Benzoic acid
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Benzoic acid[1] | |||
| Systematic IUPAC name
Benzenecarboxylic acid | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 3DMet | |||
| 636131 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.000.562 | ||
| EC Number |
| ||
| E number | E210 (preservatives) | ||
| 2946 | |||
| KEGG | |||
| MeSH | benzoic+acid | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C7H6O2 | |||
| Molar mass | 122.123 g/mol | ||
| Appearance | Colorless crystalline solid | ||
| Odor | Faint, pleasant odor | ||
| Density | 1.2659 g/cm3 (15 °C) 1.0749 g/cm3 (130 °C)[2] | ||
| Melting point | 122 °C (252 °F; 395 K)[7] | ||
| Boiling point | 250 °C (482 °F; 523 K)[7] | ||
| 1.7 g/L (0 °C) 2.7 g/L (18 °C) 3.44 g/L (25 °C) 5.51 g/L (40 °C) 21.45 g/L (75 °C) 56.31 g/L (100 °C)[2][3] | |||
| Solubility | Soluble in | ||
| Solubility in methanol | 30 g/100 g (−18 °C) 32.1 g/100 g (−13 °C) 71.5 g/100 g (23 °C)[2] | ||
| Solubility in ethanol | 25.4 g/100 g (−18 °C) 47.1 g/100 g (15 °C) 52.4 g/100 g (19.2 °C) 55.9 g/100 g (23 °C)[2] | ||
| Solubility in acetone | 54.2 g/100 g (20 °C)[2] | ||
| Solubility in olive oil | 4.22 g/100 g (25 °C)[2] | ||
1,4-dioxane
|
55.3 g/100 g (25 °C)[2] | ||
| log P | 1.87 | ||
| Vapor pressure | 0.16 Pa (25 °C) 0.19 kPa (100 °C) 22.6 kPa (200 °C)[4] | ||
| Acidity (pKa) | |||
| −70.28·10−6 cm3/mol | |||
Refractive index (nD)
|
1.5397 (20 °C) 1.504 (132 °C)[2] | ||
| Viscosity | 1.26 mPa (130 °C) | ||
| Structure | |||
Monoclinic
| |||
Planar
| |||
| 1.72 dioxane
| |||
| Thermochemistry | |||
Heat capacity (C)
|
146.7 J/mol·K[4] | ||
Std molar
entropy (S⦵298) |
167.6 J/mol·K[2] | ||
Std enthalpy of (ΔfH⦵298)formation |
−385.2 kJ/mol[2] | ||
Std enthalpy of (ΔcH⦵298)combustion |
−3228 kJ/mol[4] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Irritant | ||
| GHS labelling: | |||
  [8] [8]
| |||
| Danger | |||
| H318, H335[8] | |||
| P261, P280, P305+P351+P338[8] | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 121.5 °C (250.7 °F; 394.6 K)[7] | ||
| 571 °C (1,060 °F; 844 K)[7] | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
1700 mg/kg (rat, oral) | ||
| Safety data sheet (SDS) | JT Baker | ||
| Related compounds | |||
Other cations
|
Sodium benzoate, Potassium benzoate | ||
Related carboxylic acids
|
Hydroxybenzoic acids Aminobenzoic acids, Nitrobenzoic acids, Phenylacetic acid | ||
Related compounds
|
Benzaldehyde, Benzyl alcohol, Benzoyl chloride, Benzylamine, Benzamide, Benzonitrile | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Benzoic acid
Benzoic acid occurs naturally in many plants
History
Benzoic acid was discovered in the sixteenth century. The
Justus von Liebig and Friedrich Wöhler determined the composition of benzoic acid.[11] These latter also investigated how hippuric acid is related to benzoic acid.
In 1875 Salkowski discovered the
Production
Industrial preparations
Benzoic acid is produced commercially by
The first industrial process involved the reaction of
Laboratory synthesis
Benzoic acid is cheap and readily available, so the laboratory synthesis of benzoic acid is mainly practiced for its pedagogical value. It is a common undergraduate preparation.
Benzoic acid can be purified by recrystallization from water because of its high solubility in hot water and poor solubility in cold water. The avoidance of organic solvents for the recrystallization makes this experiment particularly safe. This process usually gives a yield of around 65%.[14]
By hydrolysis
Like other nitriles and amides, benzonitrile and benzamide can be hydrolyzed to benzoic acid or its conjugate base in acid or basic conditions.
From Grignard reagent
Bromobenzene can be converted to benzoic acid by "carboxylation" of the intermediate phenylmagnesium bromide.[15] This synthesis offers a convenient exercise for students to carry out a Grignard reaction, an important class of carbon–carbon bond forming reaction in organic chemistry.[16][17][18][19][20]
Oxidation of benzyl compounds
Benzyl alcohol[21] and benzyl chloride and virtually all benzyl derivatives are readily oxidized to benzoic acid.
Uses
Benzoic acid is mainly consumed in the production of phenol by oxidative decarboxylation at 300−400 °C:[22]
The temperature required can be lowered to 200 °C by the addition of catalytic amounts of copper(II) salts. The phenol can be converted to cyclohexanol, which is a starting material for nylon synthesis.
Precursor to plasticizers
Benzoate
Benzoic acid and its salts are used as
Typical concentrations of benzoic acid as a preservative in food are between 0.05 and 0.1%. Foods in which benzoic acid may be used and maximum levels for its application are controlled by local food laws.[25][26]
Concern has been expressed that benzoic acid and its salts may react with
Medicinal
Benzoic acid is a constituent of
Benzoic acid was used as an
Niche and laboratory uses
In teaching laboratories, benzoic acid is a common standard for calibrating a
Biology and health effects
Benzoic acid occurs naturally as do its esters in many plant and animal species. Appreciable amounts are found in most berries (around 0.05%). Ripe fruits of several
In terms of its biosynthesis, benzoate is produced in plants from cinnamic acid.
Reactions
Reactions of benzoic acid can occur at either the
Aromatic ring
Carboxyl group
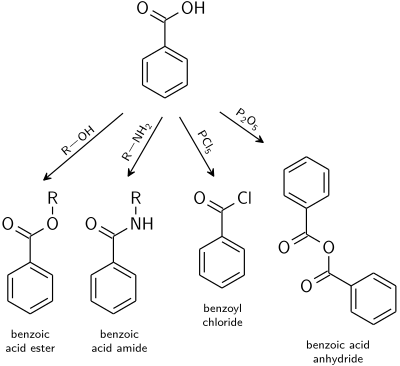
Reactions typical for carboxylic acids apply also to benzoic acid.[22]
- Benzoate esters are the product of the acid catalysed reaction with alcohols.
- Benzoic acid amides are usually prepared from benzoyl chloride.
- Dehydration to benzoic anhydride is induced with acetic anhydride or phosphorus pentoxide.
- Highly reactive acid derivatives such as phosphorus chlorides or thionyl chloride.
- Orthoesters can be obtained by the reaction of alcohols under acidic water free conditions with benzonitrile.
- Reduction to DIBAL-H, LiAlH4 or sodium borohydride.
- Hunsdiecker decarboxylationcan be achieved by heating the silver salt.
Safety and mammalian metabolism
It is excreted as
For humans, the World Health Organization's International Programme on Chemical Safety (IPCS) suggests a provisional tolerable intake would be 5 mg/kg body weight per day.[32] Cats have a significantly lower tolerance against benzoic acid and its salts than rats and mice. Lethal dose for cats can be as low as 300 mg/kg body weight.[41] The oral LD50 for rats is 3040 mg/kg, for mice it is 1940–2263 mg/kg.[32]
In Taipei, Taiwan, a city health survey in 2010 found that 30% of dried and pickled food products had benzoic acid.[42]
See also
- Niacin – Organic compound and a form of vitamin B3
References
- ISBN 978-0-85404-182-4.
- ^ a b c d e f g h i j "benzoic acid". chemister.ru. Retrieved 24 October 2018.
- ^ Seidell, Atherton; Linke, William F. (1952). Solubilities of Inorganic and Organic Compounds. Van Nostrand.
- ^ a b c Benzoic acid in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved 2014-05-23)
- ISBN 9781429254366.
- .
- ^ a b c d Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ a b c Sigma-Aldrich Co., Benzoic acid. Retrieved on 2014-05-23.
- ^ "Scientists uncover last steps for benzoic acid creation in plants". Purdue Agriculture News.
- OCLC 50969944.
- hdl:2027/hvd.hxdg3f.
- ^ Salkowski E (1875). Berl Klin Wochenschr. 12: 297–298.
{{cite journal}}: Missing or empty|title=(help) - ISBN 978-1-292-02165-2.
- ISBN 978-0-08-034715-8.
- ISBN 978-0-534-40833-6.
- Org. React.8: 28–58.
- ISBN 978-0-08-052349-1.
- ^ "The Grignard Reaction. Preparation of Benzoic Acid" (PDF). Portland Community College. Archived from the original (PDF) on 26 February 2015. Retrieved 12 March 2015.>
- ^ "Experiment 9: Synthesis of Benzoic Acid via Carbonylation of a Grignard Reagent" (PDF). University of Wisconsin-Madison. Archived from the original (PDF) on 23 September 2015. Retrieved 12 March 2015.
- ^ "Experiment 3: Preparation of Benzoic Acid" (PDF). Towson University. Archived from the original (PDF) on 13 April 2015. Retrieved 12 March 2015.>
- ISSN 1083-6160.
- ^ ISBN 978-3527306732..
- PMID 1785916.
- .
- ^ GSFA Online Food Additive Group Details: Benzoates (2006) Archived 26 September 2007 at the Wayback Machine
- ^ EUROPEAN PARLIAMENT AND COUNCIL DIRECTIVE No 95/2/EC of 20 February 1995 on food additives other than colours and sweeteners (Consleg-versions do not contain the latest changes in a law) Archived 19 April 2003 at the Wayback Machine
- ^ "Indications of the possible formation of benzene from benzoic acid in foods, BfR Expert Opinion No. 013/2006" (PDF). German Federal Institute for Risk Assessment. 1 December 2005. Archived (PDF) from the original on 26 April 2006. Retrieved 30 March 2022.
- ^ "Whitfield Ointment". Archived from the original on 9 October 2007. Retrieved 15 October 2007.
- ISBN 978-0-7817-3481-3.
- ^ Lillard, Benjamin (1919). "Troches of Benzoic Acid". Practical Druggist and Pharmaceutical Review of Reviews.
- ^ Experiment 2: Using Bomb Calorimetry to Determine the Resonance Energy of Benzene Archived 9 March 2012 at the Wayback Machine
- ^ a b c "Concise International Chemical Assessment Document 26: BENZOIC ACID AND SODIUM BENZOATE".
- PMID 5012256.
- PMID 20035037.
- PMID 15653882.
- ^ Brewster, R. Q.; Williams, B.; Phillips, R. (1955). "3,5-Dinitrobenzoic Acid". Organic Syntheses; Collected Volumes, vol. 3, p. 337.
- S2CID 13639993.
- ^ "butyrate-CoA ligase". BRENDA. Technische Universität Braunschweig. Retrieved 7 May 2014. Substrate/Product
- ^ "glycine N-acyltransferase". BRENDA. Technische Universität Braunschweig. Retrieved 7 May 2014. Substrate/Product
- PMID 6226283.
- S2CID 2553612.
- ^ Chen, Jian; Y.L. Kao (18 January 2010). "Nearly 30% dried, pickled foods fail safety inspections". The China Post.
External links
- International Chemical Safety Card 0103
- SIDS Initial Assessment Report for Benzoic Acid from the Organisation for Economic Co-operation and Development(OECD)
- ChemicalLand