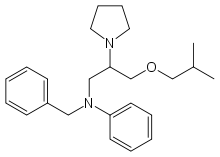
Bepridil
 | |
| Clinical data | |
|---|---|
| Trade names | Vascor |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699051 |
| Routes of administration | Oral |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | Well absorbed |
| Protein binding | 99% |
| Metabolism | Hepatic, CYP3A4-mediated |
| Elimination half-life | 42 hours |
| Excretion | Renal |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Bepridil (trade name Vascor) is an
angina pectoris
. It is no longer sold in the United States.
It is nonselective.[1]
It has been discussed as a possible option in the treatment of atrial fibrillation.[2]
It has been implicated in causing ventricular arrhythmia (torsades de pointes).
Ebola research
In June 2015 a research paper [3] was published finding bepridil to result in a 100% survival rate for mice exposed to ebola during an experiment searching for potential pharmaceutical ebola treatments; indicating its potential use in future ebola research and therapy.[4]
SARS-CoV-2 research
A research paper [5] showed that Bepridil inhibited cytopathogenic effects induced by SARS-CoV-2 in Vero E6 cells and in A549 cells in an in vitro assay.
