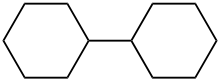
Bicyclohexyl
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
1,1′-Bi(cyclohexane) | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.001.966 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H22 | |
| Molar mass | 166.308 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 0.88273 g/cm3 |
| Melting point | 4 °C (39 °F; 277 K) |
| Insoluble | |
| Solubility in other solvents | Miscible with organic solvents |
Refractive index (nD)
|
1.4796[1] |
| Hazards | |
| GHS labelling: | |
 
| |
| Warning | |
| H315, H319, H410 | |
| P264, P273, P280, P302+P352, P305+P351+P338, P321, P332+P313, P337+P313, P362, P391, P501 | |
| Flash point | 92 °C (198 °F; 365 K) |
| 245 °C (473 °F; 518 K) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Bicyclohexyl, also known as dicyclohexyl or bicyclohexane, is an organic chemical with the formula C12H22 and a molecular mass of 166.303 g mol−1. It is a nonvolatile liquid at room temperature, with a boiling point of 227 °C (441 °F). Its structure consists of two cyclohexane rings joined by a single carbon-carbon bond.
Production
Carbazole can be denitrogenated by hydrogen to yield bicyclohexyl as the main product.[2]
When cyclohexane is exposed to radiation, bicyclohexyl is produced among other hydrocarbons.[3]
Properties
The molecule is not completely flat, and the two rings are twisted compared to each other. Liquid bicyclohexyl contains a mixture of molecules with C2 and C2h symmetry termed ee anti, and ee gauche. The carbon-carbon bond (pivot) between the rings is 1.55 Å, and the carbon-carbon length in the rings is 1.535 Å and carbon-hydrogen bond length is 1.102 Å. The torsion angle between the rings is 74.9°. The C-C-C bond angle ∠ is about 111° and C-C-H angle is 109°.[4]
The
When bicyclohexyl is heated to around 427 °C (801 °F) it slowly decomposes to cyclohexane and cyclohexene, as the pivot bond joining the two rings is the longest and weakest one.[6]
Heat of combustion is 1814.8 kcal/mol.[7]
Use
Bicyclohexyl has uses in organic synthesis as a building block and structural motif, in studying the chemistry of liquid interfaces,[8] and in surface modification of metal oxides as a solvent.[9]
See also
References
- ISBN 9781483272856.
- ISBN 9780824797560.
- .
- .
- .
- .
- .
- .
- PMID 24849332.
