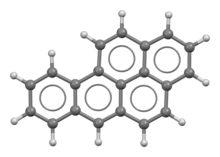
Benzo(a)pyrene
![Benzo[a]pyrene](http://upload.wikimedia.org/wikipedia/commons/thumb/f/fa/Benzo-a-pyrene.svg/220px-Benzo-a-pyrene.svg.png) | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Benzo[pqr]tetraphene[1] | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.000.026 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 3077, 3082 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H12 | |
| Molar mass | 252.316 g·mol−1 |
| Density | 1.24 g/cm3 (25 °C) |
| Melting point | 179[2] °C (354 °F; 452 K) |
| Boiling point | 495 °C (923 °F; 768 K) |
| 0.2 to 6.2 μg/L | |
| -135.7·10−6 cm3/mol | |
| Hazards[3] | |
| GHS labelling: | |
  
| |
| Danger | |
| H317, H340, H350, H360, H410 | |
| P201, P202, P261, P272, P273, P280, P281, P302+P352, P308+P313, P321, P333+P313, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Benzo[a]pyrene (BaP or B[a]P) is a
Description
Benzo[a]pyrene (BaP) is a polycyclic aromatic hydrocarbon found in coal tar with the formula C20H12. The compound is one of the benzopyrenes, formed by a benzene ring fused to pyrene, and is the result of incomplete combustion at temperatures between 300 °C (572 °F) and 600 °C (1,112 °F).
Sources
The main source of atmospheric BaP is residential wood burning.
BaP is discharged in wastewater by industries such as
History
In the 18th century, young British
Toxicity
Nervous system
Immune system
BaP has an effect on the number of
Reproductive system
In experiments with male rats, subchronic exposure to inhaled BaP has been shown to generally reduce the function of testicles and epididymis with lower sex steroid/testosterone production and sperm production.[15]
Carcinogenicity
BaP's metabolites are
In June 2016, BaP was added as benzo[def]chrysene to the REACH Candidate List of Substances of very high concern for Authorisation.[17]
Numerous studies since the 1970s have documented links between BaP and cancers.[18] It has been more difficult to link cancers to specific BaP sources, especially in humans, and difficult to quantify risks posed by various methods of exposure (inhalation or ingestion).[19] A link between vitamin A deficiency and emphysema in smokers was described in 2005 to be due to BaP, which induces vitamin A deficiency in rats.[20]
A 1996 study provided molecular evidence linking components in tobacco smoke to lung cancer. BaP was shown to cause genetic damage in lung cells that was identical to the damage observed in the DNA of most malignant lung tumours.[21]
Regular consumption of
A 2005 NCI study found an increased risk of colorectal adenomas was associated with BaP intake, and more strongly with BaP intake from all foods.[24]The detoxification enzymes cytochrome P450
Interaction with DNA
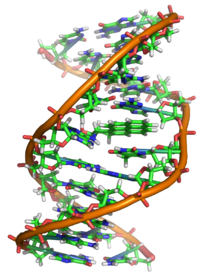
Properly speaking, BaP is a

X-ray crystallographic and nuclear magnetic resonance structure studies have shown how this binding distorts the DNA[28] by confusing the double-helical DNA structure. This disrupts the normal process of copying DNA and causes mutations, which explains the occurrence of cancer after exposure. This mechanism of action is similar to that of aflatoxin which binds to the N7 position of guanine.[29]
There are indications that benzo[a]pyrene diol epoxide specifically targets the protective
Benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide is the carcinogenic product of three enzymatic reactions:[31]
- Benzo[a]pyrene is first oxidized by 1A1 to form a variety of products, including (+)benzo[a]pyrene-7,8-epoxide.[32]
- This product is metabolized by epoxide hydrolase, opening up the epoxide ring to yield (−)benzo[a]pyrene-7,8-dihydrodiol.
- The ultimate carcinogen is formed after another reaction with cytochrome P4501A1 to yield the (+)benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide. It is this diol epoxide that covalently binds to DNA.
BaP induces cytochrome P450 1A1 (CYP1A1) by binding to the AHR (
Moreover, BaP has been found to activate a transposon, LINE1, in humans.[36]
Nucleotide excision repair
As illustrated above, (+)benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide (BPDE) forms bulky covalent DNA adducts with guanines. Most of these adducts can be efficiently eliminated from DNA by the process of nucleotide excision repair.[37] Those adducts that are not removed can cause errors during DNA replication leading to carcinogenic mutations.
See also
- Benzopyrene
- Benzo[e]pyrene
- Pyrene, a four-ring analogue
- Toxification
References
- ISBN 978-0-85404-182-4.
- ISBN 978-1-4987-5429-3.
- ^ "benzo[a]pyrene". pubchem.ncbi.nlm.nih.gov.
- ^ "Assessment of Benzo-alpha-pyrene Emissions in the Great Lakes Region" (PDF). pp. 23–24.
- PMID 11313108.
- S2CID 21302834.
- S2CID 35095622.
- ^ U.S. Environmental Protection Agency (EPA), Washington, D.C. (2002). "Iron and Steel Manufacturing Point Source Category." Code of Federal Regulations, 40 CFR Part 420.
- ^ EPA (1984). "Nonferrous Metals Manufacturing Point Source Category." Code of Federal Regulations, 40 CFR Part 421.
- ^ Pott, Percivall (1775). Chirurgical Observations …. London, England: L. Hawes, W. Clarke, and R. Collins. pp. 63–68. From p. 67: "The disease, in these people [i.e., chimney sweeps], seems to derive its origin from a lodgment of soot in the rugae of the scrotum, … "
- .
- PMID 10413421.
- PMID 18761371..
- PMID 26765097.
- PMID 18499416.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - ^ A review of human carcinogens—part F: chemical agents and related occupations
- ^ European Chemicals Agency. "ED/21/2016". ECHA. Retrieved 21 June 2016.
- ISBN 978-0-08-050576-3.
- PMID 15198916.
- ^ "Benzopyrene and Vitamin A deficiency". Researcher links cigarettes, vitamin A and emphysema. Retrieved March 5, 2005.
- S2CID 3589066.
- PMID 12351160.
- S2CID 23886438.
- S2CID 33819830.
- ^ Data presented by Daniel W. Nebert in research seminars 2007
- ^ Created from PDB 1JDG Archived 2008-09-22 at the Wayback Machine
- ^ )
- PMID 12578353.
- PMID 8042848.
- S2CID 6134471.
- PMID 17702526.
- PMID 8961944.
- ^ PMID 10331078.
- PMID 17997381.
- PMID 22442665.
- PMID 16510580.
- PMID 28607059.
External links
- International Chemical Safety Card 0104
- National Pollutant Inventory – Polycyclic Aromatic Hydrocarbon Fact Sheet
- "Lung cancer as consequence by Benzopyrene in smokers". Lung Cancer. Archived from the original on April 14, 2005. Retrieved March 5, 2005.
- "Levels of Benzopyrene in Burnt toasts". Guardian Unlimited, Special reports: Close encounters. Retrieved March 5, 2005.
- Karle, I. L. (2004). "Crystal and molecular structure of a benzo[a]pyrene-7,8-diol-9,10-epoxide N2-deoxyguanosine adduct: Absolute configuration and conformation". Proceedings of the National Academy of Sciences. 101 (6): 1433–8. PMID 14757823.
