Benzamil
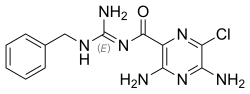
| |
| Names | |
|---|---|
| IUPAC name
3,5-diamino-N-[(1E)-amino(benzylamino)methylidene]-6-chloropyrazine-2-carboxamide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
IUPHAR/BPS |
|
| KEGG | |
| MeSH | benzamil |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C13H14ClN7O | |
| Molar mass | 319.75 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Benzamil or benzyl amiloride is a potent blocker of the
analog of amiloride, and is marketed as the hydrochloride salt (benzamil hydrochloride). As amiloride, benzamil has been studied as a possible treatment for cystic fibrosis,[4] although with disappointing results.[5]
Structure
Benzamil is a
benzyl group-containing analog of amiloride. Like amiloride, it is a guanidinium group-containing pyrazine derivative
.
Mechanism of action

Benzamil is closely related to amiloride. By adding the benzyl group to the nitrogen of the guanidinium group the activity is increased several hundredfold.[6]
Amiloride works by directly blocking the epithelial sodium channel (ENaC) thereby inhibiting sodium reabsorption in the distal convoluted tubules and collecting ducts in the kidneys (this mechanism is the same for triamterene). This promotes the loss of sodium and water from the body, but without depleting potassium.
References
- PMID 8898016.
- PMID 7573500.
- ^
Lee, Y. S.; Sayeed, M. M.; Wurster, R. D. (January 6, 1995). "Intracellular Ca2+ mediates the cytotoxicity induced by bepridil and benzamil in human brain tumor cells". Cancer Letters. 88 (1): 87–91. PMID 7850778. Archived from the originalon August 7, 2008. Retrieved 2008-05-01.
- PMID 10543294.
- S2CID 3160146.
- ^
Kleyman, T. R.; Cragoe E. J. Jr. (October 1988). "Amiloride and its analogs as tools in the study of ion transport". J Membr Biol. 105 (1): 1–21. S2CID 21071525.
External links
 Media related to Benzamil at Wikimedia Commons
Media related to Benzamil at Wikimedia Commons
