Buchner ring expansion
| Buchner ring expansion | |
|---|---|
| Named after | Eduard Buchner |
| Reaction type | Rearrangement reaction |
The Buchner ring expansion is a two-step organic

History
The Buchner ring expansion reaction was first used in 1885 by
Preparation
Preparation of ethyl-diazoacetate:
Buchner's first synthesis of cycloheptatriene derivatives in 1885 used
Preparation of the metal carbenoid:
Synthesis of the carbene in the 1960s was focused on using
Mechanism
Step 1:
The reaction mechanism of a Buchner ring expansion begins with carbene formation from ethyl-diazoacetate generated initially through photochemical or thermal reactions with extrusion of nitrogen.

The generated carbene adds to one of the double bonds of benzene to form the cyclopropane ring.

The advent of transition metal catalyzed reagents provides alternative stereospecific methods for cyclopropanation. The choices for metals include Cu, Rh and Ru with a variety of ligands.

The accepted carbene catalytic cycle[16] was proposed by Yates[17] in 1952. Initially the diazo compound oxidatively adds to the metal ligand complex. Following the extrusion of nitrogen the metal carbene is generated and reacts with an electron rich aromatic substance to reductively regenerate the metal catalyst completing the catalytic cycle.

Step 2:
The second step of the Buchner reaction involves a

The norcaradiene-cycloheptatriene

Equilibrium may be altered by varying

The

Applications
Medicine:
The importance of the Buchner ring expansion annulation chemistry is evident in the application of this synthetic sequence in the synthesis of biological compounds.
While studying an analogous reaction of carbene addition to thiophene, Stephen Matlin and Lam Chan applied the Buchner ring expansion method in 1981 to generate spiro derivatives of Penicillin.[7]

In 1998, Mander et al. synthesized the diterpenoid tropone, Harringtonolide[6] using the Buchner intramolecular ring expansion annulation chemistry. A rhodium catalyst (Rh2(mandelate)4) and DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) were used to generate the carbene. This natural product was found to have antineoplastic and antiviral properties.
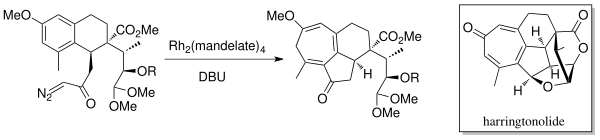
Danheiser et al. utilized
was synthesized using this ring expansion-annulation strategy with a rhodium catalyst (Rh2(OCOt-Bu)4) in ether.
Material Science:
The Buchner ring expansion method has been used to synthesize starting materials for applications in material science involving
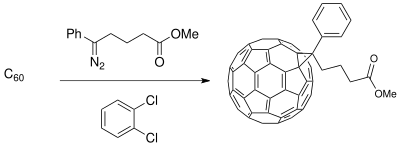
Limitations
The disadvantages of the reaction involve side reactions of the carbene moiety. The choice of solvent for the reaction needs to be considered. In addition to the potential for carbon-hydrogen bond insertion reactions, carbon-halogen carbene insertion is possible when dichloromethane is used as the solvent.[20]

Control for regioselectivity during the carbene addition is necessary to avoid side products resulting from conjugated cycloheptatriene isomers. Noels et al. used Rh(II) catalysts for carbene generation under mild reaction conditions (room temperature) to obtain regioselectively the kinetic non-conjugated cycloheptatriene isomer.[3][8][21]

See also
References
- ^ hdl:2268/237697
- ^ PMID 11277800
- ^ PMID 15575741
- ^
- ^
- ^
- doi:10.1039/A700080D
- ^
- ^ PMID 12683775
- ^
- PMID 11829610
- ^
- ProQuest 213566279
