Bat virome

The bat virome is the
Bats harbor several viruses that are
Despite the abundance of viruses associated with bats, they rarely become ill from viral infections, and rabies is the only viral illness known to kill bats. Much research has been conducted on bat virology, particularly bat immune response. Bats' immune systems differ from other mammals in their lack of several inflammasomes, which activate the body's inflammatory response, as well as a dampened stimulator of interferon genes (STING) response, which helps control host response to pathogens. Preliminary evidence indicates bats are thus more tolerant of infection than other mammals. While much research has centered on bats as a source of zoonotic disease, reviews have found mixed results on whether bats harbor more zoonotic viruses than other groups. A 2015 review found that bats do not harbor more zoonotic viruses than primates or rodents, though the three groups harbored more than other mammal orders.[4] In contrast, a 2020 review found that bats do not have more zoonotic viruses than any other bird or mammal group when viral diversity is measured relative to host diversity, as bats are the second-most diverse order of mammals.[5]
Viral diversity
| Virus family | No. sequences (n = 10,845) |
|---|---|
| Coronaviridae | 3,796(35.0%)
|
| Rhabdoviridae | 2,890(26.6%)
|
| Paramyxoviridae | 1,025(9.5%)
|
Astroviridae
|
724(6.7%)
|
| Adenoviridae | 365(3.4%)
|
| Polyomaviridae | 302(2.8%)
|
Reoviridae
|
288(2.7%)
|
| Circoviridae | 250(2.3%)
|
| Herpesviridae | 233(2.1%)
|
| Flaviviridae | 218(2.0%)
|
Picornaviridae
|
181(1.7%)
|
| Parvoviridae | 165(1.5%)
|
| Filoviridae | 123(1.1%)
|
| Hepadnaviridae | 78(< 1.0%)
|
| Papillomaviridae | 59(< 1.0%)
|
| Hantaviridae | 59(<1.0%)
|
| Caliciviridae | 43(< 1.0%)
|
| Peribunyaviridae | 31(< 1.0%)
|
| Nairoviridae | 22(< 1.0%)
|
Retroviridae
|
18(< 1.0%)
|
| Orthomyxoviridae | 8(< 1.0%)
|
| Phenuiviridae | 8(< 1.0%)
|
| Poxviridae | 6(< 1.0%)
|
Picobirnaviridae
|
4(< 1.0%)
|
Togaviridae
|
3(< 1.0%)
|
| Genomoviridae | 2(< 1.0%)
|
| Bornaviridae | 2(< 1.0%)
|
| Anelloviridae | 1(< 1.0%)
|
Viruses have been found in bat populations around the world. Bats harbor all groups of viruses in the Baltimore classification,[7] representing at least 28 families of viruses.[6] Most of the viruses harbored by bats are RNA viruses, though they are also known to have DNA viruses.[8] Bats are more tolerant of viruses than terrestrial mammals.[8] A single bat can host several different kinds of viruses without becoming ill.[9] Bats have also been shown to be more susceptible to reinfection with the same viruses, whereas other mammals, especially humans, have a greater propensity for developing varying degrees of immunity.[10][11] Their behavior and life history also make them "exquisitely suitable hosts of viruses and other disease agents", with long lifespans, the ability to enter torpor or hibernate, and their ability to traverse landscapes with daily and seasonal movement.[1]
Though bats harbor diverse viruses, they are rarely lethal to the bat host. Only the rabies virus and a few other lyssaviruses have been confirmed to kill bats.
Additionally, bats lack several
Transmission to humans

The vast majority of bat viruses have no
Bats compared to other viral reservoirs
Bats and their viruses may be the subject of more research than viruses found in other mammal orders, an example of research bias. A 2015 review found that from 1999 to 2013, there were 300–1200 papers published about bat viruses annually, compared to 12–45 publications for marsupial viruses and only 1–9 studies for sloth viruses. The same review found that bats do not have significantly greater viral diversity than other mammal groups. Bats, rodents, and primates all harbored significantly more zoonotic viruses than other mammal groups, though the differences among the aforementioned three groups were not significant (bats have no more zoonotic viruses than rodents and primates).[4] A 2020 review of mammals and birds found that the identity of the taxonomic groups did not have any impact on the probability of harboring zoonotic viruses. Instead, more diverse groups had greater viral diversity. Bat life history traits and immunity, while likely influential in determining bat viral communities, were not associated with a greater probability of viral spillover into humans.[5]
Sampling

Bats are sampled for viruses in a variety of ways. They can be tested for seropositivity for a given virus using a method like
Double-stranded DNA viruses
Adenoviruses
Herpesviruses
Diverse
Papillomaviruses
Single-stranded DNA viruses
Anelloviruses
No
Circoviruses
Parvoviruses
Several kinds of
Double-stranded RNA viruses
Reoviruses
| Virus name | Year identified | Host | Location |
|---|---|---|---|
| Nelson Bay virus | 1968 | Bat | Australia |
| Pulau virus | 1999 | Bat | Malaysia |
| Melaka virus | 2006 | Human | Malaysia |
| Kampar virus | 2006 | Human | Malaysia |
| HK23629/07 | 2007 | Human | Hong Kong |
| Miyazaki-Bali/2007 | 2007 | Human | Indonesia/Japan |
| Sikamat virus | 2010 | Human | Malaysia |
| Xi River virus | 2010 | Bat | China |
| Indonesia/2010 | 2010 | Bat | Indonesia/Italy |
Zoonotic
Some disease-causing reovirus species are associated with bats. One such virus is Melaka virus, which was linked to illness in a Malaysian man and his two children in 2006.[25][26] The man said that a bat had been in his home a week before he became ill, and the virus was closely related to other reoviruses linked to bats. Kampar virus was identified a few months later in another Malaysian man. Though he had no known contact with bats, Kampar virus is closely related to Melaka virus. Several other reovirus strains identified in ill humans are known as Miyazaki‐Bali/2007, Sikamat virus, and SI‐MRV01. No reoviruses linked to bats have caused death in humans.[25]
Other
Reoviruses include many viruses that do not cause disease in humans, including several found in bats. One reovirus species associated with bats is
Orbiviruses have been isolated from bats, including Ife virus from the straw-colored fruit bat, Japanaut virus from the common blossom bat, and Fomédé virus from Nycteris species.[25]
Positive-sense single-stranded RNA viruses
Astroviruses
Caliciviruses
Bat
SARS-CoV, SARS-CoV-2, and MERS-CoV
Several zoonotic coronaviruses are associated with bats, including severe acute respiratory syndrome coronavirus (SARS-CoV) and
The first human case of
The COVID-19 pandemic in humans started in Wuhan, China in 2019.[34] Genetic analyses of SARS-COV-2 showed that it was highly similar to viruses found in horseshoe bats, with 96% similarity to a virus isolated from the intermediate horseshoe bat. Due to similarity with known bat coronaviruses, data "clearly indicates" that the natural reservoirs of SARS-COV-2 are bats. It is yet unclear how the virus was transmitted to humans, though an intermediate host may have been involved.[3] Phylogenetic reconstruction of SARS-CoV-2 suggests that the strain that caused a human pandemic diverged from the strain found in bats decades ago, likely between 1950 and 1980.[35]
Other
Bats harbor a great diversity of coronaviruses, with sampling by the EcoHealth Alliance in China alone identifying about 400 new strains of coronavirus.[36] A study of coronavirus diversity harbored by bats in eastern Thailand revealed forty-seven coronaviruses.[37]
Flaviviruses
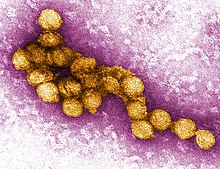
Most
Several genera of picornaviruses have been found in bats, including Kobuvirus, Sapelovirus, Cardiovirus, and Senecavirus.[18] Picornaviruses have been identified from a diverse array of bat species around the world.[7]
Negative-sense single-stranded RNA viruses
Arenaviruses are mainly associated with rodents, though some can cause illness in humans. The first arenavirus identified in bats was Tacaribe mammarenavirus, which was isolated from Jamaican fruit bats and the great fruit-eating bat. Antibody response associated with Tacaribe virus has also been found in the common vampire bat, the little yellow-shouldered bat, and Heller's broad-nosed bat. It is unclear if bats are the natural reservoir of Tacaribe virus. There has been one known human infection by Tacaribe virus, though it was accidentally acquired in a laboratory setting.[18]
Hantaviruses
Filoviruses
Marburgvirus and Ebolavirus

Ebola virus disease is a relatively rare but life-threatening illness in humans, with an average mortality rate of 50% (though individual outbreaks may be as high as 90% mortality). The first outbreaks were in 1976 in
Marburg virus disease (MVD) was first identified in 1967 during simultaneous outbreaks in
Other
Rhabdoviruses
Rabies-causing viruses

Lyssaviruses (from the genus Lyssavirus in the family Rhabdoviridae) include the rabies virus, Australian bat lyssavirus, and other related viruses, many of which are also harbored by bats. Unlike most other viruses in the family Rhabdoviridae, which are transmitted by arthropods, lyssaviruses are transmitted by mammals, most frequently through biting. All mammals are susceptible to lyssaviruses, though bats and carnivores are the most common natural reservoirs. The vast majority of human rabies cases are a result of the rabies virus, with only twelve other human cases attributed to other lyssaviruses as of 2015.[51] These rarer lyssaviruses associated with bats include Duvenhage lyssavirus (three human cases as of 2015); European bat 1 lyssavirus (one human case as of 2015); European bat 2 lyssavirus (two human cases as of 2015); and Irkut lyssavirus (one human case as of 2015). Microbats are suspected as the reservoirs of these four uncommon lyssaviruses.[51][52]
After transmission has occurred, the average human is asymptomatic for two months, though the incubation period can be as short as a week or as long as several years.[51] Italian scientist Antonio Carini was the first to hypothesize that rabies virus could be transmitted by bats, which he did in 1911. This same conclusion was reached by Hélder Queiroz in 1934 and Joseph Lennox Pawan in 1936. Vampire bats were the first to be documented with rabies; in 1953, an insectivorous bat in Florida was discovered with rabies, making it the first documented occurrence in an insectivorous species outside the vampire bats' ranges.[53] Bats have an overall low prevalence of rabies virus, with a majority of surveys of apparently healthy individuals showing rabies incidence of 0.0–0.5%.[51] Sick bats are more likely to be submitted for rabies testing than apparently healthy bats, known as sampling bias,[54] with most studies reporting rabies incidence of 5–20% in sick or dead bats.[51] Rabies virus exposure can be fatal in bats, though it is likely that the majority of individuals do not develop the disease after exposure.[51] In non-bat mammals, exposure to the rabies virus almost always leads to death.[52]

Globally, dogs are by far the most common source of human rabies deaths.
Other
Many bat lyssaviruses are not associated with infection in humans. These include Lagos bat lyssavirus, Shimoni bat lyssavirus, Khujand lyssavirus, Aravan lyssavirus, Bokeloh bat lyssavirus, West Caucasian bat lyssavirus, and Lleida bat lyssavirus.[52][51] Lagos bat lyssavirus, also known as Lagos bat virus (LBV), has been isolated from a megabat in sub-Saharan Africa.[51] This lyssavirus has four distinct lineages, all of which are found in the straw-colored fruit bat.[59]
Rhabdoviruses from other genera have been identified in bats. This includes several from the genus Ledantevirus: Kern Canyon virus, which was found in the Yuma myotis in California (US); Kolente virus from the Jones's roundleaf bat in Guinea;[60] Mount Elgon bat virus from the eloquent horseshoe bat in Kenya; Oita virus from the little Japanese horseshoe bat; and Fikirini virus from the striped leaf-nosed bat in Kenya.[61]
Orthomyxoviruses
Paramyxoviruses
Hendra, Nipah, and Menangle viruses

The first human outbreak of Nipah virus was in 1998 in Malaysia.
An additional zoonotic paramyxovirus that bats harbor is
Other
Bats host several paramyxoviruses that are not known to affect humans. Bats are the reservoir of Cedar virus, a paramyxovirus first discovered in flying foxes South East Queensland.[16] The zoonotic potential of Cedar virus is unknown.[66] In Brazil in 1979, Mapuera orthorubulavirus was isolated from the saliva of the little yellow-shouldered bat. Mapuera virus has never been associated with disease in other animals or humans, but experimental exposure of mice to the virus resulted in fatality.[16] Tioman pararubulavirus has been isolated from the urine of the small flying fox, which causes fever in some domestic pigs after exposure, but no other symptoms. Tukoko virus has been detected from Leschenault's rousette in China.[16] Bats have been suggested as the host of Porcine orthorubulavirus, though definitive evidence has not been collected.[16]
Togaviruses
Positive-sense single-stranded RNA viruses that replicate through a DNA intermediate
Retroviruses
Bats can be infected with retroviruses, including the gammaretrovirus found in horseshoe bats, Leschenault's rousette, and the greater false vampire bat. Several bat retroviruses have been identified that are similar to the Reticuloendotheliosis virus found in birds. These retroviruses were found in mouse-eared bats, horseshoe bats, and flying foxes. The discovery of varied and distinct gammaretroviruses in bat genomes indicates that bats likely played important roles in their diversification. Bats also host an extensive number of betaretroviruses, including within mouse-eared bats, horseshoe bats, and flying foxes. Bat betaretroviruses span the entire breadth of betaretrovirus diversity, similar to those of rodents, which may indicate that bats and rodents are primary reservoirs of the viruses. Betaretroviruses have infected bats for a majority of bat evolutionary history, since at least 36 million years ago.[67]
Double-stranded DNA viruses that replicate through a single-stranded RNA intermediate
See also
References
- ^ PMID 16847084.
- PMID 25742261.
An increasingly asked question is 'can we confidently link bats with emerging viruses?'. No, or not yet, is the qualified answer based on the evidence available.
- ^ PMID 32226946.
Evidence from the sequence analyses clearly indicates that the reservoir host of the virus was a bat, probably a Chinese or Intermediate horseshoe bat, and it is probable that, like SARS-CoV, an intermediate host was the source of the outbreak.
- ^ ISBN 978-1118818824.
- ^ PMID 32284401.
- ^ PMID 32528128.
- ^ PMID 27578437.
- ^ PMID 29478775.
- ^ Gorman, James (28 January 2020). "How Do Bats Live With So Many Viruses?". The New York Times. Retrieved 17 March 2020.
- S2CID 22591717.
- PMID 1773865.
- PMID 26603901.
- PMID 30804542.
- PMID 29549333.
- ^ PMID 30092093.
- ^ ISBN 978-1118818824.
- PMID 26867024.
- ^ ISBN 9781118818824.
- ^ "ICTV Master Species List 2018b.v2". International Committee on Taxonomy of Viruses (ICTV). Archived from the original on 30 March 2019. Retrieved 19 June 2019.
- PMID 25359906.
- PMID 29128758.
- ^ PMID 32088806.
- PMID 28371451.
- PMID 25497353.
- ^ ISBN 9781118818824.
- PMID 28368854.
- PMID 29789360.
- ^ ISBN 978-1118818824.
- PMID 32015507.
- ^ "Novel Coronavirus (2019-nCoV) Situation Report – 22" (PDF). World Health Organization. 11 February 2020. Retrieved 15 February 2020.
- PMID 26206723.
- ^ "Middle East respiratory syndrome coronavirus (MERS-CoV)". World Health Organization. November 2019. Retrieved 5 April 2020.
- ^ "Middle East respiratory syndrome coronavirus (MERS-CoV)". World Health Organization. 11 March 2019. Retrieved 5 April 2020.
- ^ Nsikan, Akpan (21 January 2020). "New coronavirus can spread between humans – but it started in a wildlife market". National Geographic. Archived from the original on 22 January 2020. Retrieved 23 January 2020.
- .
- ^ Aizenman, Nurith (20 February 2020). "New Research: Bats Harbor Hundreds Of Coronaviruses, And Spillovers Aren't Rare". NPR. Retrieved 5 April 2020.
- PMID 25884446.
- ^ PMID 31308502.
- PMID 25643870.
- ^ ISBN 9781118818824.
- ^ a b "Ebola virus disease". World Health Organization. 10 February 2020. Retrieved 13 April 2020.
- ^ a b "What is Ebola Virus Disease?". Centers for Disease Control and Prevention. 5 November 2019. Retrieved 13 April 2020.
Scientists do not know where Ebola virus comes from.
- PMID 25960093.
Despite concerted investigative efforts, the natural reservoir of the virus is unknown.
- ^ PMID 27959626.
The geographic ranges of many animal species, including bats, squirrels, mice and rats, dormice, and shrews, match or overlap with known outbreak sites of African filoviruses, but none of these mammals has yet been universally accepted as an EBOV reservoir.
- ISBN 978-0-323-95389-4, retrieved 2 March 2023
- .
We found published evidence from cases of serological and/or polymerase chain reaction (PCR) positivity of EVD in non- human mammal, or of EVD-linked mortality, in 28 mammal species: 10 primates, three rodents, one shrew, eight bats, one carnivore, and five ungulates
- ^ a b c d "Marburg virus disease". World Health Organization. 15 February 2018. Retrieved 14 April 2020.
- S2CID 57574565.
- ^ a b c "Filoviruses – Ebola and Marburg Viruses". BU Research Support. 12 June 2019. Retrieved 14 April 2020.
- PMID 31003196.
- ^ ISBN 978-1118818824.
- ^ PMID 21601050.
- ISBN 978-1118818824.
- PMID 21269998.
- ^ "Rabies". www.who.int. Retrieved 8 July 2020.
- ^ PMID 30890183.
- S2CID 212732288.
- ^ PMID 12203172.
- PMID 29505617.
- PMID 25487727.
- PMID 25679389.
- ^ PMID 25281398.
- PMID 28407738.
- PMID 30869046.
- PMID 25919464.
- PMID 31548390.
- ^ ISBN 9781118818824.
- PMID 26897577.
