Bone metastasis
| Bone metastasis | |
|---|---|
parotid gland cancer. Large lesions are seen on the ilium on the more distant side. Involvement of the vertebral column has caused a compression fracture. | |
| Specialty | Oncology |
Bone metastasis, or osseous metastatic disease, is a category of
Types of lesions
Under normal conditions, bone undergoes continuous remodeling through
Primary tumors
- Osteoblastic lesions[5]
- Prostate cancer
- Carcinoid
- Small cell lung cancer
- Hodgkin lymphoma
- Medulloblastoma
- Osteolytic lesions[5]
- Non-small cell lung cancer
- Thyroid cancer
- Kidney cancer
- Multiple myeloma
- Melanoma
- Non-Hodgkin lymphoma
- Langerhans cell histiocytosis
- Mixed lesions[5]
- Breast cancer
- Testicular cancer
- Ovarian cancer
- Gastrointestinal cancers
- Squamous cell skin cancers
- Hepatocellular carcinomas
Signs and symptoms
Bone metastases can cause severe pain, bone fractures, spinal cord compression, hypercalcemia, anemia, spinal instability, decreased mobility, and rapid degradation in the quality of life for patients.[6][7] Patients have described the pain as a dull ache that grows worse over time, with intermittent periods of sharp, jagged pain.[2] Even under controlled pain management, periods of breakthrough pain can occur rapidly, without warning, several times a day.[8] Pain may be worse at night and partially relieved by activity.[9] Metastases to weightbearing bones may become symptomatic early in the course of disease, as compared to metastases to the flat bones of the rib or sternum.[9]
- Effects of bone metastasis
Major complications secondary to bone metastases are termed Skeletal-Related Events (SREs).[10]
- Occurrence of pathological long bone and vertebral fractures
- Development of spinal cord compression
- Need for radiation for pain relief or to treat or prevent pathological fractures or spinal cord compression
- Requirement for surgery to bone
- Episodes of hypercalcemia of malignancy
Other symptoms include:
- Spinal Instability
- Compression of the Cauda Equina
- Cranial Nerve Palsies
- Suppression of bone marrow function (i.e. anemia)
- Decreased mobility

Sources of bone metastases
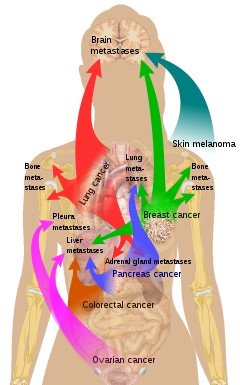
Bone is the third most common location for
Common primary tumors
Mechanism
Initial Seeding
Many cancers spread to bone, but not all bones are implicated in cancerous metastasis. The microenvironment of differing bone types is thought to play a role in its predisposition to tumor seeding.[13] For example, trabecular bone rich in red marrow and bone subject to frequent turnover are more likely to be seeded[13] at premetastatic niches formed by interactions between cancerous cells and bone substrate[14] Tumor cells are then attracted to the metastatic niche in the bone. However, the characteristics of these niches have yet to be fully elucidated.[14] Initial seeding can occur prior to the discovery of the primary tumor.[14]
Vascular Seeding
The pathogenesis of bone metastasis via the vasculature is hypothesized to be related to the Batson vertebral vein plexus, a longitudinal valveless system connected to the breast, lung, kidney, thyroid, and prostate gland that extends from the sacrum to the skull.[15] The most common locations of metastases are the pelvis, vertebral bodies, ribs and ends of long bones.[15]
Dormancy
Once established, the tumor cells can remain dormant on the bone microenvironment, radiologically undetectable, for many years.[14] The triggers which eventually awaken metastatic tumor cells are an active field of study as they could elucidate mechanisms of controlling dormancy.[14]
Tumor Cell-Bone Interactions
Tumor cells may have paraneoplastic effects, such as via the secretion of Prostaglandin E, TGF-alpha, TGF-beta, TNF, and interleukins to increase bone resorption. The destruction of bone affected by bone metastases are caused by osteoclast-mediated osteolysis.[15] The uncoupled regulation of osteoclasts and osteoblasts leads to malformation of the bone.[2] Lytic bone lesions may also lead to the pathogenesis of hypercalcemia in cancers that have spread to bone[16]
Diagnosis
Skeletal Radiography
A plain film x-ray of the entire body can identify bone metastasis. However, the sclerotic or osteolytic lesions must be at least 1 cm in diameter.[14] A combination of X-ray, CT and MRI scans may be most sensitive in the diagnosis of cancerous bone metastasis.[14]
Radionuclide Bone Scan
A
CT Scan
A
MRI
MRIs can be used to detect bone metastasis with a sensitivity of 82-100% and a specificity of 73-100%.
PET Scan
Positron emission tomography (PET) with fluorine 18–labeled fluorodeoxyglucose ( 18 F-FDG) is a powerful diagnostic tool to visualize the activity of bone metastasis.[14] Bone metastases on PET scan are usually multiple, irregularly distributed foci of increased tracer uptake without relationship to a single anatomic structure.[14] A PET scan can directly identify tumor cells with significant metabolic rate. However, it is a costly procedure and device availability may be limited.[14]
Bone Markers
Due to the high rate of bone turnover, metabolites are theorized to be capable of detecting bone metastasis.[14] Use of bone markers for detection and screening is an active field of research, though radiographic evidence remains the gold standard.[14] However, once the presence of a bone metastasis has been established, tumor metabolic markers can provide useful diagnostic and prognostic information.[14]
Treatment
The goals of treatment for bone metastases include pain control, prevention and treatment of fractures, maintenance of patient quality of life, and local tumor control.[9] Optimal treatment requires a multidisciplinary team of physicians, including medical and radiation oncologists, orthopedic surgeons, radiologists, nuclear medicine physician, palliative medicine specialists, and more.[14] Assessment of treatment is determined by multiple factors, including performance status, pain score, impact on quality of life, and overall status of clinical disease.[14] Important therapies include external-beam radiotherapy, targeted radioisotope therapy, image guided tumor ablation chemotherapy, and bone-targeting chemotherapeutic agents such as Bisphosphonates and Denosumab. Orthopedic interventions such as internal fixation or spinal decompression may be necessary in the case of loss of structural stability due to bone destruction.[14]
Pain management
The World Health Organization's pain ladder was designed for the management of cancer-associated pain. The original ladder details the management of pain using a sequence of analgesic medications, starting with non-steroidal anti-inflammatory medications and progressing to weak and strong opioids[17]
Other treatments include
Thermal ablation techniques are increasingly being used in the palliative treatment of painful metastatic bone disease. Although the majority of patients experience complete or partial relief of pain following external radiation therapy, the effect is not immediate and has been shown in some studies to be transient in more than half of patients.[20] For patients who are not eligible or do not respond to traditional therapies ( i.e. radiation therapy, chemotherapy, palliative surgery, bisphosphonates or analgesic medications), thermal ablation techniques have been explored as alternatives for pain reduction. Several multi-center clinical trials studying the efficacy of radiofrequency ablation in the treatment of moderate to severe pain in patients with metastatic bone disease have shown significant decreases in patient reported pain after treatment.[21][22] These studies are limited, however, to patients with one or two metastatic sites; pain from multiple tumors can be difficult to localize for directed therapy. More recently, cryoablation has also been explored as a potentially effective alternative as the area of destruction created by this technique can be monitored more effectively by CT than radiofrequency ablation, a potential advantage when treating tumors adjacent to critical structures.[23]
A Cochrane review of calcitonin for the treatment of metastatic bone pain suggests calcitonin yields no significant benefit in the reduction of bone pain or improvements in quality of life.[24]
Bone-Targeted Agents
Bone-Targeted Agents (BTAs) including Bisphosphonates and Denosumab, can interrupt osteoclast-mediated osteolysis.[14] Osteoclast inhibitors, most frequently used in the treatment of osteoporosis, can allow for bone healing and delay complications.[14] BTAs have been shown to decrease the incidence of Skeletal Related Events (SREs) like pathological fractures, thereby decreasing the need for surgical intervention or pain medication.[14]
Prognosis
Many cancers are predisposed to metastasize to bone.[12] The list below details the likelihood of a cancer, if in a stage of advanced metastasis, to have spread to bone at time of death:[25]
- Breast: 65-75%
- Prostate: 65-75%
- Thyroid: 60%
- Kidney: 20-25%
- Lung: 30-40%
Given the high incidence of breast, lung and prostate cancer, these patients account for > 80% of patients with bone metastases.[14]
For patients with advanced metastatic disease involving the bone, median survival from the time of diagnosis of a bone metastasis varies by primary tumor type. A list is included below:[25]
- Breast: 19–25 months
- Prostate: 12–53 months
- Thyroid: 48 months
- Kidney: 12 months
- Lung: 6–7 months
See also
References
- ^ MedlinePlus Overview bonecancer
- ^ PMID 20536932.
- ^ PMID 21111245.
- S2CID 40275522.
- ^ PMID 28584570.
- PMID 17062708.
- S2CID 44576422.
- S2CID 20516011.
- ^ a b c d Jacofsky, David (2004). "Metastatic Disease to Bone". Hospital Physician.
- ^ PMID 27101076.
- ^ List of included entries and references is found on main image page in Commons: Commons:File:Metastasis sites for common cancers.svg#Summary
- ^ ISBN 978-0781796705.
- ^
- ^ S2CID 70437912.
- ^ ISBN 978-0-323-60980-7.
- S2CID 92580605.
- PMID 32119322, retrieved 2022-09-14
- PMID 21629409.
- ^ Criteria for Palliation of Bone Metastases – Clinical Applications from International Atomic Energy Agency. Retrieved November 2011
- PMID 6178497.
- PMID 20041484.
- PMID 14722039.
- PMID 23065947.
- PMID 16856000.
- ^ PMID 15978828.
Further reading
- Bellahcène A, Castronovo V (January 1997). "Expression of bone matrix proteins in human breast cancer: potential roles in microcalcification formation and in the genesis of bone metastases". Bull Cancer. 84 (1): 17–24. PMID 9180854.
- Furger KA, Menon RK, Tuck AB, Bramwell VH, Chambers AF (November 2001). "The functional and clinical roles of osteopontin in cancer and metastasis". Curr. Mol. Med. 1 (5): 621–32. PMID 11899236.
- Ibrahim T, Leong I, Sanchez-Sweatman O, et al. (2000). "Expression of bone sialoprotein and osteopontin in breast cancer bone metastases". Clin. Exp. Metastasis. 18 (3): 253–60. S2CID 27673069.
- Chung, Leland W.K.; Isaacs, William B.; Simons, Jonathan W. (2007). Prostate Cancer: Biology, Genetics, and the New Therapeutics. Humana Press. ISBN 978-1-59745-224-3.
