Bone marrow
| Bone marrow | |
|---|---|
| Identifiers | |
| Latin | medulla ossium |
| MeSH | D001853 |
| TA98 | A13.1.01.001 |
| TA2 | 388 |
| FMA | 9608 |
| Anatomical terminology] | |
Bone marrow is a semi-solid
Human marrow produces approximately 500 billion blood cells per day, which join the
Bone marrow transplants can be conducted to treat severe diseases of the bone marrow, including certain forms of cancer such as leukemia. Several types of stem cells are related to bone marrow. Hematopoietic stem cells in the bone marrow can give rise to hematopoietic lineage cells, and mesenchymal stem cells, which can be isolated from the primary culture of bone marrow stroma, can give rise to bone, adipose, and cartilage tissue.[7]
Structure
The composition of marrow is dynamic, as the mixture of cellular and non-cellular components (connective tissue) shifts with age and in response to systemic factors. In humans, marrow is colloquially characterized as "red" or "yellow" marrow (
Hematopoietic components
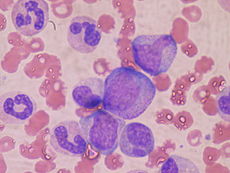
At the cellular level, the main functional component of bone marrow includes the progenitor cells which are destined to mature into blood and lymphoid cells. Human marrow produces approximately 500 billion blood cells per day.
| Group | Cell type | Average fraction |
Reference range |
|---|---|---|---|
| Myelopoietic cells |
Myeloblasts | 0.9 | 0.2–1.5 |
| Promyelocytes | 3.3% | 2.1–4.1 | |
Neutrophilic myelocytes |
12.7% | 8.2–15.7 | |
Eosinophilic myelocytes |
0.8% | 0.2–1.3 | |
Neutrophilic metamyelocytes |
15.9% | 9.6–24.6 | |
Eosinophilic metamyelocytes |
1.2% | 0.4–2.2 | |
Neutrophilic band cells |
12.4% | 9.5–15.3 | |
Eosinophilic band cells |
0.9% | 0.2–2.4 | |
Segmented neutrophils |
7.4% | 6.0–12.0 | |
Segmented eosinophils |
0.5% | 0.0–1.3 | |
Segmented basophils and mast cells |
0.1% | 0.0–0.2 | |
| Erythropoietic cells |
Pronormoblasts |
0.6% | 0.2–1.3 |
Basophilic normoblasts |
1.4% | 0.5–2.4 | |
Polychromatic normoblasts |
21.6% | 17.9–29.2 | |
Orthochromatic normoblast |
2.0% | 0.4–4.6 | |
| Other cell types |
Megakaryocytes | < 0.1% | 0.0-0.4 |
| Plasma cells | 1.3% | 0.4-3.9 | |
| Reticular cells | 0.3% | 0.0-0.9 | |
| Lymphocytes | 16.2% | 11.1-23.2 | |
| Monocytes | 0.3% | 0.0-0.8 |
Stroma
The
- fibroblasts (reticular connective tissue)
- macrophages, which contribute especially to red blood cell production, as they deliver iron for hemoglobinproduction.
- adipocytes(fat cells)
- osteoblasts(synthesize bone)
- osteoclasts(resorb bone)
- sinusoids. These derive from endothelial stem cells, which are also present in the bone marrow.[11]
Function
Central hematopoietic and antigen-responsive organ
That bone marrow is a priming site for T-cell responses to blood-borne antigens was first described in 2003.[13] Mature circulating naïve T cells home to bone marrow sinuses after they have passed through arteries and arterioles.[14] They transmigrate sinus endothelium and enter the parenchyma which contains dendritic cells (DCs). These have a capacity of antigen uptake, processing, and presentation.[13] Cognate interactions between antigen-specific T cells and antigen-presenting DCs (APCs) in parenchyma lead to rapid T-APC cluster formation followed by T cell activation, T cell proliferation and T cell re-circulation to blood.[13] These findings were corroborated and extended in 2013 by in situ two-photon dynamic imaging of mice skulls.[15]
Importance for storage and long-term survival of memory B and memory T cells
Bone marrow is a nest for migratory memory T cells[16] and a sanctuary for plasma cells.[17] This has implications for adaptive immunity and vaccinology.[17] Memory B and T cells persist in the parenchyma in dedicated survival niches organized by stromal cells.[18] This memory can be maintained over long time periods in the form of quiescent cells[18] or by repeated antigenic restimulation.[19] Bone marrow protects and optimizes immunological memory during dietary restriction.[20] In cancer patients, cancer-reactive memory T cells can arise in bone marrow spontaneously or after specific vaccination.[21] Bone marrow is a center of a variety of immune activities: i) hematopoiesis, ii) osteogenesis, iii) immune responses, iv) distinction between self and non-self antigens, v) central immune regulatory function, vi) storage of memory cells, vii) immune surveillance of the central nervous system, viii) adaptation to energy crisis, ix) provision of mesenchymal stem cells for tissue repair. [22]
Mesenchymal stem cells
The bone marrow stroma contains
Bone marrow barrier
The blood vessels of the bone marrow constitute a barrier, inhibiting immature blood cells from leaving the marrow. Only mature blood cells contain the membrane proteins, such as aquaporin and glycophorin, that are required to attach to and pass the blood vessel endothelium.[23] Hematopoietic stem cells may also cross the bone marrow barrier, and may thus be harvested from blood.[citation needed]
Lymphatic role
The red bone marrow is a key element of the
Compartmentalization
As food
People have used animal bone-marrow in cuisine worldwide for millennia, as in the famed Milanese Ossobuco.[25]
Clinical significance
Disease
The normal bone marrow architecture can be damaged or displaced by
To diagnose diseases involving the bone marrow, a
Application of stem cells in therapeutics
Bone marrow derived stem cells have a wide array of application in regenerative medicine.[28]
Imaging
Medical imaging may provide a limited amount of information regarding bone marrow.
When "yellow" fatty marrow becomes replaced by tissue with more cellular composition, this change is apparent as decreased brightness on T1-weighted sequences. Both normal "red" marrow and pathologic marrow lesions (such as cancer) are darker than "yellow" marrow on T1-weight sequences, although can often be distinguished by comparison with the MR signal intensity of adjacent soft tissues. Normal "red" marrow is typically equivalent or brighter than skeletal muscle or intervertebral disc on T1-weighted sequences.[8][31]
Fatty marrow change, the inverse of red marrow
Histology

Bone marrow examination is the pathologic analysis of samples of bone marrow obtained via biopsy and bone marrow aspiration. Bone marrow examination is used in the diagnosis of a number of conditions, including leukemia, multiple myeloma, anemia, and pancytopenia. The bone marrow produces the cellular elements of the blood, including platelets, red blood cells and white blood cells. While much information can be gleaned by testing the blood itself (drawn from a vein by phlebotomy), it is sometimes necessary to examine the source of the blood cells in the bone marrow to obtain more information on hematopoiesis; this is the role of bone marrow aspiration and biopsy.[citation needed]
The ratio between
Donation and transplantation
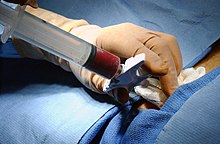
In a
Bone marrow stem cells can be induced to become neural cells to treat neurological illnesses,
Harvesting
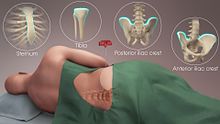
The stem cells are typically harvested directly from the red marrow in the
Another option is to administer certain drugs that stimulate the release of stem cells from the bone marrow into circulating blood.
Persistent viruses
Using quantitative Polymerase Chain Reaction (qPCR) and Next-generation Sequencing (NGS) a maximum of five DNA viruses per individual have been identified. Included were several herpesviruses, hepatitis B virus, Merkel cell polyomavirus, and human papillomavirus 31. Given the reactivation and/or oncogenic potential of these viruses, their repercussion on hematopoietic and malignant disorders calls for further studies.[44]
Fossil record

The earliest fossilised evidence of bone marrow was discovered in 2014 in
See also
- LOC100272216 protein
- Myelonecrosis
- National Marrow Donor Program
- Gift of Life Marrow Registry
- List of distinct cell types in the adult human body
References
- ^ Schmidt, Richard F.; Lang, Florian; Heckmann, Manfred (30 November 2010). What are the organs of the immune system?. Institute for Quality and Efficiency in Health Care. pp. 3/7.
- )
- ^ Arikan, Hüseyin; Çiçek, Kerim (2014). "Haematology of amphibians and reptiles: a review" (PDF). North-Western Journal of Zoology. 10: 190–209.
- ^ Katherine, Abel (2013). Official CPC Certification Study Guide. American Medical Association.
- S2CID 9755621.
- ^ PMID 27015419.
- ISBN 9780323548038.
- ^ PMID 27726743.
- PMID 8249729.
- PMID 29296956.
- ^ ISBN 978-0-7817-9516-6.
- ^ Appendix A:IV in Wintrobe's clinical hematology (9th edition). Philadelphia: Lea & Febiger (1993).
- ^ a b c Feuerer, Markus; Beckhove, Philipp; Garbi, Natalio (10 August 2003). "Bone marrow as a priming site for T-cell responses to blood-borne antigen". Nature Medicine (9): 1151–1157.
- ^ Mazo, I.B.; von Adrian, U.H. (1999). "Adhesion and homing of blood-borne cells in bone marrow microvessels". Journal of Leukocyte Biology. 66 (1): 25–32.
- ^ Milo, Idan; Sapoznikov, Anita; Kalchenko, Vyacheslav (2013). "Dynamic imaging reveals promiscuous crosspresentation of blood-borne antigens to naïve CD8+ T cells in the bone marrow". Blood. 122 (2): 193–208.
- ^ Di Rosa, Francesca; Pabst, Reinhard (2005). "The bone marrow: A nest for migratory memory T cells". Trends in Immunology. 26 (7): 360–366.
- ^ a b Salaming, Stephan A.; Nolte, Martijn A. "The bone marrow as sanctuary for plasma cells and memory T-cells: Implications for adaptive immunity and vaccinology". Cells. 10 (6): 1508.
- ^ a b Chang, Hyun-Dong; Radbruch, Andreas (19 May 2021). "Maintenance of quiescent immune memory in the bone marrow". European Journal of Immunology. 51: 1592–1601.
- ^ Mahnke, Yolanda; Schwendemann, Jochen; Beckhove, Philipp; Schirrmacher, Volker (9 June 2005). "Maintenance of long-term tumour-specific T-cell memory by residual dormant tumour cells". Immunology. 115 (3): 325–336.
- ^ Collins, Nicolas; Han, Seong-Ji; Enamorado, Michel (22 August 2019). "The bone marrow protects and optimizes immunological memory during dietary restriction". Cell. 178 (5): 1088–1101.
- ^ Schirrmacher, Volker (12 October 2015). "Cancer-reactive memory T cells from bone marrow: Spontaneous induction and therapeutic potential (Review)". International Journal of Oncology. 47: 2005–2016.
- ^ Schirrmacher, Volker (2023). "Bone marrow: The central immune system". Immuno. 3 (3): 289–329.
- ^ "The Red Cell Membrane: structure and pathologies" (PDF). Australian Centre for Blood Diseases/Monash University. Retrieved 24 January 2015.
- ^ The Lymphatic System. Allonhealth.com. Retrieved 5 December 2011.
- ^ Fabricant, Florence. "Begging for Bones: A New Craving for Marrow". The New York Times. 16 September 1998.
- S2CID 205381050.
- ^ a b "Bone Marrow Aspiration and Biopsy". Lab Tests Online UK. Retrieved 16 February 2013.
- ^
Mahla RS (2016). "Stem cells application in regenerative medicine and disease threpeutics". International Journal of Cell Biology. 2016 (7): 1–24. PMID 27516776.
- PMID 30035055.
- PMID 26230953.
- PMID 8249729.
- S2CID 20115888.
- PMID 15893536.
- ^ "Definition: 'M:E Ratio'". Stedman's Medical Dictionary via MediLexicon.com. 2006. Archived from the original on 10 May 2013. Retrieved 20 December 2012.
- ^ "Bone marrow transplantation". UpToDate.com. Retrieved 12 April 2014.
- ^ "Antibody Transforms Stem Cells Directly into Brain Cells". Science Daily. 22 April 2013. Retrieved 24 April 2013.
- Wake Forest Baptist Medical Center. 28 February 2013. Archived from the originalon 8 August 2017. Retrieved 5 March 2013.
- ^ "Bone marrow 'frees men of HIV drugs'". BBC. 3 July 2013. Retrieved 3 July 2013.
- ^ "Stem-Cell Transplants Erase HIV in Two Men". PopSci. 3 July 2013. Retrieved 3 July 2013.
- ^ "HIV Returns in Two Men Thought 'Cured' by Bone Marrow Transplants". RH Reality Check. 10 December 2013. Retrieved 10 December 2013.
- ^ National Marrow Donor Program Donor Guide Archived 8 September 2008 at the Wayback Machine. Marrow.org. Retrieved 5 November 2012.
- ^ Bone marrow donation: What to expect when you donate. Mayo Clinic. Retrieved 16 February 2013.
- PMID 16098183.
- PMID 33968803.
- ^ PMID 24648231.
Further reading
- Nature Bone Marrow Transplantation (Nature Publishing Group) – specialist scientific journal with articles on bone marrow biology and clinical uses.
- Cooper, B (2011). "The origins of bone marrow as the seedbed of our blood: from antiquity to the time of Osler". Baylor University Medical Center Proceedings. 24 (2): 115–8. PMID 21566758.
- Wang J, Liu X, Lu H, Jiang C, Cui X, Yu L, Fu X, Li Q, Wang J (2015). "CXCR4(+)CD45(-) BMMNC subpopulation is superior to unfractionated BMMNCs for protection after ischemic stroke in mice". Brain Behav. Immun. 45: 98–108. PMID 25526817.
