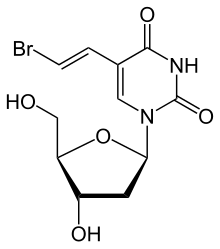
Brivudine
 | |
| Clinical data | |
|---|---|
| Trade names | Zostex, Mevir, Brivir, many others |
| Other names | BVDU |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
faeces | |
| Identifiers | |
| |
JSmol) | |
| Specific rotation | +9°±1° |
| Density | 1.86 g/cm3 |
| Melting point | 165 to 166 °C (329 to 331 °F) (decomposes) |
| |
| |
| | |
Brivudine (trade names Zostex, Mevir, Brivir, among others) is an
Medical uses
Brivudine is used for the treatment of herpes zoster in adult patients. It is taken orally once daily, in contrast to aciclovir, valaciclovir and other antivirals.[1] A study has found that it is more effective than aciclovir, but this has been disputed because of a possible conflict of interest on part of the study authors.[2]
Contraindications
The drug is contraindicated in patients undergoing
Adverse effects
The drug is generally well tolerated. The only common side effect is
Interactions
Brivudine interacts strongly and in rare cases lethally with the anticancer drug
There are no other relevant interactions. Brivudine does not significantly influence the cytochrome P450 enzymes in the liver.[1]
Pharmacology
Spectrum of activity
The drug inhibits
Mechanism of action
Brivudine is an analogue of the
Pharmacokinetics
Brivudine is well and rapidly absorbed from the gut and undergoes
Highest blood plasma concentrations are reached after one hour. Brivudine is almost completely (>95%)
-
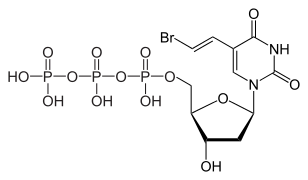
Brivudine 5'-triphosphate, the active metabolite
-
Bromovinyluracil (BVU), the main inactive metabolite
-
The acetic acid derivative predominantly found in urine
Chemistry
The molecule has three
Manufacturing
Main supplier is Berlin Chemie, now part of Italy's Menarini Group. In Central America is provided by Menarini Centro America and Wyeth.[citation needed]
History
The substance was first synthesized by scientists at the University of Birmingham in the UK in 1976. It was shown to be a potent inhibitor of HSV-1 and VZV by Erik De Clercq at the Rega Institute for Medical Research in Belgium in 1979. In the 1980s the drug became commercially available in East Germany, where it was marketed as Helpin by a pharmaceutical company called Berlin-Chemie. Only after the indication was changed to the treatment of herpes zoster in 2001 did it become more widely available in Europe.[7][8]
Brivudine is approved for use in a number of European countries including Austria, Belgium, Germany, Greece, Italy, Portugal, Spain and Switzerland.[9]
Etymology
The name brivudine derives from the chemical nomenclature bromo-vinyl-deoxyuridine or BVDU for short. It is sold under trade names such as Bridic, Brival, Brivex, Brivir, Brivirac, Brivox, Brivuzost, Zerpex, Zonavir, Zostex, and Zovudex.[9]
Research
A
See also
Related antiviral drugs
- Aciclovir
- Valacyclovir, a prodrug form of aciclovir
- Famciclovir, an analogue of Penciclovir with greater oral availability
- Foscarnet, an intravenous antiviral for aciclovir-resistant VZV
- Penciclovir, a topical preparation
Vaccines and other treatments
- Zostavax, a live virus Herpes zoster (shingles) vaccine
- Varivax, a live virus Varicella Zoster (chickenpox) vaccine
- Shingrix, a recombinant subunit vaccine for shingles
- VZV immune globulin, an antibody-based treatment for immune-suppressed patients with zoster
References
- ^ ISBN 978-3-85200-181-4.
- ^ "Brivudin (Zostex) besser als Aciclovir (Zovirax a.a.)?". Arznei-telegramm (in German). 5/2007.
- ^ "UAW – Aus Fehlern lernen - Potenziell tödlich verlaufende Wechselwirkung zwischen Brivudin (Zostex) und 5-Fluoropyrimidinen" (PDF). Deutsches Ärzteblatt (in German). 103 (27). 7 July 2006.
- ^ ISBN 3-7692-3483-9.
- PMID 6651877.
- ISBN 3-8047-1763-2.
- PMID 15548377.
- ^ Tringali C, ed. (2012). Bioactive Compounds from Natural Sources (2nd ed.). CRC Press. p. 170.
- ^ a b "Brivudine". drugs.com.
- PMID 25879115.