Butyric acid
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Butanoic acid[1] | |||
| Other names
Ethylacetic acid
1-Propanecarboxylic acid Propylformic acid C4:0 ( Lipid numbers ) | |||
| Identifiers | |||
3D model (
JSmol ) |
| ||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider | |||
| DrugBank |
| ||
ECHA InfoCard
|
100.003.212 | ||
| EC Number |
| ||
IUPHAR/BPS |
| ||
| KEGG |
| ||
| MeSH | Butyric+acid | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII |
| ||
| UN number | 2820 | ||
CompTox Dashboard (EPA)
|
| ||
| |||
| |||
| Properties | |||
| C 3H 7COOH | |||
| Molar mass | 88.106 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Odor | Unpleasant, similar to vomit or body odor | ||
| Density | 1.135 g/cm3 (−43 °C)[2] 0.9528 g/cm3 (25 °C)[3] | ||
| Melting point | −5.1 °C (22.8 °F; 268.0 K)[3] | ||
| Boiling point | 163.75 °C (326.75 °F; 436.90 K)[3] | ||
| Sublimes at −35 °C ΔsublH | |||
| Miscible | |||
| Solubility | Miscible with ethanol, ether. Slightly soluble in CCl4 | ||
| log P | 0.79 | ||
| Vapor pressure | 0.112 kPa (20 °C) 0.74 kPa (50 °C) 9.62 kPa (100 °C)[4] | ||
Henry's law
constant (kH) |
5.35·10−4 L·atm/mol | ||
| Acidity (pKa) | 4.82 | ||
| −55.10·10−6 cm3/mol | |||
Thermal conductivity
|
1.46·105 W/m·K | ||
Refractive index (nD)
|
1.398 (20 °C)[3] | ||
| Viscosity | 1.814 cP (15 °C)[5] 1.426 cP (25 °C) | ||
| Structure | |||
| Monoclinic (−43 °C)[2] | |||
| C2/m[2] | |||
a = 8.01 Å, b = 6.82 Å, c = 10.14 Å[2] α = 90°, β = 111.45°, γ = 90°
| |||
| 0.93 D (20 °C)[5] | |||
| Thermochemistry | |||
Heat capacity (C)
|
178.6 J/mol·K[4] | ||
Std molar
entropy (S⦵298) |
222.2 J/mol·K[5] | ||
Std enthalpy of (ΔfH⦵298)formation |
−533.9 kJ/mol[4] | ||
Std enthalpy of (ΔcH⦵298)combustion |
2183.5 kJ/mol[4] | ||
| Hazards | |||
| GHS labelling: | |||
 [6] [6]
| |||
| Danger | |||
| H314[6] | |||
| P280, P305+P351+P338, P310[6] | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 71 to 72 °C (160 to 162 °F; 344 to 345 K)[6] | ||
| 440 °C (824 °F; 713 K)[6] | |||
Explosive limits
|
2.2–13.4% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
2000 mg/kg (oral, rat) | ||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Related carboxylic acids
|
Pentanoic acid
| ||
Related compounds
|
1-Butanol Butyraldehyde Methyl butyrate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Butyric acid ( and an important component in the mammalian gut.
History
Butyric acid was first observed in an impure form in 1814 by the French chemist
Occurrence
Triglycerides of butyric acid compose 3–4% of butter. When butter goes rancid, butyric acid is liberated from the glyceride by hydrolysis.[11] It is one of the fatty acid subgroup called short-chain fatty acids. Butyric acid is a typical carboxylic acid that reacts with bases and affects many metals.[12] It is found in
In humans, butyric acid is one of two primary endogenous agonists of human hydroxycarboxylic acid receptor 2 (HCA2), a Gi/o-coupled G protein-coupled receptor.[16][17]
Butyric acid is present as its octyl ester in parsnip (Pastinaca sativa)[18] and in the seed of the ginkgo tree.[19]
Production
Industrial
In industry, butyric acid is produced by
- H2 + CO + CH3CH=CH2 → CH3CH2CH2CHObutyric acid
It can be separated from aqueous solutions by saturation with salts such as calcium chloride. The calcium salt, Ca(C4H7O2)2 · H2O, is less soluble in hot water than in cold.
Microbial biosynthesis
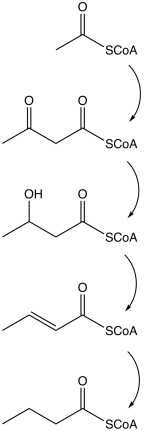
Butyrate is produced by several fermentation processes performed by obligate anaerobic bacteria.[20] This fermentation pathway was discovered by Louis Pasteur in 1861. Examples of butyrate-producing species of bacteria:
- Clostridium butyricum
- Clostridium kluyveri
- Clostridium pasteurianum
- Faecalibacterium prausnitzii
- Fusobacterium nucleatum
- Butyrivibrio fibrisolvens
- Eubacterium limosum
The pathway starts with the
- C6H12O6 → C4H8O2 + 2CO2 + 2H2
Other pathways to butyrate include succinate reduction and crotonate disproportionation.
| Action | Responsible enzyme |
|---|---|
| Acetyl coenzyme A converts into acetoacetyl coenzyme A |
acetyl-CoA-acetyl transferase
|
| Acetoacetyl coenzyme A converts into β-hydroxybutyryl CoA |
β-hydroxybutyryl-CoA dehydrogenase
|
| β-hydroxybutyryl CoA converts into crotonyl CoA |
crotonase
|
| Crotonyl CoA converts into butyryl CoA (CH3CH2CH2C=O−CoA) |
butyryl CoA dehydrogenase
|
| A phosphate group replaces CoA to form butyryl phosphate | phosphobutyrylase
|
| The phosphate group joins butyrate |
butyrate kinase |
Several species form
- Clostridium acetobutylicum, the most prominent acetone and butanol producer, used also in industry
- Clostridium beijerinckii
- Clostridium tetanomorphum
- Clostridium aurantibutyricum
These bacteria begin with butyrate fermentation, as described above, but, when the pH drops below 5, they switch into butanol and acetone production to prevent further lowering of the pH. Two molecules of butanol are formed for each molecule of acetone.
The change in the pathway occurs after acetoacetyl CoA formation. This intermediate then takes two possible pathways:
- acetoacetyl CoA → acetoacetate → acetone
- acetoacetyl CoA → butyryl CoA → butyraldehyde → butanol
For commercial purposes Clostridium species are used preferably for butyric acid or butanol production. The most common species used for probiotics is the Clostridium butyricum.[21]
Fermentable fiber sources
Highly-fermentable fiber residues, such as those from
Fructans are another source of prebiotic soluble dietary fibers which can be digested to produce butyrate.
Reactions
Butyric acid reacts as a typical carboxylic acid: it can form amide, ester, anhydride, and chloride derivatives.[29] The latter, butyryl chloride, is commonly used as the intermediate to obtain the others.
Uses
Butyric acid is used in the preparation of various butyrate esters. It is used to produce cellulose acetate butyrate (CAB), which is used in a wide variety of tools, paints, and coatings, and is more resistant to degradation than cellulose acetate.[30] CAB can degrade with exposure to heat and moisture, releasing butyric acid.[31]
Low-molecular-weight esters of butyric acid, such as methyl butyrate, have mostly pleasant aromas or tastes.[7] As a consequence, they are used as food and perfume additives. It is an approved food flavoring in the EU FLAVIS database (number 08.005).
Due to its powerful odor, it has also been used as a fishing bait additive.
Pharmacology
| Inhibited enzyme | IC50 (nM) | Entry note |
|---|---|---|
| HDAC1 | 16,000 | |
HDAC2 |
12,000 | |
| HDAC3 | 9,000 | |
| HDAC4 | 2,000,000 | Lower bound |
HDAC5 |
2,000,000 | Lower bound |
| HDAC6 | 2,000,000 | Lower bound |
| HDAC7 | 2,000,000 | Lower bound |
| HDAC8 | 15,000 | |
| HDAC9 | 2,000,000 | Lower bound |
CA1 |
511,000 | |
CA2 |
1,032,000 | |
GPCR target |
pEC50 |
Entry note |
FFAR2 |
2.9–4.6 | Full agonist |
FFAR3 |
3.8–4.9 | Full agonist |
| HCA2 | 2.8 | Agonist |
Pharmacodynamics
Butyric acid (pKa 4.82) is fully
Like other
Pharmacokinetics
Butyrate that is produced in the colon through microbial fermentation of dietary fiber is primarily absorbed and metabolized by
Metabolism
Butyric acid is metabolized by various human
- Adenosine triphosphate + butyric acid + coenzyme A → adenosine monophosphate + pyrophosphate + butyryl-CoA
As a
In humans, the butyrate precursor tributyrin, which is naturally present in butter, is metabolized by triacylglycerol lipase into dibutyrin and butyrate through the reaction:[46]
- Tributyrin + H2O → dibutyrin + butyric acid
Biochemistry
Butyrate has numerous effects on
In the mammalian gut
Butyrate is essential to host immune homeostasis.[37] Although the role and importance of butyrate in the gut is not fully understood, many researchers argue that a depletion of butyrate-producing bacteria in patients with several vasculitic conditions is essential to the pathogenesis of these disorders. A depletion of butyrate in the gut is typically caused by an absence or depletion of butyrate-producing-bacteria (BPB). This depletion in BPB leads to microbial dysbiosis. This is characterized by an overall low biodiversity and a depletion of key butyrate-producing members. Butyrate is an essential microbial metabolite with a vital role as a modulator of proper immune function in the host. It has been shown that children lacking in BPB are more susceptible to allergic disease[48] and Type 1 Diabetes.[49] Butyrate is also reduced in a diet low in dietary fiber, which can induce inflammation and have other adverse affects insofar as these short-chain fatty acids activate PPAR-γ.[50]
Butyrate exerts a key role for the maintenance of immune homeostasis both locally (in the gut) and systemically (via circulating butyrate). It has been shown to promote the differentiation of regulatory T cells. In particular, circulating butyrate prompts the generation of extrathymic regulatory T cells. The low-levels of butyrate in human subjects could favor reduced regulatory T cell-mediated control, thus promoting a powerful immuno-pathological T-cell response.[51] On the other hand, gut butyrate has been reported to inhibit local pro-inflammatory cytokines. The absence or depletion of these BPB in the gut could therefore be a possible aide in the overly-active inflammatory response. Butyrate in the gut also protects the integrity of the intestinal epithelial barrier. Decreased butyrate levels therefore lead to a damaged or dysfunctional intestinal epithelial barrier.[52]
In a 2013 research study conducted by Furusawa et al., microbe-derived butyrate was found to be essential in inducing the differentiation of colonic regulatory T cells in mice. This is of great importance and possibly relevant to the pathogenesis and vasculitis associated with many inflammatory diseases because regulatory T cells have a central role in the suppression of inflammatory and allergic responses.[53] In several research studies, it has been demonstrated that butyrate induced the differentiation of regulatory T cells in vitro and in vivo.[54] The anti-inflammatory capacity of butyrate has been extensively analyzed and supported by many studies. It has been found that microorganism-produced butyrate expedites the production of regulatory T cells, although the specific mechanism by which it does so unclear.[55] More recently, it has been shown that butyrate plays an essential and direct role in modulating gene expression of cytotoxic T-cells.[56] Butyrate also has an anti-inflammatory effect on neutrophils, reducing their migration to wounds. This effect is mediated via the receptor HCA1[57]
In the gut microbiomes found in the class Mammalia, omnivores and herbivores have butyrate-producing bacterial communities dominated by the butyryl-CoA:acetate CoA-transferase pathway, whereas carnivores have butyrate-producing bacterial communities dominated by the butyrate kinase pathway.[58]
The odor of butyric acid, which emanates from the sebaceous follicles of all mammals, works on the tick as a signal.
Immunomodulation and inflammation
Butyrate's effects on the immune system are mediated through the inhibition of class I
Butyrate has established antimicrobial properties in humans that are mediated through the
Similar to other HCA2 agonists studied, butyrate also produces marked anti-inflammatory effects in a variety of tissues, including the brain, gastrointestinal tract, skin, and vascular tissue.[63][64][65] Butyrate binding at FFAR3 induces neuropeptide Y release and promotes the functional homeostasis of colonic mucosa and the enteric immune system.[66]
Cancer
Butyrate has been shown to be a critical mediator of the colonic inflammatory response. It is responsible for about 70% of energy from the colonocytes, being a critical SCFA in colon
In summary, the production of
Conversely, some researchers have sought to eliminate butyrate and consider it a potential cancer driver.[74] Studies in mice indicate it drives transformation of MSH2-deficient colon epithelial cells.[75]
Potential treatments from butyrate restoration
Owing to the importance of butyrate as an inflammatory regulator and immune system contributor, butyrate depletions could be a key factor influencing the pathogenesis of many
Addiction
Butyric acid is an
Butyrate salts and esters
The butyrate or butanoate
Examples
Salts
Esters
- Butyl butyrate
- Butyryl-CoA
- Cellulose acetate butyrate (aircraft dope)
- Estradiol benzoate butyrate
- Ethyl butyrate
- Methyl butyrate
- Pentyl butyrate
- Tributyrin
See also
- List of saturated fatty acids
- Hershey's milk chocolate
- Histone
- Histone-modifying enzyme
- Histone acetylase
- Histone deacetylase
- Hydroxybutyric acids
- α-Hydroxybutyric acid
- β-Hydroxybutyric acid
- γ-Hydroxybutyric acid
- Oxobutyric acids
- 2-Oxobutyric acid(α-ketobutyric acid)
- 3-Oxobutyric acid(acetoacetic acid)
- 4-Oxobutyric acid(succinic semialdehyde)
- β-Methylbutyric acid
Notes
- ^ Most of the butyrate that is absorbed into blood plasma from the colon enters the circulatory system via the portal vein; most of the butyrate that enters the circulatory system by this route is taken up by the liver.[39]
References
This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Butyric Acid". Encyclopædia Britannica (11th ed.). Cambridge University Press.
- ISBN 978-0-85404-182-4.
- ^ .
- ^ ISBN 978-1-4200-9084-0.
- ^ a b c d e Butanoic acid in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved 27 October 2020)
- ^ a b c "Butanoic acid". Chemister.ru. 19 March 2007. Retrieved 27 October 2020.
- ^ a b c d e Sigma-Aldrich Co., Butyric acid. Retrieved on 27 October 2020.
- ^ ISBN 978-3527306732.
- ^ Chevreul (1815) "Lettre de M. Chevreul à MM. les rédacteurs des Annales de chimie" (Letter from Mr. Chevreul to the editors of the Annals of Chemistry), Annales de chimie, 94 : 73–79; in a footnote spanning pages 75–76, he mentions that he had found a substance that is responsible for the smell of butter.
- ^ Chevreul (1817) "Extrait d'une lettre de M. Chevreul à MM. les Rédacteurs du Journal de Pharmacie" (Extract of a letter from Mr. Chevreul to the editors of the Journal of Pharmacy), Journal de Pharmacie et des sciences accessoires, 3 : 79–81. On p. 81, he named butyric acid: "Ce principe, que j'ai appelé depuis acid butérique, … " (This principle [i.e., constituent], which I have since named "butyric acid", … )
- ^ E. Chevreul, Recherches chimiques sur les corps gras d'origine animale [Chemical researches on fatty substances of animal origin] (Paris, France: F.G. Levrault, 1823), pages 115–133.
- .
- ^ ICSC 1334 – Butyric acid. Inchem.org (23 November 1998). Retrieved on 2020-10-27.
- ^ PMID 29231905.
- ^ PMID 26963409.
- ^ "Butyric acid". The Good Scents Company. Retrieved 26 October 2020.
- ^ PMID 21454438.
- ^ a b Offermanns S, Colletti SL, IJzerman AP, Lovenberg TW, Semple G, Wise A, Waters MG. "Hydroxycarboxylic acid receptors". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 13 July 2018.
- S2CID 23512190.
- ISBN 978-0-7167-1007-3. Retrieved 11 October 2018.
- PMID 18218779.
- .
- ^ PMID 14747692.
- PMID 11157351.
- PMID 1779073.
- ^ PMID 27446020.
- ^ "Frequently asked questions in the area of diet and IBS". Department of Gastroenterology Translational Nutrition Science, Monash University, Victoria, Australia. Retrieved 24 March 2016.
- S2CID 20666740.
- PMID 25680668.
- ISBN 978-0-85186-884-4.
- ^ Lokensgard, Erik (2015). Industrial Plastics: Theory and Applications (6th ed.). Cengage Learning.
- ^ Williams, R. Scott. "Care of Plastics: Malignant plastics". WAAC Newsletter. Vol. 24, no. 1. Conservation OnLine. Retrieved 29 May 2017.
- ^ Freezer Baits Archived 25 January 2010 at the Wayback Machine, nutrabaits.net
- .
- ^ Japanese Whalers Injured by Acid-Firing Activists Archived 8 June 2010 at the Wayback Machine, newser.com, 10 February 2010
- ^ a b c d "Butyric acid". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 13 July 2018.
- ^ a b "Butanoic acid and Sodium butyrate". BindingDB. The Binding Database. Retrieved 27 October 2020.
- ^ PMID 25875123.
Short-chain fatty acids (SCFAs) such as acetate, butyrate, and propionate, which are produced by gut microbial fermentation of dietary fiber, are recognized as essential host energy sources and act as signal transduction molecules via G-protein coupled receptors (FFAR2, FFAR3, OLFR78, GPR109A) and as epigenetic regulators of gene expression by the inhibition of histone deacetylase (HDAC). Recent evidence suggests that dietary fiber and the gut microbial-derived SCFAs exert multiple beneficial effects on the host energy metabolism not only by improving the intestinal environment, but also by directly affecting various host peripheral tissues.
- ^
- ^ PMID 26868600.
- ISSN 0312-5963. Retrieved 18 March 2024.
the middle and inferior rectal veins drain the lower part of the rectum and venous blood is returned to the inferior vena cava. Therefore, drugs absorbed in the latter system will be delivered preferentially to the systemic circulation, bypassing the liver and avoiding first-pass metabolism
- PMID 15717057.
Other in vivo studies in our laboratories indicated that several compounds including acetate, propionate, butyrate, benzoic acid, salicylic acid, nicotinic acid, and some β-lactam antibiotics may be transported by the MCT at the BBB.21 ... Uptake of valproic acid was reduced in the presence of medium-chain fatty acids such as hexanoate, octanoate, and decanoate, but not propionate or butyrate, indicating that valproic acid is taken up into the brain via a transport system for medium-chain fatty acids, not short-chain fatty acids.
- ^ PMID 23789956.
Monocarboxylate transporters (MCTs) are known to mediate the transport of short chain monocarboxylates such as lactate, pyruvate and butyrate. ... MCT1 and MCT4 have also been associated with the transport of short chain fatty acids such as acetate and formate which are then metabolized in the astrocytes [78]. ... SLC5A8 is expressed in normal colon tissue, and it functions as a tumor suppressor in human colon with silencing of this gene occurring in colon carcinoma. This transporter is involved in the concentrative uptake of butyrate and pyruvate produced as a product of fermentation by colonic bacteria.
- ^ a b Butyric acid. University of Alberta. Retrieved 15 August 2015.
{{cite encyclopedia}}:|website=ignored (help) - ^ "Butanoate metabolism – Reference pathway". Kyoto Encyclopedia of Genes and Genomes. Kanehisa Laboratories. 1 November 2017. Retrieved 1 February 2018.
- PMID 21531334.
- ^ "triacylglycerol lipase – Homo sapiens". BRENDA. Technische Universität Braunschweig. Retrieved 25 May 2015.
- S2CID 22633025.
- PMID 31279007.
- PMID 30356183.
- S2CID 220337072.
- PMID 25435420.
- PMID 30077182.
- PMID 29067994.
- S2CID 4408815.
- PMID 24226773.
- PMID 30258117.
- PMID 33064972.
- PMID 25343515.
- ^
- PMID 22194341.
- PMID 21464244.
- PMID 22517765.
- PMID 25766751.
- PMID 23526298.
- PMID 26773933.
- PMID 25545642.
- PMID 24567795.
- S2CID 148568580.
- PMID 15213223.
- PMID 19707587.
- S2CID 18573671.
- PMID 22517765.
- PMID 18655767.
- ^ "Low-carb diet cuts risk of colon cancer, study finds | University of Toronto Media Room". media.utoronto.ca. Retrieved 4 May 2016.
- PMID 25036629.
- PMID 1612357.
- PMID 21989194.
- PMID 23643695.
- ^ PMID 25486626.
- S2CID 40791780.
- PMID 28682229.



