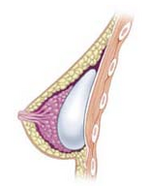
Breast implant
| Breast implant | |
|---|---|
A video of a doctor marking the chest for implants | |
| Specialty | Plastic surgery |
A breast implant is a
Complications of implants may include breast pain, rashes, skin changes, infection, rupture, cosmetic changes to the breasts such as asymmetry and hardness, and a fluid collection around the breast.[1]
A rare complication associated with textured surfaced implants is a type of lymphoma (cancer of the immune system) known as breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL).
There are four general types of breast implants, defined by their filler material: saline solution, silicone gel, structured and composite filler. The saline implant has an
In surgical practice, for the reconstruction of a breast, the tissue expander device is a temporary breast prosthesis used to form and establish an implant pocket for the future permanent breast implant. For the correction of male breast defects and deformities, the pectoral implant is the breast prosthesis used for the reconstruction and the aesthetic repair of a man's chest wall (see: gynecomastia and mastopexy).
Uses
A
- primary reconstruction: the replacement of breast tissues damaged by trauma (tuberous breast deformity).
- revision and reconstruction: to revise (correct) the outcome of a previous breast reconstruction surgery.
- primary augmentation: to aesthetically augment the size, form, and feel of the breasts.
The
surgery is determined by the procedure employed, the type of incisions, the breast implant (type and materials), and the pectoral locale of the implant pocket.Recent research has indicated that mammograms should not be done with any greater frequency than that used in normal procedure in patients undergoing breast surgery, including breast implant, augmentation, mastopexy, and breast reduction.[2]
Psychology
The
Post-operative patient surveys about mental health and quality-of-life, reported improved physical health, physical appearance, social life, self-confidence, self-esteem, and satisfactory sexual functioning. Furthermore, the women reported long-term satisfaction with their breast implant outcomes; some despite having medical complications that required surgical revision, either corrective or aesthetic. Likewise, in Denmark, 8% of breast augmentation patients had a pre-operative history of psychiatric hospitalization.[4][5][6][7][8][9][10][11][12][13]
In 2008, the longitudinal study Excess Mortality from Suicide and other External Causes of Death Among Women with Cosmetic Breast Implants (2007), reported that women who sought breast implants are almost 3 times as likely to commit suicide as are women who have not sought breast implants. Compared to the standard suicide-rate for women of the general populace, the suicide-rate for women with augmented breasts remained constant until 10-years post-implantation, yet, it increased to 4.5 times greater at the 11-year mark, and so remained until the 19-year mark, when it increased to 6 times greater at 20-years post-implantation. Moreover, additional to the suicide-risk, women with breast implants also faced a trebled death-risk from alcoholism and the abuse of prescription and recreational drugs.[14][15] Although seven studies have statistically connected a woman's breast augmentation to a greater suicide-rate, the research indicates that breast augmentation surgery does not increase the death rate; and that, in the first instance, it is the psychopathologically-inclined woman who is more likely to undergo a breast augmentation procedure.[16][17][18][19][20][21]
The study Effect of Breast Augmentation Mammoplasty on Self-Esteem and Sexuality: A Quantitative Analysis (2007), reported that the women attributed their improved
Complications
The
Safety
In the 1990s, several reports reviewed the few studies evaluating any increased risk of systemic and auto-immune diseases among women with breast implants. The conclusion at that time was that there was no evidence establishing a causal connection between the implantation of silicone breast implants and either type of disease.
A published study of U.S. women with similar results was published in 2019 by Coroneos and his colleagues at MD Anderson Medical Center.[31] The data were based on two studies with a combined total of almost 100,000 women with breast implants, but many dropped out of the study within a few years of their breast implant surgery. However, of the women in the study for at least two years, the researchers reported an 800% increase in Sjögren syndrome, 700% increase in scleroderma, and almost 600% increase in rheumatoid arthritis among women with breast implants compared to the general population of women of the same age and demographics.
Recent research on women who reported autoimmune and other system symptoms but were not diagnosed with an autoimmune disease evaluated whether the women's symptoms changed after their implants were removed. A 2020 study on the effectiveness of explant surgery on women with breast implant illness found that nearly all of 750 women who underwent explant surgery reported a significant improvement in their health within a month after their surgery. Researchers focused on the following symptoms: hair loss, memory loss, dry eyes and/or blurred vision, numbness or tingling in the extremities, chronic fatigue, joint pain, rashes, breast pain, food intolerance, flu-like symptoms, and difficulty breathing. The same authors also published a study on the impact of breast implant removal on breathing difficulties and found a statistically significant improvement in well-established objective measures of pulmonary function following explant surgery.[32]
| Year | Country | Systemic Review Group | Conclusions |
|---|---|---|---|
| 1991–93 | United Kingdom | Independent Expert Advisory Group (IEAG) | There was no evidence of an increased risk of connective-tissue disease in patients who had undergone silicone-gel breast implant emplacement, and no cause for changing either breast implant practice or policy in the U.K. |
| 1996 | United States | U.S. Institute of Medicine (IOM)[33] | There was "insufficient evidence for an association of silicone gel- or saline-filled breast implants with defined connective tissue disease." |
| 1996 | France | Agence Nationale pour le Developpement de l'Evaluation Medicale (ANDEM) [National Agency for Medical Development and Evaluation][34] | French original: "Nous n'avons pas observé de connectivité ni d'autre pathologie auto-immune susceptible d'être directement ou indirectement induite par la présence d'un implant mammaire en particulier en gel de silicone...."
English translation: "We did not observe connective tissue diseases to be directly or indirectly associated by the presence of a breast implant, in particular one of silicone gel...." |
| 1997 | Australia | Therapeutic Devices Evaluation Committee (TDEC) | The "current, high-quality literature suggest that there is no association between breast implants and connective tissue disease-like syndromes (atypical connective tissue diseases)."[35] |
| 1998 | Germany | Federal Institute for Medicine and Medical Products | Reported that "silicone breast implants neither cause auto-immune diseases nor rheumatic diseases and have no disadvantageous effects on pregnancy, breast-feeding capability, or the health of children who are breast-fed. There is no scientific evidence for the existence of silicone allergy, silicone poisoning, atypical silicone diseases or a new silicone disease."[36] |
| 2000 | United States | Federal court-ordered review[37] | "No evidence of an association between... silicone-gel-filled breast implants specifically, and any of the individual CTDs, all definite CTDs combined, or other auto-immune or rheumatic conditions." |
| 2000 | European Union | European Committee on Quality Assurance & Medical Devices in Plastic Surgery (EQUAM) | "Additional medical studies have not demonstrated any association between silicone-gel filled breast implants and traditional auto-immune or connective tissue diseases, cancer, nor any other malignant disease. . . . EQUAM continues to believe that there is no scientific evidence that silicone allergy, silicone intoxication, atypical disease or a 'new silicone disease' exists."[38] |
| 2001 | United Kingdom | UK Independent Review Group (UK-IRG) | "There is no evidence of an association with an abnormal immune response or typical or atypical connective tissue diseases or syndromes."[39] |
| 2001 | United States | Court-appointed National Science Panel review[40] | The panel evaluated established and undifferentiated connective tissue diseases (CTD), and concluded there was no causal evidence between breast implants and these CTDs. |
| 2003 | Spain | Science and Technology Options Assessment (STOA) | The STOA report to the European Parliament Petitions Committee reported that the current scientific evidence demonstrates no solid, causal evidence linking SBI [silicone breast implants] to severe diseases, e.g. breast cancer, connective tissue diseases.[41] |
| 2009 | European Union | International Committee for Quality Assurance, Medical Technologies & Devices in Plastic Surgery panel (IQUAM) | The consensus statement of the Transatlantic Innovations conference (April 2009) indicated that additional medical studies demonstrated no association between silicone gel-filled breast implants and carcinoma, or any metabolic, immune, or allergic disorder.[42] |
Implant rupture

Because a breast implant is a Class III medical device of limited product-life, the principal rupture-rate factors are its age and design; nonetheless, a breast implant device can retain its mechanical integrity for decades in a woman's body.[43] When a saline breast implant ruptures, leaks, and empties, it quickly deflates, and thus can be readily explanted (surgically removed). In some cases, saline implant rupture can result in an infection due to bacteria or mold that had been within the implant, though this is uncommon.[44] The follow-up report, Natrelle Saline-filled Breast Implants: a Prospective 10-year Study (2009) indicated rupture-deflation rates of 3–5 per cent at 3-years post-implantation, and 7–10 per cent rupture-deflation rates at 10-years post-implantation.[45] In a study of his 4761 augmentation mammaplasty patients, Eisenberg reported that overfilling saline breast implants 10-13% significantly reduced the rupture-deflation rate to 1.83% at 8-years post-implantation.[46]

When a silicone breast implant ruptures it usually does not deflate, yet the filler gel does leak from it, which can migrate to the implant pocket; therefore, an intracapsular rupture (in-capsule leak) can become an extracapsular rupture (out-of-capsule leak), and each occurrence is resolved by explantation. Although the leaked silicone filler-gel can migrate from the chest tissues to elsewhere in the woman's body, most clinical complications are limited to the breast and armpit areas, usually manifested as granulomas (inflammatory nodules) and axillary lymphadenopathy (enlarged lymph glands in the armpit area).[47][48][49]
The suspected mechanisms of breast implant rupture are:
- damage during implantation
- damage during (other) surgical procedures
- chemical degradation of the breast implant shell
- trauma (blunt trauma, penetrating trauma, blast trauma)
- mechanical pressure of traditional mammographic breast examination[50]
Silicone implant rupture can be evaluated using magnetic resonance imaging; from the long-term
The study Safety and Effectiveness of Mentor's MemoryGel Implants at 6 Years (2009), which was a branch study of the U.S. FDA's core
Furthermore, The Effect of Study design Biases on the Diagnostic Accuracy of Magnetic Resonance Imaging for Detecting Silicone Breast Implant Ruptures: a Meta-analysis (2011) reported that the breast-screening MRIs of asymptomatic women might overestimate the incidence of breast implant rupture.[59] In the event, the U.S. Food and Drug Administration emphasised that "breast implants are not lifetime devices. The longer a woman has silicone gel-filled breast implants, the more likely she is to experience complications."[60]
Capsular contracture
The human body's

The cause of capsular contracture is unknown, but the common incidence factors include bacterial contamination, device-shell rupture, filler leakage, and hematoma. The surgical implantation procedures that have reduced the incidence of capsular contracture include submuscular emplacement, the use of breast implants with a textured surface (polyurethane-coated);[61][62][63] limited pre-operative handling of the implants, limited contact with the chest skin of the implant pocket before the emplacement of the breast implant, and irrigation of the recipient site with triple-antibiotic solutions.[64][65]
The correction of capsular contracture might require an open capsulotomy (surgical release) of the collagen-fiber capsule, or the removal, and possible replacement, of the breast implant. Furthermore, in treating capsular contracture, the closed capsulotomy (disruption via external manipulation) once was a common maneuver for treating hard capsules, but now is a discouraged technique, because it can rupture the breast implant. Non-surgical treatments for collagen-fiber capsules include massage, external
Repair and revision surgeries
When the patient is unsatisfied with the outcome of the augmentation mammoplasty; or when technical or medical complications occur; or because of the breast implants' limited product life, it is likely she might require replacing the breast implants. Common revision surgery indications include major and minor medical complications,
Systemic disease

In the 1990s, the national health ministries of the listed countries reviewed the pertinent studies for causal links among silicone-gel breast implants and systemic and diagnosed autoimmune diseases and breast cancer.
A 2021 study by Cleveland Clinic physicians investigated the outcome of breast implant removal on women with breast implants who had reported difficulty breathing or tightness in their chest; this is one of the symptoms associated with breast implant illness.
Platinum toxicity
Platinum is a catalyst used in the making of silicone implant polymer shells and other silicone devices used in medicine. The literature indicates that small amounts of platinum leaches (leaks) from these implants and is present in the surrounding tissue. The FDA reviewed the available studies from the medical literature on platinum and breast implants in 2002 and concluded there was little evidence suggesting toxicity from platinum in implant patients.[76] The FDA revisited this study and additional literature several years later, reaffirming prior conclusions that platinum catalysts used in implants is likely not ionized and therefore would not represent a risk to women.[77]
Anaplastic large-cell lymphoma
The FDA has identified that breast implants may be associated with a rare form of cancer called anaplastic large-cell lymphoma (ALCL), which some experts believe is believed to be associated with chronic bacterial inflammation.[78] Similar ALCL phenomena have been seen with other types of medical implants including vascular access ports, orthopedic hip implants, and jaw (TMJ) implants. The causal association between breast implants and ALCL was conclusively established in December 2013, when researchers at MD Anderson Cancer Center published a study of 60 women with breast implants who were diagnosed with ALCL in the breast. In 2015, plastic surgeons published an article reviewing 37 articles in the literature on 79 patients and collected another 94 unreported cases, resulting in 173 women with breast implants who had developed ALCL of the breast. They concluded that "Breast implant-associated ALCL is a novel manifestation of site- and material-specific lymphoma originating in a specific scar location, presenting a wide array of diverse characteristics and suggesting a multifactorial cause." They stated that "There was no preference for saline or silicone fill or for cosmetic or reconstructive indications." Where implant history was known, the patient had received at least one textured-surface device. In 2016, the World Health Organization (WHO) officially recognized BIA-ALCL.[79]
As of April 2022, the FDA has received 1,130 global medical device reports (MDRs) of BIA-ALCL, including 59 deaths.
The current lifetime risk of BIA-ALCL in the U.S. is unknown, but estimates have ranged between one in 70,000 and one in 500,000 women with breast implants, according to the MD Anderson Cancer Center.[84] Countries with breast implant registries have the best data on the risks of BIA-ALCL. For example, as of October 2020, the Therapeutic Goods Administration of Australia and New Zealand reported a "1:3,345 risk with Allergan Biocell and a 1:86,029 risk with Mentor Siltex."[82] AIn the U.S, estimates of the risk of BIA-ALCL in textured implants ranges from 1.79 per 1,000 (1 woman with BIA-ALCL per 559 implants) to 2.82 per 1,000 (1 woman per 355 implants)[2]. As of April 2022, the FDA reported 1,130 medical device reports (MDRs) of BIA-ALCL. Of those MDRs, 59 of the women died . FDA states that 798 of the 1,130 MDRs for BIA-ALCL involved textured breast implants, 37 MDRs involved smooth-surfaced implants, and 295 MDRs did not specify whether the implants were textured or smooth.[80] In some cases, the women who developed BIA-ALCL after smooth breast implants had textured expanders prior to their implants. Most patients had BIA-ALCL affect only one breast with only 8 patients diagnosed with bilateral BIA-ALCL. The most common reported symptom of BIA-ALCL according to the FDA was seroma, followed by breast swelling and pain, capsular contracture, and peri-implant mass or lump. Furthermore, 84 percent of the reported implants were made by Allergan.[80]
A study conducted at Memorial Sloan Kettering cancer center of women undergoing prophylactic mastectomies indicated that 1 in 355 women were subsequently diagnosed with BIA-ALCL. [85] Almost all those women had Allergan biocell implants and expanders. These reports strongly suggest that BIA-ALCL develops primarily in patients with textured implants or expanders. In all cases, however, many researchers suspect that BIA-ALCL is an under-recognized, misdiagnosed, and under-reported complication of breast implants.
The ASPS and the Plastic Surgery Foundation (PSF) have partnered with the FDA to study this condition and in doing so created the Patient Registry and Outcomes for Breast Implants and Anaplastic Large Cell Lymphoma Etiology and Epidemiology (PROFILE). The United States FDA strongly encourages all physicians to report cases to PROFILE in an effort to better understand the role of breast implants in ALCL and the management of this disease.[86]
Other lymphomas and squamous-cell carcinoma
In September, 2022, the FDA announced new information regarding other types of cancers related to breast implants, based on medical device reports (MDRs) of cases of patients with squamous-cell carcinoma or various lymphomas found in the scar tissue around breast implants. The agency reported 10 MDRs about squamous-sell carcinoma found in the capsule scar tissue and 12 MDRs about various lymphomas also in the scar tissue around the breast implants. These new cases do not overlap with cases of BIA-ALCL. SCC and other lymphomas have been found in smooth-surface and textured implants as well as silicone-gel-filled and saline-filled implants. Patients who reported symptoms specified swelling, pain, lumps, and changes to their skin. As of 2022, neither the incidence rate nor prevalence is known.[87]
Surgical procedures
Incision types
Breast implant emplacement is performed with five types of surgical incisions:
- Inframammary: an incision made to the pectoralis muscle interface; yet, IMF implantation can produce thicker, slightly more visible surgical scars.
- Periareolar: a border-line incision along the periphery of the mammoplasty procedure. In periareolar emplacement, the incision is around the medial-half (inferior half) of the areola's circumference. Silicone gel implants can be difficult to emplace via periareolar incision, because of the short, five-centimetre length (~ 5.0 cm) of the required access-incision. Aesthetically, because the scars are at the areola's border (periphery), they usually are less visible than the IMF-incision scars of women with light-pigment areolae; when compared to cutaneous-incision scars, the modified epitheliaof the areolae are less prone to (raised) hypertrophic scars.
- Transaxillary: an incision made to the axilla (armpit), from which the dissection tunnels medially, to emplace the implants, either bluntly or with an endoscope (illuminated video microcamera), without producing visible scars on the breast proper; yet, it is likelier to produce inferior asymmetry of the implant-device position. Therefore, surgical revision of transaxillary emplaced breast implants usually requires either an IMF incision or a periareolar incision.
- Transumbilical: a trans-umbilical breast augmentation (TUBA) is a less common implant-device emplacement technique wherein the incision is at the umbilicus (navel), and the dissection tunnels superiorly, up towards the bust. The TUBA approach allows emplacing the breast implants without producing visible scars upon the breast proper; but makes appropriate dissection and device-emplacement more technically difficult. A TUBA procedure is performed bluntly—without the endoscope's visual assistance—and is not appropriate for emplacing (pre-filled) silicone-gel implants, because of the great potential for damaging the elastomer silicone shell of the breast implant during its manual insertion through the short (~2.0 cm) incision at the navel, and because pre-filled silicone gel implants are incompressible, and cannot be inserted through so small an incision.[88]
- Transabdominal: as in the TUBA procedure, in the transabdominoplasty breast augmentation (TABA), the breast implants are tunneled superiorly from the abdominal incision into bluntly dissected implant pockets, whilst the patient simultaneously undergoes an abdominoplasty.[89]
Implant pocket placement
The five
- Subglandular: the breast implant is emplaced to the pectoralis major muscle (major muscle of the chest), which most approximates the plane of normal breast tissue, and affords the most aesthetic results. Yet, in women with thin pectoral soft-tissue, the subglandular position is likelier to show the ripples and wrinkles of the underlying implant. Moreover, the capsular contractureincidence rate is slightly greater with subglandular implantation.
- Subfascial: the breast implant is emplaced beneath the pectoralis major muscle; the subfascial position is a variant of the subglandular position for the breast implant.[90] The technical advantages of the subfascial implant-pocket technique are debated; proponent surgeons report that the layer of fascial tissue provides greater implant coverage and better sustains its position.[91]
- Subpectoral (dual plane): the breast implant is emplaced beneath the pectoralis major muscle, after the surgeon releases the inferior muscular attachments, with or without partial dissection of the subglandular plane. Resultantly, the upper pole of the implant is partially beneath the pectoralis major muscle, while the lower pole of the implant is in the subglandular plane. This implantation technique achieves maximal coverage of the upper pole of the implant, whilst allowing the expansion of the implant's lower pole; however, "animation deformity", the movement of the implants in the subpectoral plane can be excessive for some patients.[92]
- Submuscular: the breast implant is emplaced beneath the pectoralis minor muscle, or both—and suturing it, or them, to the pectoralis major muscle. In breast reconstructionsurgery, the submuscular implantation approach effects maximal coverage of the breast implants. This technique is rarely used in cosmetic surgery due to high risk of animation deformities.
- Prepectoral or subcutaneous: in a breast reconstruction following a skin-sparing or skin- and nipple-sparing mastectomy, the implant is placed above the pectoralis major muscle without dissecting it so that the implant fills directly the volume of the mammary gland that has been removed. To avoid the issue of capsular contracture, the implant is often covered frontally or completely with a mesh in biomaterial, either biological or synthetic.
Post-surgical recovery
The surgical scars of a breast augmentation mammoplasty develop approximately at 6-weeks post-operative, and fade within months. Depending upon the daily-life physical activities required of the woman, the breast augmentation patient usually resumes her normal life at 1-week post-operative. Moreover, women whose breast implants were emplaced beneath the chest muscles (submuscular placement) usually have a longer, slightly more painful convalescence, because of the healing of the incisions to the chest muscles. Usually, she does not exercise or engage in strenuous physical activities for approximately 6 weeks. During the initial post-operative recovery, the woman is encouraged to regularly exercise (flex and move) her arm to alleviate pain and discomfort; if required, analgesic indwelling medication catheters can alleviate pain[93][94] Moreover, significantly improved patient recovery has resulted from refined breast-device implantation techniques (submuscular, subglandular) that allow 95 per cent of women to resume their normal lives at 24-hours post-procedure, without bandages, fluid drains, pain pumps, catheters, medical support brassières, or narcotic pain medication.[95][96][97][98]
Types


Today, there are three types of breast implants commonly used for mammaplasty, breast reconstruction, and breast augmentation procedures:[99]
- saline implant filled with sterile saline solution.
- silicone implant filled with viscous silicone gel.
- structured implants using nested elastomer silicone shells and two saline filled lumen.
A fourth type of implant, composite (or alternative-composite) implants, have largely been discontinued. These types featured fillers such as soy oil and polypropylene string. Other discontinued materials include ox cartilage, Terylene wool, ground rubber, silastic rubber, and teflon-silicone prostheses.[100]
Saline implants
The saline breast implant—filled with
The technical goal of saline-implant technology was a physically less invasive surgical technique for emplacing an empty breast implant device through a smaller surgical incision.[102] In surgical praxis, after having emplaced the empty breast implants to the implant pockets, the plastic surgeon then filled each device with saline solution, and, because the required insertion-incisions are short and small, the resultant incision-scars will be smaller and shorter than the surgical scars usual to the long incisions required for inserting pre-filled, silicone-gel implants.
When compared to the results achieved with a silicone-gel breast implant, the saline implant can yield acceptable results, of increased breast-size, smoother hemisphere-contour, and realistic texture; yet, it is likelier to cause cosmetic problems, such as the rippling and the wrinkling of the breast-envelope skin, accelerated lower breast pole stretch, and technical problems, such as the presence of the implant being noticeable to the eye and to the touch. The occurrence of such cosmetic problems is likelier in the case of the woman with very little breast tissue, and in the case of the woman who requires post-mastectomy breast reconstruction; thus, the silicone-gel implant is the technically superior prosthetic device for breast augmentation, and for breast reconstruction. In the case of the woman with much breast tissue, for whom sub-muscular emplacement is the recommended surgical approach, saline breast implants can produce an aesthetic result much like that afforded by silicone breast implants.[103][104] Ultrasound examination and outcome studies have revealed that saline and silicone breast implants look and feel similar.[105]
Silicone gel implants
As a medical device technology, there are five generations of silicone breast implant, each defined by common model-manufacturing techniques.[citation needed]
The modern prosthetic breast was invented in 1961 by the American
First generation
The Cronin–Gerow Implant, prosthesis model 1963, was a silicone rubber envelope-sac, shaped like a teardrop, which was filled with viscous silicone-gel. To reduce the rotation of the emplaced breast implant upon the chest wall, the model 1963 prosthesis was affixed to the implant pocket with a fastener-patch, made of Dacron material (Polyethylene terephthalate), which was attached to the rear of the breast implant shell.[106]
Second generation
In the 1970s, manufacturers presented the second generation of breast implant prostheses that featured functional developments and aesthetic improvements to the technology:
- the first technological developments were a thinner-gauge device-shell, and a filler gel of low-cohesion silicone, which improved the functionality and the verisimilitude (size, appearance, and texture) of the silicone-gel breast implant. Yet, in clinical practice, second-generation breast implants proved fragile, and had greater incidences[spelling?] of shell rupture, and of filler leakage ("silicone-gel bleed") through the intact device shell. The consequent, increased incidence-rates of medical complications (e.g. capsular contracture) precipitated faulty-product, class action-lawsuits, by the U.S. government, against the Dow Corning Corporation, and other manufacturers of breast prostheses.
- the second technological development was a polyurethane foam coating for the shell of the breast implant; the coating reduced the incidence of capsular contracture, by causing an inflammatory reaction that impeded the formation of a capsule of fibrous collagen tissue around the breast implant. Nevertheless, despite that prophylactic measure, the medical use of polyurethane-coated breast implants was briefly discontinued, because of the potential health-risk posed by 2,4-toluenediamine (TDA), a carcinogenic by-product of the chemical breakdown of the polyurethane foam coating of the breast implant proven to cause liver and skin cancers in animal-model studies.[107]
- After reviewing the medical data, the U.S. Food and Drug Administration concluded that TDA-induced breast cancer was an infinitesimal health-risk to women with breast implants, and did not justify legally requiring physicians to explain the matter to their patients. In the event, polyurethane-coated breast implants remain in plastic surgery practice in Europe and in South America; and no manufacturer has sought FDA approval for medical sales of such breast implants in the U.S.[108]
- the third technological development was the double lumen breast implant device, a double-cavity prosthesis composed of a silicone breast implant contained within a saline breast implant. The two-fold, technical goal was: (i) the cosmetic benefits of silicone-gel (the inner lumen) enclosed in saline solution (the outer lumen); (ii) a breast implant device the volume of which is post-operatively adjustable. Nevertheless, the more complex design of the double-lumen breast implant suffered a device-failure rate greater than that of single-lumen breast implants. The contemporary versions of second-generation breast implant devices (presented in 1984) are the "Becker Expandable" models of breast implant, which are primarily used for breast reconstruction.
Third and fourth generations
In the 1980s, the models of the third and of the fourth generations of breast implant devices were sequential advances in manufacturing technology, such as elastomer-coated shells that decreased gel-bleed (filler leakage), and a thicker (increased-cohesion) filler gel. Sociologically, the manufacturers of prosthetic breasts then designed and made anatomic models (natural breast) and shaped models (round, tapered) that realistically corresponded with the breast- and body- types of women. The tapered models of breast implant have a uniformly textured surface, which reduces the rotation of the prosthesis within the implant pocket; the round models of breast implant are available in smooth-surface- and textured-surface- types.
Fifth generation
Since the mid-1990s, the fifth generation of silicone-gel breast implant is made of a high-strength, highly cohesive silicone gel that mostly eliminates the occurrences of filler leakage ("silicone gel bleed") and of the migration of the silicone filler from the implant pocket to elsewhere in the woman's body. These implants are commonly referred to as "gummy bear breast implants" for their firm, pliant consistency, which is similar to gummy candies. The studies Experience with Anatomical Soft Cohesive Silicone gel Prosthesis in Cosmetic and Reconstructive Breast Implant Surgery (2004) and Cohesive Silicone gel Breast Implants in Aesthetic and Reconstructive Breast Surgery (2005) reported low incidence-rates of capsular contracture and of device-shell rupture; and greater rates of improved medical-safety and technical-efficacy than that of early generation breast implant devices.[109][110][111]
Breast-feeding
The presence of breast implants currently presents no contraindication to breast feeding, and no evidence to support that the practice may present health issues to a breast feeding infant is recognized by the USFDA.
Women with breast implants may have functional breast-feeding difficulties; mammoplasty procedures that feature periareolar incisions are especially likely to cause breast-feeding difficulties. Surgery may also damage the lactiferous ducts and the nerves in the nipple-areola area.[112][113][114]
Functional breast-feeding difficulties arise if the surgeon cut the milk ducts or the major nerves innervating the breast, or if the milk glands were otherwise damaged. Milk duct and nerve damage are more common if the incisions cut tissue near the nipple. The milk glands are most likely to be affected by subglandular implants (under the gland), and by large-sized breast implants, which pinch the lactiferous ducts and impede milk flow. Small-sized breast implants, and submuscular implantation, cause fewer breast-function problems; however, it is impossible to predict whether a woman who undergoes breast augmentation will be able to successfully breast feed since some women are able to breast-feed after periareolar incisions and subglandular placement and some are not able to after augmentation using submuscular and other types of surgical incisions.[114]
Mammography

The presence of
The breast cancer studies Cancer in the Augmented Breast: Diagnosis and Prognosis (1993) and Breast Cancer after Augmentation Mammoplasty (2001) of women with breast implant prostheses reported no significant differences in disease-stage at the time of the diagnosis of cancer; prognoses are similar in both groups of women, with augmented patients at a lower risk for subsequent cancer recurrence or death.[117][118] Conversely, the use of implants for breast reconstruction after breast cancer mastectomy appears to have no negative effect upon the incidence of cancer-related death.[119] That patients with breast implants are more often diagnosed with palpable—but not larger—tumors indicates that equal-sized tumors might be more readily palpated in augmented patients, which might compensate for the impaired mammogram images.[120] The ready palpability of the breast-cancer tumor(s) is consequent to breast tissue thinning by compression, innately in smaller breasts a priori (because they have lesser tissue volumes), and that the implant serves as a radio-opaque base against which a cancerous tumor can be differentiated.[121]

The breast implant has no clinical bearing upon lumpectomy breast-conservation surgery for women who developed breast cancer after the implantation procedure, nor does the breast implant interfere with external beam radiation treatments (XRT); moreover, the post-treatment incidence of breast-tissue fibrosis is common, and thus a consequent increased rate of capsular contracture.[122] There is tentative evidence that women who have had breast augmentation, have worse breast cancer prognosis.[123] The use of implants for breast reconstruction after breast cancer mastectomy appears to have no negative effect upon cancer-related death.[119][124]
There have been multiple reported cases of other adverse effects of mammography of women with breast implants; ruptures resulting from pressure exerted on the breast implant make up a majority of these cases.[125] Compression may also lead to pain or exacerbate already existing pain in the breasts.[125]
History

19th century
Since the late nineteenth century, breast implants have been used to surgically augment the size (volume), modify the shape (contour), and enhance the feel (tact) of a woman's breasts. In 1895, surgeon Vincenz Czerny effected the earliest breast implant emplacement when he used the patient's autologous adipose tissue, harvested from a benign lumbar lipoma, to repair the asymmetry of the breast from which he had removed a tumor.[126] In 1889, surgeon Robert Gersuny experimented with paraffin injections, with disastrous results arising from the breakup of the paraffin into smaller bodies following the procedure.[127]
20th century
From the first half of the twentieth century, physicians used other substances as breast implant fillers—
In the mid-twentieth century, Morton I. Berson, in 1945, and Jacques Maliniac, in 1950, each performed flap-based breast augmentations by rotating the patient's chest wall tissue into the breast to increase its volume. Furthermore, throughout the 1950s and the 1960s, plastic surgeons used synthetic fillers—including
FDA approval
In 1988, twenty-six years after the 1962 introduction of breast implants filled with silicone gel, the
- In 1997, the complications from the emplacement of silicone breast implants. The IOM's review of the safety and efficacy of silicone gel-filled breast implants, reported that the "evidence suggests diseases or conditions, such as connective tissue diseases, cancer, neurological diseases, or other systemic complaints or conditions are no more common in women with breast implants, than in women without implants" subsequent studies and systemic review found no causal link between silicone breast implants and disease.[130]

- In 1998, the U.S. FDA approved adjunct study protocols for silicone-gel filled implants only for breast reconstruction patients and for revision-surgery patients; and also approved the Dow Corning Corporation's Investigational Device Exemption (IDE) study for silicone-gel breast implants for a limited number of breast augmentation-, reconstruction-, and revision-surgery patients.[130]
- In 1999, the Institute of Medicine published the Safety of Silicone Breast Implants (1999) study that reported no evidence that saline-filled and silicone-gel filled breast implant devices caused systemic health problems; that their use posed no new health or safety risks; and that local complications are "the primary safety issue with silicone breast implants", in distinguishing among routine and local medical complications and systemic health concerns."[130][131][132]
- In 2000, the FDA approved saline breast implant Premarket Approval Applications (PMA) containing the type and rate data of the local medical complications experienced by the breast surgery patients.[133] "Despite complications experienced by some women, the majority of those women still in the Inamed Corporation and Mentor Corporation studies, after three years, reported being satisfied with their implants."[130] The premarket approvals were granted for breast augmentation, for women at least 18 years old, and for women requiring breast reconstruction.[134][135]
- In 2006, for the Inamed Corporation and for the Mentor Corporation, the U.S. Food and Drug Administration lifted its restrictions against using silicone-gel breast implants for breast reconstruction and for augmentation mammoplasty. Yet, the approval was conditional upon accepting FDA monitoring, the completion of 10-year-mark studies of the women who already had the breast implants, and the completion of a second, 10-year-mark study of the safety of the breast implants in 40,000 other women.[136] The FDA warned the public that breast implants do carry medical risks, and recommended that women who undergo breast augmentation should periodically undergo MRI examinations to screen for signs of either shell rupture or of filler leakage, or both conditions; and ordered that breast surgery patients be provided with detailed, informational brochures explaining the medical risks of using silicone-gel breast implants.[130]
- In March 2019, the FDA hosted a public meeting of their General and Plastic Surgery Devices Advisory Panel to discuss safety issues of silicone and saline breast implants, including BIA-ALCL and breast implant illness.[137] One of the major topics of that public meeting was the evidence that Allergan BIOCELL textured breast implants were the type of breast implants most likely to cause BIA-ALCL. Following the committee meeting, the FDA requested that Allergan recall their BIOCELL textured breast implants and tissue expanders, and in July 2019 Allergan took those BIOCELL textured implants and expanders off the market. Allergan subsequently announced they would contact all customers who had purchased their products to ensure they were aware of the recall.[138]
The U.S. Food and Drug Administration established the age ranges for women seeking breast implants; for breast reconstruction, silicone-gel filled implants and saline-filled implants were approved for women of all ages; for breast augmentation, saline implants were approved for women 18 years of age and older; silicone implants were approved for women 22 years of age and older.
See also
- Breast augmentation
- Breast enlargement supplements
- Breast reconstruction
- Breast reduction plasty
- Mastopexy (breast lift)
- Poly Implant Prothèse
- Polypropylene breast implants
- Trans-umbilical breast augmentation (TUBA)
References
- ^ "Risks and Complications of Breast Implants". FDA. 21 October 2019. Retrieved 30 October 2019.
- ABIM Foundation, American Society of Plastic Surgeons, archived from the originalon 19 July 2014, retrieved 25 July 2014
- S2CID 32599107.
- PMID 15596635.
- S2CID 753343.
- S2CID 8925060.
- S2CID 45574374.
- PMID 16181718.
- PMID 11994621.
- S2CID 23169107.
- ^ "Important Information for Women About Breast Augmentation with Inamed Silicone Gel-Filled Implants" (PDF). Food and Drug Administration. 2006. Archived from the original (PDF) on 2007-01-03.
- ^ Dolan, Eric W. (2023-10-06). "Breast implants have a positive impact on female sexuality, according to new research". PsyPost. Retrieved 2023-10-09.
- S2CID 15228702.
- ^ "Breast Implants Linked with Suicide in Study". Reuters. 2007-08-08. Archived from the original on 2008-12-21.
- ^ Manning A (2007-08-06). "Breast Implants Linked to Higher Suicide Rates". USA Today. Archived from the original on 2011-03-18. Retrieved 2010-04-26.
- ^ PMID 11306343.
- PMID 12623911.
- S2CID 34929987.
- PMID 16777929.
- S2CID 22285852.
- ^ National Plastic Surgery Procedural Statistics, 2006. Arlington Heights, Illinois, American Society of Plastic Surgeons, 2007
- ^ "Plastic Surgery Helps Self-Esteem". Psych Central.com. Archived from the original on 2010-06-19.
- PMID 23864111.
- ^ a b "Important Information for Women About Breast Augmentation with INAMED Silicone-Filled Breast Implants" (PDF). Food and Drug Administration. 2006-11-03. Archived from the original (PDF) on 2007-01-03. Retrieved 2007-05-04.
- ^ "Important Information for Augmentation Patients About Mentor MemoryGel Silicone Gel-Filled Breast Implants" (PDF). 2006-11-03. Archived from the original (PDF) on 16 October 2014. Retrieved 11 October 2014.
- ^ "Saline-Filled Breast Implant Surgery: Making An Informed Decision (Mentor Corporation)". FDA Breast Implant Consumer Handbook - 2004. 2004-01-13. Archived from the original on 2006-11-26. Retrieved 2007-05-04.
- ^ "FDA NEWS RELEASE". Food and Drug Administration. Archived from the original on 2011-11-03. Retrieved 2011-11-09.
- ^ a b Diamond BA; Hulka BS; Kerkvliet NI; Tugwell P. "Silicone Breast Implants in Relation to Connective Tissue Diseases and Immunologic Dysfunction: A Report by a National Science Panel to the Honorable Sam C. Pointer Jr. Coordinating Judge for the Federal Breast Implant Multi-District Litigation". 1998.
- PMID 10717013.
- ^ PMID 30329056.
- S2CID 52284936.
- ^ S2CID 235801794.
- S2CID 29456173.
- ^ Benadiba L (2004). "Histoire des protheses mammaires" (in French). Archived from the original on 29 January 2015. Retrieved 12 October 2015.
- ISBN 0642735794. Archived from the original(PDF) on 2007-01-01. Retrieved 2006-12-29.
- ^ "German Society for Senology, Declaration of Consensus for the Security of Silicone Breast Implants". 24 September 1998.
- ^ PMID 10717013.
- ^ [1] Archived December 27, 2005, at the Wayback Machine
- ^ [2] Archived June 23, 2006, at the Wayback Machine
- PMID 11710703.
- ^ Gorgojo L, Gonzalez J, Wisbaum W, Martin-Moreno J (30 May 2003). Health risks posed by silicone implants in general, with a special attention to breast implants (PDF) (Report). Archived from the original (PDF) on 2003-08-29. Retrieved 2019-01-28.
- S2CID 29112694.
- PMID 11000165.
- ^ "Risks and Complications of Breast Implants". U.S. Food and Drug Administration. 28 September 2020. Retrieved 14 October 2021.
- PMID 19233001.
- PMID 33712871.
- S2CID 25947224.
- S2CID 31982669. Archived from the originalon May 24, 2009.
- ^ "Study of Rupture of Silicone Gel-filled Breast Implants (MRI Component)". FDA Breast Implant Consumer Handbook - 2004. 2000-05-22. Archived from the original on 2007-06-09. Retrieved 2007-05-04.
- ^ a b c "Local Complications". FDA Breast Implant Consumer Handbook - 2004. 2004-06-08. Archived from the original on 2007-05-13. Retrieved 2007-05-04.
- ^ MRI of a ruptured silicone breast implant Archived 2013-09-26 at the Wayback Machine 2013-04-05
- PMID 12860765.
- S2CID 30442865.
- ^ "FDA summary of clinical issues (MS Word document)". Food and Drug Administration. Archived from the original on 2008-03-08.
- S2CID 25722841.
- S2CID 34380204.
- S2CID 39525474.
- ^ "Expert Advisory Panel on Breast Implants: Record of Proceedings". HealthCanada. 2005-09-29. Archived from the original on 2007-11-07. Retrieved 2007-05-04.
- PMID 21364405.
- ^ AFP (18 September 2011). "Breast implants safe, but not for life: US experts". The Independent. Archived from the original on 3 August 2016.
- S2CID 35420582.
- S2CID 29643167.
- PMID 19338905.
- S2CID 39767802.
- S2CID 35238465.
- S2CID 2784003.
- PMID 19331987.
- S2CID 251008.
- S2CID 8451166.
- PMID 15457046.
- S2CID 27630646.
- S2CID 20584928.
- ^ Grigg, Martha; Bondurant, Stuart (2000). "A Report of a Study by the Institute of Medicine". Information for Women About the Safety of Silicone Breast Implants – via NCBI.
{{cite book}}:|journal=ignored (help) - S2CID 22285852.
- ^ "Labeling for Approved Breast Implants". U.S. Food and Drug Administration. 3 December 2021. Archived from the original on 2019-09-11. Retrieved 19 January 2022.
- PMID 12627791.
- ^ "FDA Backgrounder on Platinum in Silicone Breast Implants". U.S. Food and Drug Administration. 16 June 2006. Archived from the original on 2006-07-15.
- ^ Office of the Commissioner. "Safety Alerts for Human Medical Products - Breast Implants: Update - Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL)". www.fda.gov. Archived from the original on 28 April 2018. Retrieved 28 April 2018.
- PMID 26980727.
- ^ a b c "Breast Implants - Medical Device Reports of Breast Implant-Associated Anaplastic Large Cell Lymphoma". www.fda.gov. 20 August 2020. Retrieved 18 August 2021.
- PMID 24323027.
- ^ a b Clemens, Mark. "Breast Implant Associated Anaplastic Large Cell Lymphoma (BIA-ALCL) Archived 2017-03-26 at the Wayback Machine" (2017).
- PMID 28184418.
- ^ "Implant-associated ALCL Facts | The MD Anderson Foundation". www.mdanderson.org. Archived from the original on 2017-12-09. Retrieved 2017-12-08.
- PMID 32008941.
- ^ "Breast Implant Associated ALCL: PROFILE Project | The Plastic Surgery Foundation". www.thepsf.org. Archived from the original on 2017-05-07. Retrieved 2017-04-25.
- ^ "Breast Implants: Reports of Squamous Cell Carcinoma and Various Lymphomas in Capsule Around Implants: FDA Safety Communication". U.S. Food and Drug Administration. September 8, 2022.
- PMID 8415961.
- S2CID 44430032.
- PMID 12560720.
- PMID 14758271.
- PMID 11964998.
- PMID 19083538.
- S2CID 13441800.
- PMID 11964998.
- S2CID 11180810.
- S2CID 26419990.
- S2CID 21392313.
- ^ "Choosing Your Breast Implants" (Web). Minneapolis Plastic Surgery, LTD. Archived from the original on 24 November 2016. Retrieved 23 November 2016.
- ISBN 9781524659073. Retrieved 7 June 2019.
- ^ S2CID 41156555.
- ^ Arion HG (1965). "Retromammary Prosthesis". Comptes Rendus de la Société Française de Gynécologie. 5.
- S2CID 136191732.
- S2CID 252437143.
- PMID 32766047.
- ^ Cronin TD, Gerow FJ (1963). "Augmentation Mammaplasty: A New "natural feel" Prosthesis". Excerpta Medica International Congress Series. 66: 41.
- PMID 9637796.
- PMID 11471963.
- S2CID 35392851.
- S2CID 24661896.
- PMID 11471959.
- ^ Breastfeeding after Breast Surgery Archived 2010-12-30 at the Wayback Machine, La Leche League, contains references.
- ^ Breastfeeding and Breast Implants Archived 2010-12-31 at the Wayback Machine, Selected Bibliography April 2003, LLLI Center for Breastfeeding Information
- ^ Slate.com, 11 December 2009
- PMID 1404718.
- PMID 31981297.
- PMID 8374874.
- S2CID 26010159.
- ^ PMID 15743498.
- S2CID 36277679.
- S2CID 220562548.
- PMID 16864033.
- PMID 23637132.
- PMID 23359049.
- ^ a b "Breast Implant Adverse Events During Mammography". Federal Drug Administration. 18 January 2018. Archived from the original on 2020-09-19.
- ^ Czerny V (1895). "Plastischer Ersatz der Brusthus durch ein Lipoma". Zentralblatt für Chirurgie. 27: 72.
- PMID 16860452.
- from the original on 2007-03-13.
- ^ Anderson N (1997). "Lawsuit Science: Lessons from the Silicone Breast Implant Controversy". New York Law School Law Review. 41 (2): 401–07.
- ^ a b c d e f g h "FDA Breast Implant Consumer Handbook - 2004". Food and Drug Administration. Archived from the original on September 17, 2008.
- )
- )
- ^ "FDA study". Food and Drug Administration. Archived from the original on January 13, 2008.
- ^ "FDA approval". fda.gov. Archived from the original on 30 March 2009. Retrieved 28 April 2018.
- ^ "FDA approval". fda.gov. Archived from the original on 26 May 2009. Retrieved 28 April 2018.
- ^ "FDA Approves Silicone Gel-Filled Breast Implants". FDA. Archived from the original on 2008-07-26. Retrieved 2008-07-01.
- ^ "March 25-26, 2019: General and Plastic Surgery Devices Panel of the Medical Devices Advisory Committee Meeting Announcement". U.S. Food and Drug Administration. April 25, 2019.
- ^ "The FDA Requests Allergan Voluntarily Recall Natrelle BIOCELL Textured Breast Implants and Tissue Expanders from the Market to Protect Patients: FDA Safety Communication". U.S. Food and Drug Administration. June 1, 2020.
- ^ a b "Breast Implant Questions and Answers". U.S. Food and Drug Administration. October 2010. Archived from the original on 2010-11-12.
External links
- Article "Expander-Implant Breast Reconstruction" at Medscape