Boekelheide reaction
| Boekelheide reaction | |
|---|---|
| Named after | Virgil Carl Boekelheide |
| Reaction type | Rearrangement reaction |
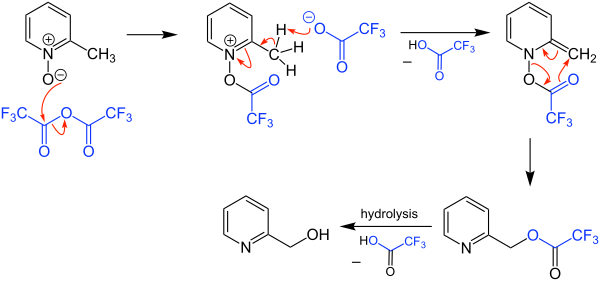
The Boekelheide reaction is a rearrangement of α-picoline-N-oxides to hydroxymethylpyridines. It is named after Virgil Boekelheide who first reported it in 1954.[1] Originally the reaction was carried out using acetic anhydride, which typically required a period at reflux (~140 °C). The reaction can be performed using trifluoroacetic anhydride (TFAA), which often allows for a room temperature reaction.[2]

Mechanism
The mechanism of the Boekelheide reaction begins by an acyl transfer from the trifluoroacetic anhydride to the sigmatropic rearrangement which furnishes the trifluoroacetylated methylpyridine. Hydrolysis of the trifluoroacetate releases the hydroxymethylpyridine.
Wikimedia Commons has media related to Boekelheide reaction.
References
- .
- .
