Borrelia burgdorferi
| Borrelia burgdorferi | |
|---|---|
| |
| Borrelia burgdorferi | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Spirochaetota |
| Class: | Spirochaetia |
| Order: | Spirochaetales |
| Family: | Borreliaceae |
| Genus: | Borrelia |
| Species: | B. burgdorferi
|
| Binomial name | |
| Borrelia burgdorferi Johnson et al. 1984 emend. Baranton et al. 1992
| |
Borrelia burgdorferi is a
Microbiology
Borrelia burgdorferi is named after the researcher Willy Burgdorfer, who first isolated the bacterium in 1982.[6]
| Test type | Test | Characteristics |
| Colony characters | Size | Small[7] |
| Type | Round[7] | |
| Color | White[7] | |
| Shape | Raised[7] | |
| Morphological characters | Shape | Spirochete[8] |
| Physiological characters | Motility | +[8] |
| Growth at 6.5% NaCl | +[8] | |
| Biochemical characters | Gram staining | - |
| Oxidase | -[9] | |
| Catalase | -[9] | |
| Oxidative-Fermentative | Fermentative[10] | |
| β-Galactosidase | +[11] | |
| Utilization of | Glycerol | +[12] |
| Galactose | +[13] | |
| D-Glucose | +[13] | |
| D-Mannose | +[13] |
Borrelia burgdorferi is a microaerophile, requiring small amounts of oxygen in order to undergo glycolysis and survive. Like all other Borrelia sps., this bacterium is also gram-negative and a spirochete. Borrelia colonies are often smaller, rounded, and white with an elevated center.
Morphology
B. burgdorferi resembles other
B. burgdorferi is a microaerobic, motile spirochete with seven to 11 bundled perisplasmic flagella set at each end that allow the bacterium to move in low- and high-viscosity media alike, which is related to its high virulence factor.[15]
Metabolism
B. burgdorferi is a slow-growing microaerophilic spirochete with a doubling time of 24 to 48 hours.[16]
Transformation
Bacterial
Life cycle
B. burgdorferi circulates between Ixodes ticks and a vertebrate host in an enzootic cycle.[2] B. burgdorferi living in a tick is mainly acquired through blood meals from an infected, competent vertebrate host,[21] but rare cases of transovarial transmission exist.[22] Once a tick is infected, it will then transmit B. burgdorferi by feeding on another vertebrate to complete the cycle.[23] Ticks can transmit B. burgdorferi to humans, but humans are dead-end hosts, unlikely to continue the life cycle of the spirochete.[24] Nymphs molt into adult ticks, which usually feed on larger mammals that are not able to support the survival of B. burgdorferi.[25]
Disease
B. burgdorferi is the
Clinical presentation of Lyme disease is best known for the characteristic bull's-eye rash (also known as 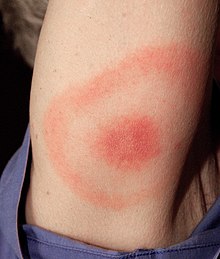
B. burgdorferi infections have been found in possible association with
Progression of the disease follows three stages.
Stage 1
Stage 1 is known as the Early Localized stage and occurs approximately 3 days - 1 month after inoculation. It affects the local area around the bite and is characterized by local swelling and / or a red "bull's-eye" rash (also known as
Stage 2
Stage 2 is known as the Early Disseminated stage and occurs weeks - months after infection if left untreated. The bacteria spreads via the blood through the body to affect the organs. It often presents with general symptoms such as fever, chills, fatigue, and lymphadenopathy as well as the organ-specific symptoms. It can affect the
Stage 3
Stage 3 is known as the Late Disseminated stage and occurs months - years after the initial infection. Effects of the 3rd stage include encephalitis or meningitis,[29] as well as migratory arthropathies (most commonly of the knee).[29]
Anaplasmosis and babesiosis are also common tick-borne pathogens carried by the Ixodes tick that infect humans similarly to Borrelia burgdorferi.[30] Consequently, it is possible for an Ixodes tick to coinfect a host with either two or all other diseases. When a host is coinfected, the combined effects of the diseases act synergistically, often proving to cause worse symptoms than a single infection alone[30] Coinfected humans tend to display a more severe manifestation of Lyme disease. In addition, they tend to acquire a wider range of secondary symptoms, such as influenza-like symptoms.[30] More studies and research must be done to determine the synergistic effect of co-infection and its effect on the human body.
Variation of severity
So far, there are three factors that may contribute to the severity of the clinical manifestation of Lyme Disease. The presence of ribosomal spacers, plasmids, and the outer surface protein C (OspC) are indicators of the severity of the infection.[31] Additionally, humans, themselves, vary in their response to the infection.[31] The variation in response leads to different clinical manifestations and different infections to different organs.[citation needed]
Molecular pathogenesis
After the pathogen is transmitted, it will acclimate to the mammalian conditions. Borrelia burgdorferi will change its glycoproteins and proteases on its plasma membrane to facilitate its dissemination throughout the blood.[31] While infecting, B. burgdorferi will express proteins that will interact with endothelial cells, platelets, chondrocytes, and the extracellular matrix.[31] This interaction inhibits proper function of the infected areas, leading to the pathological manifestations of Lyme disease. In response, the host will initiate an inflammatory response to attempt to remove the infection.[31]
Borrelia burgdorferi, also, expresses at least seven plasminogen binding proteins for interference of factor H at the activation level. This is part of a complement system evasion strategy that leads to downstream blocking of immune response.[32]
In addition, Borrelia burgdorferi has a strategy to directly inhibit the classical pathway of complement system. A borrelial lipoprotein BBK32, expressed on the surface of Borrelia burgdorferi, binds the initiating protease complex C1 of the classical pathway. More specifically, BBK32 interacts with C1r subunit of C1. C-terminal domain of the BBK32 protein mediates the binding. As a result, C1 is trapped in an inactive form.[33]
Genome
B. burgdorferi (B31 strain) was the third microbial
The genomic variations of B. burgdorferi contribute to varying degrees of infection and dissemination.[36] Each genomic group has varying antigens on its membrane receptor, which are specific to the infection of the host. One such membrane receptor is the surface protein OspC.[36] The OspC surface protein is shown to be a strong indicator of the identification of genomic classification and the degree of dissemination.[36] Varying number of OspC loci are indications and determinants for the variations of B. burgdorferi.[36] The surface protein is also on the forefront of current vaccine research for Lyme disease via Borrelia.[37]
Bacteriophage
Relatively few bacteriophages are known to infect B. burgdorferi. Several phage particles were isolated and some evidence suggested that they had an 8-kb dsDNA genome. Among the best-studied Borrelia phages is φBB-1, a phage with a polyhedral head and a contractile tail of 90 nm in length.[38][39] φBB-1 was the first bacteriophage that provided evidence of transduction for lateral gene transfer in Borrelia species that cause Lyme Disease.[40] Current research aims to use bacteriophages as way of identifying virulence factors in spirochaetes that lead to Lyme Disease.[citation needed]
Evolution
Genetically diverse B. burgdorferi strains, as defined by the sequence of ospC, are maintained within the Northeastern United States. Balancing selection may act upon ospC or a nearby sequence to maintain the genetic variety of B. burgdorferi.[41] Balancing selection is the process by which multiple versions of a gene are kept within the gene pool at unexpectedly high frequencies. Two major models that control the selection balance of B.burgdorferi is negative frequency-dependent selection and multiple-niche polymorphism.[42] These models may explain how B. burgdorferi have diversified, and how selection may have affected the distribution of the B. burgdorferi variants, or the variation of specific traits of the species, in certain environments.[citation needed]
Negative-frequency dependent selection
In negative frequency-dependent selection, rare and uncommon variants will have a selective advantage over variants that are very common in an environment.[42] For B. burgdorferi, low-frequency variants will be advantageous because potential hosts will be less likely to mount an immunological response to the variant-specific OspC outer protein.[42]
Multiple-niche polymorphism
Ecological niches are all of the variables in an environment, such as the resources, competitors, and responses, that contribute to the organism's fitness. Multiple-niche polymorphism states that diversity is maintained within a population due to the varying amount of possible niches and environments.[42] Therefore, the more various niches the more likelihood of polymophrism and diversity. For B. burgdorferi, varying vertebrae niches, such as deer and mice, can affect the overall balancing selection for variants.[42]
See also
References
- ISBN 978-1-913652-61-6.
- ^ PMID 34161868.
- ^ CDC (2016-02-08). "New Lyme-disease-causing bacteria species discovered". Centers for Disease Control and Prevention. Retrieved 2019-01-18.
- PMID 18452798.
- ISBN 978-1-904455-58-5.
- PMID 7043737.
- ^ PMID 3693538.
- ^ PMID 9440510.
- ^ PMID 22500025.
- PMID 26185064, retrieved 2022-04-21
- PMID 22763966.
- PMID 21750672.
- ^ PMID 1611204.
- PMID 26519910.
- PMID 10995478.
- PMID 18770608.
- PMID 15514047.
- PMID 21400675.
- PMID 22974303.
- PMID 34113333.
- PMID 29336985.
- PMID 23238242.
- ^ PMID 22974303.
- PMID 22230951.
- ^ PMID 18452798.
- ^ "Signs and Symptoms, Lyme Disease". Centers For Disease Control. March 4, 2015. Retrieved 2015-07-16.
- PMID 16507206.
- ^ PMID 20930075.
- ^ ISBN 978-0-321-73360-3.
- ^ PMID 17041141.
- ^ a b c d e Weis, Janet (2011). "Critical Needs and Gaps in Understanding Prevention, Amelioration, and Resolution of Lyme and Other Tick-Borne Diseases: The Short-Term and Long-Term Outcomes: Workshop Report". The National Academies: 97–101.
- PMID 23810413.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ Garcia, B.L., Zhi, H., Wager, B., Höök, M. & Skare, J.T. 2016, "Borrelia burgdorferi BBK32 Inhibits the Classical Pathway by Blocking Activation of the C1 Complement Complex", PLoS Pathogens, vol. 12, no. 1
- ^ S2CID 4388492.
- PMID 10672174.
- ^ PMID 7768799.
- PMID 23407755.
- PMID 11075907.
- )
- PMID 11466280.
- S2CID 10877654.
- ^ ISBN 9781904455585.
Further reading
- Velázquez, Encarna; Peix, Álvaro; Gómez-Alonso, Alberto (March 2011). "Microorganismos y cáncer: evidencias científicas y nuevas hipótesis" [Microorganisms and cancer: scientific evidence and new hypotheses]. Cirugía Española (in Spanish). 89 (3): 136–144. PMID 21292247.
- Koči, Juraj; Bista, Sandhya; Chirania, Payal; Yang, Xiuli; Kitsou, Chrysoula; Rana, Vipin Singh; Yas, Ozlem Buyuktanir; Sonenshine, Daniel E.; Pal, Utpal (17 March 2021). "Antibodies against EGF-like domains in Ixodes scapularis BM86 orthologs impact tick feeding and survival of Borrelia burgdorferi". Scientific Reports. 11 (1): 6095. PMID 33731754.
