Bioinorganic chemistry
Bioinorganic chemistry is a field that examines the role of
As a mix of biochemistry and inorganic chemistry, bioinorganic chemistry is important in elucidating the implications of electron-transfer proteins, substrate bindings and activation, atom and group transfer chemistry as well as metal properties in biological chemistry. The successful development of truly interdisciplinary work is necessary to advance bioinorganic chemistry.[2]
Composition of living organisms
About 99% of
.History
Themes in bioinorganic chemistry
Several distinct systems are of identifiable in bioinorganic chemistry. Major areas include:
Metal ion transport and storage
A diverse collection of
Enzymology
Many reactions in life sciences involve water and metal ions are often at the catalytic centers (active sites) for these enzymes, i.e. these are metalloproteins. Often the reacting water is a ligand (see metal aquo complex). Examples of hydrolase enzymes are carbonic anhydrase, metallophosphatases, and metalloproteinases. Bioinorganic chemists seek to understand and replicate the function of these metalloproteins.
Metal-containing electron transfer proteins are also common. They can be organized into three major classes:
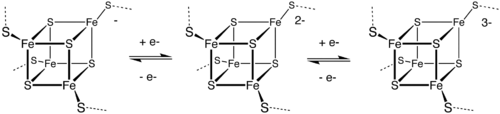
Toxicity
Several metal ions are toxic to humans and other animals. The bioinorganic chemistry of lead in the context of its toxicity has been reviewed.[4]
Oxygen transport and activation proteins
Aerobic life make extensive use of metals such as iron, copper, and manganese.

Bioorganometallic chemistry
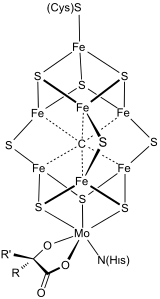
Metals in medicine
A number of drugs contain metals. This theme relies on the study of the design and mechanism of action of metal-containing pharmaceuticals, and compounds that interact with endogenous metal ions in enzyme active sites. The most widely used anti-cancer drug is
Environmental chemistry
Environmental chemistry traditionally emphasizes the interaction of heavy metals with organisms.
Biomineralization
Biomineralization is the process by which living organisms produce
Types of inorganic substances in biology
Alkali and alkaline earth metals
The abundant inorganic elements act as
Transition metals
The transition metals are usually present as trace elements in organisms, with zinc and iron being most abundant.[18][19][20] These metals are used as protein cofactors and signalling molecules. Many are essential for the activity of enzymes such as catalase and oxygen-carrier proteins such as hemoglobin.[21] These cofactors are tightly to a specific protein; although enzyme cofactors can be modified during catalysis, cofactors always return to their original state after catalysis has taken place. The metal micronutrients are taken up into organisms by specific transporters and bound to storage proteins such as ferritin or metallothionein when not being used.[22][23] Cobalt is essential for the functioning of vitamin B12.[24]
Main group compounds
Many other elements aside from metals are bio-active. Sulfur and phosphorus are required for all life. Phosphorus almost exclusively exists as phosphate and its various esters. Sulfur exists in a variety of oxidation states, ranging from sulfate (SO42−) down to sulfide (S2−). Selenium is a trace element involved in proteins that are antioxidants. Cadmium is important because of its toxicity.[25]
See also
References
- ISBN 0-935702-72-5
- PMID 33787237.
- ^ PMID 1872381.
- PMID 28731294.
- ISBN 978-1-84755-177-1.
- PMID 29227923.
- ISBN 978-0-470-03525-2.
- ISBN 978-0-19-504977-0.
- ISBN 978-0-521-87473-1.
- doi:10.1177/11.6.799.
- PMID 15295599.
- PMID 8312724.
- PMID 17845714.
- ISBN 978-0-08-037941-8.
- PMID 15119939.
- PMID 2452594.
- PMID 16922804.
- ISSN 1868-0402
- PMID 9498359. Archived from the originalon 2011-04-30.
- PMID 14551660.
- S2CID 14863354.
- PMID 16793761.
- PMID 17194590.
- ISSN 1868-0402
- ^ Maret, Wolfgang; Moulis, Jean-Marc (2013). "Chapter 1. The Bioinorganic Chemistry of Cadmium in the Context of its Toxicity". In Astrid Sigel, Helmut Sigel and Roland K. O. Sigel (ed.). Cadmium: From Toxicology to Essentiality. Metal Ions in Life Sciences. Vol. 11. Springer. pp. 1–30.
Literature
- Heinz-Bernhard Kraatz (editor), Nils Metzler-Nolte (editor), Concepts and Models in Bioinorganic Chemistry, John Wiley and Sons, 2006, ISBN 3-527-31305-2
- Ivano Bertini, Harry B. Gray, Edward I. Stiefel, Joan Selverstone Valentine, Biological Inorganic Chemistry, University Science Books, 2007, ISBN 1-891389-43-2
- Wolfgang Kaim, Brigitte Schwederski "Bioinorganic Chemistry: Inorganic Elements in the Chemistry of Life." John Wiley and Sons, 1994, ISBN 0-471-94369-X
- Rosette M. Roat-Malone, Bioinorganic Chemistry : A Short Course, ISBN 0-471-15976-X
- J.J.R. Fraústo da Silva and R.J.P. Williams, The biological chemistry of the elements: The inorganic chemistry of life, 2nd Edition, ISBN 0-19-850848-4
- Lawrence Que, Jr., ed., Physical Methods in Bioinorganic Chemistry, University Science Books, 2000, ISBN 1-891389-02-5
External links
- The Society of Biological Inorganic Chemistry (SBIC)'s home page
- The French Bioinorganic Chemistry Society
- Glossary of Terms in Bioinorganic Chemistry
- Metal Coordination Groups in Proteins by Marjorie Harding
- European Bioinformatics Institute
- MetalPDB: A database of metal sites in biomolecular structures
