). "Preparation of oxo-substituted α-chloro ethers and their reaction with samarium diiodide". Arkivoc: KU–242A.
- Leo A. Paquette. "Methyl 3-(hydroxymethyl)-4-methyl-2-methylenepentanoate". Organic Syntheses; Collected Volumes, vol. 10, p. 77.
- PMC 6236354.
- .
