PD-L1
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 9: 5.45 – 5.47 Mb | Chr 19: 29.34 – 29.37 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
Programmed death-ligand 1 (PD-L1) also known as cluster of differentiation 274 (CD274) or B7 homolog 1 (B7-H1) is a protein that in humans is encoded by the CD274 gene.[5]
Programmed death-ligand 1 (PD-L1) is a 40kDa type 1
History
PD-L1 also known as B7-H1 was characterized at the Mayo Clinic in 1999 as an immune regulatory molecule.[7] At that time, it was concluded that B7-H1 helps tumor cells evade anti-tumor immunity.[8] In 2003, B7-H1 was shown to be expressed on myeloid cells as checkpoint protein and was proposed as potential target in cancer immunotherapy in human clinic.[9]
Binding
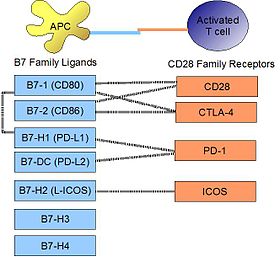
PD-L1 binds to its receptor,
Signaling
Engagement of PD-L1 with its receptor
Regulation
By interferons
Upon
On macrophages and monocytes
PD-L1 is notably expressed on
Role of microRNAs
Resting human cholangiocytes express PD-L1 mRNA, but not the protein, due to translational suppression by microRNA miR-513.[19] Upon treatment with interferon-gamma, miR-513 was down-regulated, thereby lifting suppression of PD-L1 protein. In this way, interferon-gamma can induce PD-L1 protein expression by inhibiting gene-mediated suppression of mRNA translation. Whereas the Epstein-Barr viral (EBV) latent membrane protein-1 (LMP1) is a known potent inducer of PD-L1, the EBV miRNA miR-BamH1 fragment H rightward open reading frame 1 (BHRF1) 2-5p has been shown to regulate LMP1 induced PD-L1 expression.[20]
Epigenetic regulation
PD-L1 promoter DNA methylation may predict survival in some cancers after surgery.[21]
Clinical significance
Cancer
PD-L1 is shown to be highly expressed in a variety of malignancies, particularly lung cancer. In order to anticipate the effectiveness of gene therapy or systemic immunotherapy in blocking the PD-1 and PD-L1 checkpoints, PD-L1 might be employed as a prognostic marker and a target for anti-cancer immunity.[22] i.e. upregulation of PD-L1 may allow cancers to evade the host immune system. For example, an analysis of 196 tumor specimens from patients with renal cell carcinoma found that high tumor expression of PD-L1 was associated with increased tumor aggressiveness and a 4.5-fold increased risk of death.[23]
Many
Upregulation of PD-L1 on immune cells (especially
PD-L1 analysis in TNBC is essential for selecting patients eligible for immunotherapy. Inter-observer and intra-observer agreement among the pathologists were found to be substantial. Cases around the 1% cut-off value are specifically challenging.[31]
Listeria monocytogenes
In a mouse model of intracellular infection, L. monocytogenes induced PD-L1 protein expression in T cells, NK cells, and macrophages. PD-L1 blockade (using blocking antibodies) resulted in increased mortality for infected mice. Blockade reduced TNFα and nitric oxide production by macrophages, reduced granzyme B production by NK cells, and decreased proliferation of L. monocytogenes antigen-specific CD8 T cells (but not CD4 T cells).[32] This evidence suggests that PD-L1 acts as a positive costimulatory molecule in intracellular infection.
Autoimmunity
PD-1/PD-L1 interaction is thought to play a role in preventing destructive autoimmunity, especially during inflammatory conditions. The best example is in the stomach, where PD-1 expression protects the
In humans, PD-L1 was found to have altered expression in pediatric patients with
See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000120217 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000016496 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Entrez Gene: CD274 CD274 molecule".
- PMID 15240681.
- S2CID 21397460.
- S2CID 27694471.
- S2CID 12499214.
- PMID 18585785.
- PMID 20208540.
- S2CID 85034305.
- PMID 21739608.
- PMID 17414316.
- S2CID 11169726.
- PMID 12421930.
- PMID 12697896.
- S2CID 195259881.
- PMID 19155478.
- PMID 31856276.
- PMID 27835597.
- PMID 35720000.
- PMID 15569934.
- PMID 24217091.
- ^ "Immune checkpoint inhibitors to treat cancer". www.cancer.org. Retrieved 2017-03-27.
- PMID 28884042.
- PMID 30936321.
- PMID 32439799.
- S2CID 251068796.
- S2CID 248083925.
- ^ Zaakouk, M.; Van Bockstal, M.; Galant, C.; Callagy, G.; Provenzano, E.; Hunt, R.; D’Arrigo, C.; Badr, N.M.; O’Sullivan, B.; Starczynski, J.; Tanchel, B.; Mir, Y.; Lewis, P.; Shaaban, A.M. Inter- and Intra-Observer Agreement of PD-L1 SP142 Scoring in Breast Carcinoma—A Large Multi-Institutional International Study. Cancers 2023, 15, 1511. https://doi.org/10.3390/cancers15051511
- PMID 17971153.
- PMID 35899973.
- PMID 12847137.
- PMID 18650228.
External links
- CD274+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: Q9NZQ7 (Programmed cell death 1 ligand 1) at the PDBe-KB.
