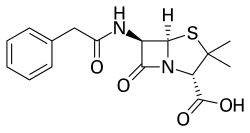
Benzylpenicillin
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Pfizerpen, other |
| Other names | Penicillin G potassium,[2] penicillin G sodium |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a685013 |
| Pregnancy category |
|
QJ51CE01 (WHO) | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 30% oral [3] |
| Protein binding | 60% |
| Metabolism | Liver |
| Elimination half-life | 30 min |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Benzylpenicillin, also known as penicillin G (PenG
Side effects include
Benzylpenicillin is on the World Health Organization's List of Essential Medicines.[8]
Medical uses
Antimicrobial potency
As an antibiotic, benzylpenicillin is noted to possess effectiveness mainly against gram-positive organisms. Some gram-negative organisms such as Neisseria gonorrhoeae and Leptospira weilii are also reported to be susceptible to benzylpenicillin.[9]
Adverse effects
Adverse effects can include hypersensitivity reactions including urticaria, fever, joint pains, rashes, angioedema, anaphylaxis, serum sickness-like reaction. Rarely central nervous system toxicity including convulsions (especially with high doses or in severe renal impairment), interstitial nephritis, haemolytic anaemia, leucopenia, thrombocytopenia, and coagulation disorders. Also reported diarrhoea (including antibiotic-associated colitis). Benzylpenicillin has relatively low toxicity, except for in the nervous system, in which it is one of the most active drugs among β-lactam agents.[7] In addition, benzylpenicillin is an irritant, a health hazard, and an environmental hazard.[10]
Benzylpenicillin serum concentrations can be monitored either by traditional microbiological assay or by more modern chromatographic techniques. Such measurements can be useful to avoid central nervous system toxicity in any person receiving large doses of the drug on a chronic basis, but they are especially relevant to patients with kidney failure, who may accumulate the drug due to reduced urinary excretion rates.[11][12]
Manufacture
Benzylpenicillin is produced by fermentation of Penicillium chrysogenum.[10] The production of benzylpenicillin involves fermentation, recovery and purification of the penicillin.[13]
The fermentation process of the production of benzylpenicillin creates the product. The presence of the product in solution inhibits the reaction and reduces the product rate and yield. Thus, in order to obtain the most product and increase the rate of reaction, it is continuously extracted.[14] This is done by mixing the mold with either glucose, sucrose, lactose, starch, or dextrin, nitrate, ammonium salt, corn steep liquor, peptone, meat or yeast extract, and small amounts of inorganic salts.[15]
The recovery of the benzylpenicillin is the most important part of the production process because it affects the later purification steps if done incorrectly.[13] There are several techniques used to recover benzylpenicillin: aqueous two-phase extraction, liquid membrane extraction, microfiltration, and solvent extraction.[13] Extraction is more commonly used in the recovery process.[medical citation needed]
In the purification step, the benzylpenicillin is separated from the extraction solution. This is normally done by using a separation column.[16]
Synonyms
References
- PMID 565366.
- ^ a b "Penicillin G Injection - FDA prescribing information, side effects and uses". www.drugs.com. Archived from the original on 20 December 2016. Retrieved 10 December 2016.
- PMID 32119447. Retrieved 7 December 2023.
- ISBN 978-0-323-06947-2.
Natural penicillin (PenG), penicillinase-resistant penicillin (methicillin), extended-spectrum penicillin (amoxicillin), and broad-spectrum penicillin (carbenicillin) all have the same core β-lactam ring, which is essential for antimicrobial activity.
- ^ "Australian Product Information – BENPEN" (PDF). Seqirus Pty Ltd.
- ^ ISBN 9789241547659.
- ^ a b Castle SS (2007). "Penicillin G". In Enna SJ, Bylund DB (eds.). xPharm: The Comprehensive Pharmacology Reference. New York: Elsevier. pp. 1–6.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Penicillin G" (PDF). Toku-E. 10 October 2010. Archived from the original (PDF) on 3 March 2016. Retrieved 11 June 2012.
- ^ a b c "Benzylpenicillin". Molecule of the Week. American Chemical Society. Retrieved 9 November 2022.
- S2CID 40884225.
- ^ Baselt R (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. pp. 1195–1196.
- ^ .
- ^ Barros J (4 January 2016). "Use Extraction to Improve Penicillin G Recovery". Discover Chemistry. American Chemical Society. Archived from the original on 6 May 2019. Retrieved 6 May 2019.
- ^ "Separation and Purification of Pharmaceuticals and Antibiotics" (PDF). Mitsubishi Chemical Corporation. pp. 312–324.
- PMID 6983856.
- PMID 20259048.
- ^ "Penicillin G". PubChem. National Center for Biotechnology Information, National Library of Medicine. Retrieved 26 December 2020.
