Cadogan–Sundberg indole synthesis
Source: Wikipedia, the free encyclopedia.
Reaction in organic chemistry
| Cadogan–Sundberg indole synthesis | |
|---|---|
| Named after | John Cadogan Richard J. Sundberg |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000509 |
The Cadogan–Sundberg indole synthesis, or simply Cadogan indole synthesis, is a
indoles from o-nitrostyrenes with the use of trialkyl phosphites, such as triethyl phosphite.[1][2][3][4][5][6][7]
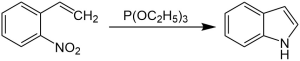
Mechanism
o-nitrostyrene first reacts with triethyl phosphite, and the nitro group is converted to a nitroso group. The nitroso group then reacts with the alkene, and N-hydroxylindole is formed, which reacts again with triethyl phosphite to form the indole.[1][2][3][4]

Application
The Cadogan–Sundberg indole synthesis has been used as an intermediate step in the total synthesis of Tjipanazole E,[8] transforming 2-[trans-2-[5-Chloro-2-nitrophenyl)vinyl]-5-chloro-1H-indole to 5,5’-Dichloro-2,2’-biindole.
References
- ^ .
- ^ ISBN 9780470638859.
- ^ .
- ^ .
- ISBN 9781118695692.
- .
- ISBN 978-3-319-03979-4.
- PMID 14507214.
This chemical reaction article is a stub. You can help Wikipedia by expanding it. |
