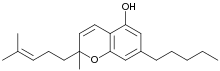
Cannabichromene
| |

| |
| Names | |
|---|---|
| IUPAC name
2-Methyl-2-(4-methylpent-3-enyl)-7-pentyl-5-chromenol
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.236.929 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H30O2 | |
| Molar mass | 314.469 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cannabichromene (CBC), also called cannabichrome, cannanbichromene, pentylcannabichromene or cannabinochromene,[1] exhibits anti-inflammatory properties in vitro, which may, theoretically, contribute to cannabis analgesic effects.[2] It is a
Biosynthesis
Within the Cannabis plant, CBC occurs mainly as cannabichromenic acid (CBCA, 2-COOH-CBC, CBC-COOH). Geranyl pyrophosphate and olivetolic acid combine to produce cannabigerolic acid (CBGA; the sole intermediate for all other phytocannabinoids), which is cyclized by the enzyme CBCA synthase to form CBCA. Over time, or when heated above 93 °C, CBCA is decarboxylated, producing CBC. See also the biosynthetic scheme image below.[citation needed]

Pharmacology
Cannabichromene has been hypothesized to affect THC psychoactivity, though in vivo effects have not been demonstrated.
References
- ^ "Cannabichromene". PubChem. National Center for Biotechnology Information. 16 February 2019. Retrieved 12 February 2019.
- PMID 28120232.
- ^ PMID 28120231.
- PMID 26836472.
- S2CID 827221.
- ^ "What Is CBC (Cannabichromene)?". CNBS. Retrieved 2019-03-31.
- S2CID 1341744.
- PMID 33395525
- ^ PMID 33230154.
- PMID 25311884.
