Bleomycin
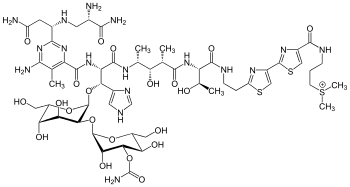 Bleomycin A2 | |
| Clinical data | |
|---|---|
| Trade names | Blenoxane |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682125 |
| License data | |
| Pregnancy category |
|
subcutaneous, intrapleural[2] | |
| Drug class | Glycopeptide antibiotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100% and 70% following intramuscular and subcutaneous administrations, respectively, and 45% following both intraperitoneal and intrapleural administrations[2] |
| Elimination half-life | two hours[2] |
| Excretion | Kidney (60–70%)[2] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Bleomycin is a medication used to treat
Common
Bleomycin was discovered in 1962.
Medical uses
Cancer
Bleomycin is mostly used to treat
Other uses
It may also be put inside the chest to help prevent the recurrence of a pleural effusion due to cancer.[6] However, for scarring down the pleura, talc appears to be the better option although indwelling pleural catheters are at least as effective in reducing the symptoms of an effusion(such as dyspnea).[11][12]
While potentially effective against bacterial infections, its toxicity prevents its use for this purpose.[6] It has been studied in the treatment of warts but is of unclear benefit.[13]
Side effects
The most common
Lifetime cumulative dose
Bleomycin should not exceed a lifetime cumulative dose greater than 400 units.[18] Pulmonary toxicities, most commonly presenting as pulmonary fibrosis, are associated with doses of bleomycin greater than 400 units.[18]
Mechanism of action
Bleomycin acts by induction of
Biosynthesis
Bleomycin is a
The biosynthesis of the bleomycin aglycon can be visualized in three stages:[citation needed]
- NRPS-mediated formation of P-3A from Ser, Asn, His, and Ala
- PKS-mediated elongation of P-3A by malonyl CoA and AdoMet to yield P-4
- NRPS-mediated elongation of P-4 by Thr to P-5 that is further elongated by β-Ala, Cys, and Cys to get P-6m.
On the basis of the bleomycin structure and the deduced functions of individual NRPS and PKS domains and modules, a linear model for the bleomycin megasynthetase-templated assembly of the bleomycin peptide/polyketide/peptide aglycon was proposed from nine amino acids and one acetate.[citation needed]
Biosynthesis of bleomycin is completed by glycosylation of the aglycones. Bleomycin naturally occurring-analogues have two to three sugar molecules, and DNA cleavage activities of these analogues have been assessed,[21][22] primarily by the plasmid relaxation and break light assays.
History
Bleomycin was first discovered in 1962 when the Japanese scientist
Research
Bleomycin is used in research to induce pulmonary fibrosis in mice.[24]
See also
- Flagellate pigmentation from bleomycin
- Pingyangmycin (Bleomycin A5)
References
- ^ "Bleomycin Use During Pregnancy". Drugs.com. 9 August 2019. Retrieved 16 February 2020.
- ^ a b c d "Bleomycin- bleomycin sulfate injection, powder, lyophilized, for solution". DailyMed. 31 December 2019. Retrieved 16 February 2020.
- FDA. Retrieved 22 October 2023.
- ^ "Bleo-Kyowa Powder for solution for injection - Summary of Product Characteristics (SmPC)". (emc). 31 August 2018. Archived from the original on 16 February 2020. Retrieved 16 February 2020.
- ^ "Bleomycin". European Medicines Agency (EMA).
- ^ a b c d e f g h i j k l m n o p q r "Bleomycin Sulfate". The American Society of Health-System Pharmacists. Archived from the original on 8 September 2015. Retrieved 1 August 2015.
- PMID 32315458.
- ISBN 9780471899792. Archivedfrom the original on 5 March 2016.
- ISBN 9780429774546.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- |intentional=yes}}.)
- PMID 29634827.
- PMID 22972052.
- ^ Thompson M. "Bleomycin and Anaesthesia" (PDF). Anaesthesia Western Australia. Archived from the original (PDF) on 8 September 2017. Retrieved 8 September 2017.
- PMID 19265174.
- PMID 12735422.
- S2CID 12671326.
- ^ a b "bleomycin [TUSOM | Pharmwiki]". tmedweb.tulane.edu. Retrieved 2 February 2022.
- ^ Takimoto CH, Calvo E. "Principles of Oncologic Pharmacotherapy" Archived 15 May 2009 at the Wayback Machine in Pazdur R, Wagman LD, Camphausen KA, Hoskins WJ (Eds) Cancer Management: A Multidisciplinary Approach Archived 4 October 2013 at the Wayback Machine. 11 ed. 2008.
- PMID 10650103.
- PMID 28256838.
- S2CID 33531845.
- PMID 5953301.
- PMID 26289670.
In our studies, mice developed classic PF with structural alteration of the lung following intravenous bleomycin treatment
Further reading
- Claussen CA, Long EC (September 1999). "Nucleic Acid recognition by metal complexes of bleomycin". Chemical Reviews. 99 (9): 2797–2816. PMID 11749501.
- Shen B, Du L, Sanchez C, Edwards DJ, Chen M, Murrell JM (December 2001). "The biosynthetic gene cluster for the anticancer drug bleomycin from Streptomyces verticillus ATCC15003 as a model for hybrid peptide-polyketide natural product biosynthesis". Journal of Industrial Microbiology & Biotechnology. 27 (6): 378–385. S2CID 3022217.
