Carbon cycle

| Part of a series on the |
| Carbon cycle |
|---|
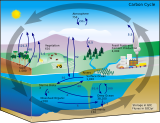 |
The carbon cycle is that part of the biogeochemical cycle by which carbon is exchanged among the biosphere, pedosphere, geosphere, hydrosphere, and atmosphere of Earth. Other major biogeochemical cycles include the nitrogen cycle and the water cycle. Carbon is the main component of biological compounds as well as a major component of many minerals such as limestone. The carbon cycle comprises a sequence of events that are key to making Earth capable of sustaining life. It describes the movement of carbon as it is recycled and reused throughout the biosphere, as well as long-term processes of carbon sequestration (storage) to and release from carbon sinks.
To describe the dynamics of the carbon cycle, a distinction can be made between the fast and slow carbon cycle. The fast carbon cycle is also referred to as the biological carbon cycle. Fast carbon cycles can complete within years, moving substances from atmosphere to biosphere, then back to the atmosphere. Slow or geological cycles (also called deep carbon cycle) can take millions of years to complete, moving substances through the Earth's crust between rocks, soil, ocean and atmosphere.[2]
Humans have disturbed the carbon cycle for many centuries. They have done so by
Main compartments
The carbon cycle was first described by
- Atmosphere
- Terrestrial biosphere
- Ocean, including dissolved inorganic carbon and living and non-living marine biota
- Sediments, including fossil fuels, freshwater systems, and non-living organic material.
- Earth's interior (mantle and crust). These carbon stores interact with the other components through geological processes.
The carbon exchanges between reservoirs occur as the result of various chemical, physical, geological, and biological processes. The ocean contains the largest active pool of carbon near the surface of the Earth.[7] The natural flows of carbon between the atmosphere, ocean, terrestrial ecosystems, and sediments are fairly balanced; so carbon levels would be roughly stable without human influence.[8][9]
Atmosphere
Carbon in the Earth's atmosphere exists in two main forms: carbon dioxide and methane. Both of these gases absorb and retain heat in the atmosphere and are partially responsible for the greenhouse effect.[7] Methane produces a larger greenhouse effect per volume as compared to carbon dioxide, but it exists in much lower concentrations and is more short-lived than carbon dioxide. Thus, carbon dioxide contributes more to the global greenhouse effect than methane.[11]
Carbon dioxide is removed from the atmosphere primarily through photosynthesis and enters the terrestrial and oceanic biospheres. Carbon dioxide also dissolves directly from the atmosphere into bodies of water (ocean, lakes, etc.), as well as dissolving in precipitation as raindrops fall through the atmosphere. When dissolved in water, carbon dioxide reacts with water molecules and forms carbonic acid, which contributes to ocean acidity. It can then be absorbed by rocks through weathering. It also can acidify other surfaces it touches or be washed into the ocean.[12]
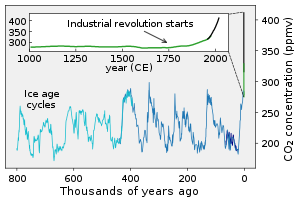
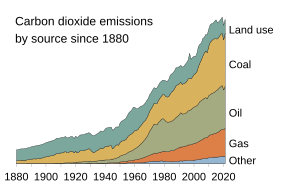
Human activities over the past two centuries have increased the amount of carbon in the atmosphere by nearly 50% as of year 2020, mainly in the form of carbon dioxide, both by modifying ecosystems' ability to extract carbon dioxide from the atmosphere and by emitting it directly, e.g., by burning fossil fuels and manufacturing concrete.[3][7]
In the far future (2 to 3 billion years), the rate at which carbon dioxide is absorbed into the soil via the carbonate–silicate cycle will likely increase due to expected changes in the sun as it ages. The expected increased luminosity of the Sun will likely speed up the rate of surface weathering.[13] This will eventually cause most of the carbon dioxide in the atmosphere to be squelched into the Earth's crust as carbonate.[14][15][16] Once the concentration of carbon dioxide in the atmosphere falls below approximately 50 parts per million (tolerances vary among species), C3 photosynthesis will no longer be possible.[15] This has been predicted to occur 600 million years from the present, though models vary.[17]
Once the oceans on the Earth evaporate in about 1.1 billion years from now,[13] plate tectonics will very likely stop due to the lack of water to lubricate them. The lack of volcanoes pumping out carbon dioxide will cause the carbon cycle to end between 1 billion and 2 billion years into the future.[18]
Terrestrial biosphere
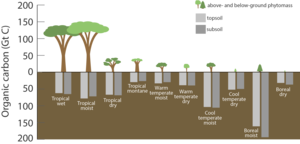
The terrestrial biosphere includes the organic carbon in all land-living organisms, both alive and dead, as well as carbon stored in
Because carbon uptake in the terrestrial biosphere is dependent on biotic factors, it follows a diurnal and seasonal cycle. In CO2 measurements, this feature is apparent in the

Carbon leaves the terrestrial biosphere in several ways and on different time scales. The combustion or respiration of organic carbon releases it rapidly into the atmosphere. It can also be exported into the ocean through rivers or remain sequestered in soils in the form of inert carbon.[23] Carbon stored in soil can remain there for up to thousands of years before being washed into rivers by erosion or released into the atmosphere through soil respiration. Between 1989 and 2008 soil respiration increased by about 0.1% per year.[24] In 2008, the global total of CO2 released by soil respiration was roughly 98 billion tonnes[citation needed], about 3 times more carbon than humans are now putting into the atmosphere each year by burning fossil fuel (this does not represent a net transfer of carbon from soil to atmosphere, as the respiration is largely offset by inputs to soil carbon).[citation needed] There are a few plausible explanations for this trend, but the most likely explanation is that increasing temperatures have increased rates of decomposition of soil organic matter, which has increased the flow of CO2. The length of carbon sequestering in soil is dependent on local climatic conditions and thus changes in the course of climate change.[25]
| Pool | Quantity (gigatons) |
|---|---|
| Atmosphere | 720 |
| Ocean (total) | 38,400 |
| Total inorganic | 37,400 |
| Total organic | 1,000 |
| Surface layer | 670 |
| Deep layer | 36,730 |
| Lithosphere | |
| Sedimentary carbonates | > 60,000,000 |
| Kerogens | 15,000,000 |
| Terrestrial biosphere (total) | 2,000 |
| Living biomass | 600 – 1,000 |
| Dead biomass | 1,200 |
| Aquatic biosphere | 1 – 2 |
| Fossil fuels (total) | 4,130 |
| Coal | 3,510 |
| Oil | 230 |
| Gas | 140 |
| Other (peat) | 250 |
Ocean
The ocean can be conceptually divided into a surface layer within which water makes frequent (daily to annual) contact with the atmosphere, and a deep layer below the typical mixed layer depth of a few hundred meters or less, within which the time between consecutive contacts may be centuries. The dissolved inorganic carbon (DIC) in the surface layer is exchanged rapidly with the atmosphere, maintaining equilibrium. Partly because its concentration of DIC is about 15% higher[26] but mainly due to its larger volume, the deep ocean contains far more carbon—it is the largest pool of actively cycled carbon in the world, containing 50 times more than the atmosphere[7]—but the timescale to reach equilibrium with the atmosphere is hundreds of years: the exchange of carbon between the two layers, driven by thermohaline circulation, is slow.[7]
Carbon enters the ocean mainly through the dissolution of atmospheric carbon dioxide, a small fraction of which is converted into carbonate. It can also enter the ocean through rivers as dissolved organic carbon. It is converted by organisms into organic carbon through photosynthesis and can either be exchanged throughout the food chain or precipitated into the oceans' deeper, more carbon-rich layers as dead soft tissue or in shells as calcium carbonate. It circulates in this layer for long periods of time before either being deposited as sediment or, eventually, returned to the surface waters through thermohaline circulation.[8]
Oceans are basic (with a current
Geosphere

The geologic component of the carbon cycle operates slowly in comparison to the other parts of the global carbon cycle. It is one of the most important determinants of the amount of carbon in the atmosphere, and thus of global temperatures.[29]
Most of the Earth's carbon is stored inertly in the Earth's lithosphere.[7] Much of the carbon stored in the Earth's mantle was stored there when the Earth formed.[30] Some of it was deposited in the form of organic carbon from the biosphere.[31] Of the carbon stored in the geosphere, about 80% is limestone and its derivatives, which form from the sedimentation of calcium carbonate stored in the shells of marine organisms. The remaining 20% is stored as kerogens formed through the sedimentation and burial of terrestrial organisms under high heat and pressure. Organic carbon stored in the geosphere can remain there for millions of years.[29]
Carbon can leave the geosphere in several ways. Carbon dioxide is released during the
Types of dynamic

The fast carbon cycle operates through the biosphere, see diagram at start of article ↑
There is a fast and a slow carbon cycle. The fast cycle operates in the biosphere and the slow cycle operates in rocks. The fast or biological cycle can complete within years, moving carbon from atmosphere to biosphere, then back to the atmosphere. The slow or geological cycle may extend deep into the mantle and can take millions of years to complete, moving carbon through the Earth's crust between rocks, soil, ocean and atmosphere.[2]
The fast carbon cycle involves relatively short-term
The slow (or deep) carbon cycle involves medium to long-term
Sub-processes within fast carbon cycle
Terrestrial carbon in the water cycle

The movement of terrestrial carbon in the water cycle is shown in the diagram on the right and explained below: [38]
- Atmospheric particles act as cloud condensation nuclei, promoting cloud formation.[39][40]
- Raindrops absorb
- Burning and volcanic eruptions produce highly condensed polycyclic aromatic molecules (i.e. black carbon) that is returned to the atmosphere along with greenhouse gases such as CO2.[43][44]
- Terrestrial plants fix atmospheric CO2 through photosynthesis, returning a fraction back to the atmosphere through respiration.[45] Lignin and celluloses represent as much as 80% of the organic carbon in forests and 60% in pastures.[46][47]
- Water absorbs plant and settled aerosol-derived
- Organic carbon derived from the terrestrial biosphere and in situ photo-oxidation), resulting in a flux of CO2 from rivers to the atmosphere that are the same order of magnitude as the amount of carbon sequestered annually by the terrestrial biosphere.[56][57][58] Terrestrially-derived macromolecules such as lignin [59] and black carbon [60] are decomposed into smaller components and monomers, ultimately being converted to CO2, metabolic intermediates, or biomass.
- Lakes, reservoirs, and
- Primary production is typically enhanced in estuarine waters are a source of CO2 to the atmosphere, globally.[65]
- open ocean typically absorb CO2 from the atmosphere.[65]
- The marine biological pump sequesters a small but significant fraction of the absorbed CO2 as organic carbon in marine sediments (see below).[70][38]
Terrestrial runoff to the ocean

Terrestrial and marine ecosystems are chiefly connected through
Riverine transport, being the main connective channel of these pools, will act to transport
Biological pump in the ocean

The ocean
Most carbon incorporated in organic and inorganic biological matter is formed at the sea surface where it can then start sinking to the ocean floor. The deep ocean gets most of its nutrients from the higher water column when they sink down in the form of marine snow. This is made up of dead or dying animals and microbes, fecal matter, sand and other inorganic material.[83]
The biological pump is responsible for transforming
A single phytoplankton cell has a sinking rate around one metre per day. Given that the average depth of the ocean is about four kilometres, it can take over ten years for these cells to reach the ocean floor. However, through processes such as coagulation and expulsion in predator fecal pellets, these cells form aggregates. These aggregates have sinking rates orders of magnitude greater than individual cells and complete their journey to the deep in a matter of days.[85]
About 1% of the particles leaving the surface ocean reach the seabed and are consumed, respired, or buried in the sediments. The net effect of these processes is to remove carbon in organic form from the surface and return it to DIC at greater depths, maintaining a surface-to-deep ocean gradient of DIC.
Sub-processes within slow carbon cycle

Slow or deep carbon cycling is an important process, though it is not as well-understood as the relatively fast carbon movement through the atmosphere, terrestrial biosphere, ocean, and geosphere.[86] The deep carbon cycle is intimately connected to the movement of carbon in the Earth's surface and atmosphere. If the process did not exist, carbon would remain in the atmosphere, where it would accumulate to extremely high levels over long periods of time.[87] Therefore, by allowing carbon to return to the Earth, the deep carbon cycle plays a critical role in maintaining the terrestrial conditions necessary for life to exist.
Furthermore, the process is also significant simply due to the massive quantities of carbon it transports through the planet. In fact, studying the composition of basaltic
Carbon in the lower mantle

Carbon principally enters the mantle in the form of
However, carbonates descending to the lower mantle encounter other fates in addition to forming diamonds. In 2011, carbonates were subjected to an environment similar to that of 1800 km deep into the Earth, well within the lower mantle. Doing so resulted in the formations of

Accordingly, carbon can remain in the lower mantle for long periods of time, but large concentrations of carbon frequently find their way back to the lithosphere. This process, called carbon outgassing, is the result of carbonated mantle undergoing decompression melting, as well as mantle plumes carrying carbon compounds up towards the crust.[98] Carbon is oxidised upon its ascent towards volcanic hotspots, where it is then released as CO2. This occurs so that the carbon atom matches the oxidation state of the basalts erupting in such areas.[99]

Carbon in the core
Although the presence of carbon in the Earth's core is well-constrained, recent studies suggest large inventories of carbon could be stored in this region.[
Human influence on fast carbon cycle
Climate change

as represented in a stylised model
Current trends in climate change lead to higher ocean temperatures and
The exchanges of carbon between the atmosphere and other components of the Earth system, collectively known as the carbon cycle, currently constitute important negative (dampening) feedbacks on the effect of anthropogenic carbon emissions on climate change. Carbon sinks in the land and the ocean each currently take up about one-quarter of anthropogenic carbon emissions each year.[108][105]
These feedbacks are expected to weaken in the future, amplifying the effect of anthropogenic carbon emissions on climate change.[109] The degree to which they will weaken, however, is highly uncertain, with Earth system models predicting a wide range of land and ocean carbon uptakes even under identical atmospheric concentration or emission scenarios.[110][105][111] Arctic methane emissions indirectly caused by anthropogenic global warming also affect the carbon cycle and contribute to further warming.
Fossil carbon extraction and burning

The largest and one of the fastest growing human impacts on the carbon cycle and biosphere is the extraction and burning of
.As of 2020[update], about 450 gigatons of fossil carbon have been extracted in total; an amount approaching the carbon contained in all of Earth's living terrestrial biomass.[112] Recent rates of global emissions directly into the atmosphere have exceeded the uptake by vegetation and the oceans.[114][115][116][117] These sinks have been expected and observed to remove about half of the added atmospheric carbon within about a century.[112][118][119] Nevertheless, sinks like the ocean have evolving saturation properties, and a substantial fraction (20–35%, based on coupled models) of the added carbon is projected remain in the atmosphere for centuries to millennia.[120][121]
Halocarbons
Halocarbons are less prolific compounds developed for diverse uses throughout industry; for example as
Land use changes
Since the invention of agriculture, humans have directly and gradually influenced the carbon cycle over century-long timescales by modifying the mixture of vegetation in the terrestrial biosphere.[118] Over the past several centuries, direct and indirect human-caused land use and land cover change (LUCC) has led to the loss of biodiversity, which lowers ecosystems' resilience to environmental stresses and decreases their ability to remove carbon from the atmosphere. More directly, it often leads to the release of carbon from terrestrial ecosystems into the atmosphere.
Deforestation for agricultural purposes removes forests, which hold large amounts of carbon, and replaces them, generally with agricultural or urban areas. Both of these replacement land cover types store comparatively small amounts of carbon so that the net result of the transition is that more carbon stays in the atmosphere. However, the effects on the atmosphere and overall carbon cycle can be intentionally and/or naturally reversed with reforestation.
See also
- Biogeochemical cycle – Chemical transfer pathway between Earth's biological and non-biological parts
- Climate change mitigation – Actions to reduce net greenhouse gas emissions to limit climate change
- Carbon dioxide in Earth's atmosphere – Atmospheric constituent and greenhouse gas
- Carbon sequestration – Storing carbon in a carbon pool (natural as well as enhanced or artificial processes)
- Carbonate–silicate cycle – Geochemical transformation of silicate rocks
- Ocean acidification – Decrease of pH levels in the ocean
- Permafrost carbon cycle – Sub-cycle of the larger global carbon cycle
References
- ^ a b c Riebeek, Holli (16 June 2011). "The Carbon Cycle". Earth Observatory. NASA. Archived from the original on 5 March 2016. Retrieved 5 April 2018.
- ^ ISBN 9781136294822.
- ^ NOAAGlobal Monitoring Laboratory/Earth System Research Laboratories. Retrieved 30 October 2020.
- ^ "What is Ocean Acidification?". National Ocean Service, National Oceanic and Atmospheric Administration. Retrieved 30 October 2020.
- ISBN 978-0-375-42222-5.
- ISBN 9781400837076.
- ^ PMID 11030643.
- ^ .
- ^ "An Introduction to the Global Carbon Cycle" (PDF). University of New Hampshire. 2009. Archived (PDF) from the original on 8 October 2016. Retrieved 6 February 2016.
- ^ A Year In The Life Of Earth's CO2 Archived 26 December 2021 at the Wayback Machine NASA: Goddard Space Flight Center, 17 November 2014.
- ^ Forster, P.; Ramawamy, V.; Artaxo, P.; Berntsen, T.; Betts, R.; Fahey, D.W.; Haywood, J.; Lean, J.; Lowe, D.C.; Myhre, G.; Nganga, J.; Prinn, R.; Raga, G.; Schulz, M.; Van Dorland, R. (2007). "Changes in atmospheric constituents and in radiative forcing". Climate Change 2007: The Physical Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change.
- ^ "Many Planets, One Earth // Section 4: Carbon Cycling and Earth's Climate". Many Planets, One Earth. 4. Archived from the original on 17 April 2012. Retrieved 24 June 2012.
- ^ S2CID 73722450.
- ISSN 0148-0227.
- ^ arXiv:0912.2482 [astro-ph.EP].
- .
- .
- ISBN 978-0-521-11294-9.
- ^ .
- ^ Rice, Charles W. (January 2002). "Storing carbon in soil: Why and how?". Geotimes. 47 (1): 14–17. Archived from the original on 5 April 2018. Retrieved 5 April 2018.
- .
- doi:10.1039/b809492f.
- .
- S2CID 4412623.
- ^ PMID 33139706.
- ^ Sarmiento, J.L.; Gruber, N. (2006). Ocean Biogeochemical Dynamics. Princeton University Press, Princeton, New Jersey, US.
- PMID 10102806.
- S2CID 128987509.
- ^ a b "The Slow Carbon Cycle". NASA. 16 June 2011. Archived from the original on 16 June 2012. Retrieved 24 June 2012.
- ^ a b The Carbon Cycle and Earth's Climate Archived 23 June 2003 at the Wayback Machine Information sheet for Columbia University Summer Session 2012 Earth and Environmental Sciences Introduction to Earth Sciences I
- ^ Berner, Robert A. (November 1999). "A New Look at the Long-term Carbon Cycle" (PDF). GSA Today. 9 (11): 1–6. Archived (PDF) from the original on 13 February 2019.
- ^ S2CID 210305910.
- ^ NASA Earth Observatory (16 June 2011). "The Fast Carbon Cycle". Archive.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- PMID 11904360.
- doi:10.3390/sci1010017.).
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License "Archived copy". Archived from the original on 16 October 2017. Retrieved 5 July 2020.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License "Archived copy". Archived from the original on 16 October 2017. Retrieved 5 July 2020.{{cite web}}: CS1 maint: archived copy as title (link) CS1 maint: bot: original URL status unknown (link - ISSN 0273-0979.
- ^ NASA Earth Observatory (16 June 2011). "The Slow Carbon Cycle". Archive.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ doi:10.3389/fmars.2017.00007..
 Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - .
- .
- S2CID 129377411.
- S2CID 41290209.
- .
- PMID 27462728.
- PMID 9657713.
- S2CID 5019983.
- PMID 18954979.
- S2CID 94252768.
- S2CID 3461265.
- S2CID 205246638.
- .
- .
- S2CID 140536437.
- ^ Linsley, Ray K. (1975). "Solutions Manual to Accompany Hydrology for Engineers".
- .
- S2CID 4345881.
- S2CID 1728636.
- ^ S2CID 4460910.
- doi:10.1038/ngeo1817.
- .
- .
- .
- .
- S2CID 8889134.
- ^ PMID 21329201.
- ISBN 9781461591467.
- .
- .
- S2CID 205079291.
- S2CID 10255391.
- ^ S2CID 251168582..
 Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - S2CID 128411658.
- .
- S2CID 53512466.
- S2CID 53418968.
- ^ S2CID 4399374.
- PMID 21329201.
- S2CID 205243485.
- ^ Sigman DM & GH Haug. 2006. The biological pump in the past. In: Treatise on Geochemistry; vol. 6, (ed.). Pergamon Press, pp. 491–528
- .
- S2CID 16787695.
- .
- ISSN 0967-0637.
- ^ doi:10.5670/oceanog.2001.06..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - ^ De La Rocha C.L. (2006) "The Biological Pump". In: Treatise on Geochemistry; vol. 6, Pergamon Press, pp. 83–111.
- S2CID 204027259.
- ^ "The Deep Carbon Cycle and our Habitable Planet | Deep Carbon Observatory". deepcarbon.net. Archived from the original on 27 July 2020. Retrieved 19 February 2019.
- .
- ^ Dasgupta, Rajdeep (10 December 2011). "From Magma Ocean to Crustal Recycling: Earth's Deep Carbon Cycle". Archived from the original on 24 April 2016. Retrieved 9 March 2019.
- ^ "Carbon cycle reaches Earth's lower mantle: Evidence of carbon cycle found in 'superdeep' diamonds From Brazil". ScienceDaily. Retrieved 6 February 2019.
- S2CID 129243867.
- ^ PMID 21402927.
- (PDF) from the original on 18 July 2021.
- S2CID 39125834.
- ISBN 9780128113011. Retrieved 7 February 2019.
- S2CID 205335268.
- S2CID 49365059.
- ISSN 0012-821X.
- .
- ^ "Does Earth's Core Host a Deep Carbon Reservoir? | Deep Carbon Observatory". deepcarbon.net. Archived from the original on 27 July 2020. Retrieved 9 March 2019.
- PMID 25453077.
- ISSN 1752-0908.
- ^ "Overview of greenhouse gases". U.S. Environmental Protection Agency. 23 December 2015. Retrieved 2 November 2020.
- ^ "The known unknowns of plastic pollution". The Economist. 3 March 2018. Retrieved 17 June 2018.
- ^ hdl:1885/163968..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - .
- (PDF) from the original on 26 April 2019.
- hdl:10871/26418.
- ISBN 9781107415324.
- .
- ^ Hausfather, Zeke; Betts, Richard (14 April 2020). "Analysis: How 'carbon-cycle feedbacks' could make global warming worse". Carbon Brief. Archived from the original on 16 April 2020. Retrieved 4 January 2022.
- ^ doi:10.5194/essd-11-1783-2019..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - ^ IPCC (2007) 7.4.5 Minerals Archived 25 May 2016 at the Wayback Machine in Climate Change 2007: Working Group III: Mitigation of Climate Change,
- ^ Buis, Alan; Ramsayer, Kate; Rasmussen, Carol (12 November 2015). "A Breathing Planet, Off Balance". NASA. Archived from the original on 14 November 2015. Retrieved 13 November 2015.
- ^ "Audio (66:01) - NASA News Conference - Carbon & Climate Telecon". NASA. 12 November 2015. Archived from the original on 17 November 2015. Retrieved 12 November 2015.
- ^ St. Fleur, Nicholas (10 November 2015). "Atmospheric Greenhouse Gas Levels Hit Record, Report Says". The New York Times. Archived from the original on 11 November 2015. Retrieved 11 November 2015.
- AP News. Archivedfrom the original on 17 November 2015. Retrieved 11 November 2015.
- ^ ISBN 9780444873910.
- ^ "Figure 8.SM.4" (PDF). Intergovernmental Panel on Climate Change Fifth Assessment Report. p. 8SM-16. Archived (PDF) from the original on 13 March 2019.
- hdl:2268/12933.
- .
- NOAAGlobal Monitoring Laboratory/Earth System Research Laboratories.
- ^ Sciance, Fred (29 October 2013). "The Transition from HFC- 134a to a Low -GWP Refrigerant in Mobile Air Conditioners HFO -1234yf" (PDF). General Motors Public Policy Center. Archived (PDF) from the original on 15 October 2015. Retrieved 1 August 2018.
External links
- Carbon Cycle Science Program – an interagency partnership.
- NOAA's Carbon Cycle Greenhouse Gases Group
- Global Carbon Project – initiative of the Earth System Science Partnership
- UNEP – The present carbon cycle – Climate Change Archived 15 September 2008 at the Wayback Machine carbon levels and flows
- NASA's Orbiting Carbon Observatory Archived 9 September 2018 at the Wayback Machine