Carbon dioxide

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Carbon dioxide
| |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 3DMet | |||
| 1900390 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.004.271 | ||
| EC Number |
| ||
| E number | E290 (preservatives) | ||
| 989 | |||
| KEGG | |||
| MeSH | Carbon+dioxide | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1013 (gas), 1845 (solid) | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CO2 | |||
| Molar mass | 44.009 g·mol−1 | ||
| Appearance | Colorless gas | ||
| Odor |
| ||
| Density |
| ||
| Critical point (T, P) | 304.128(15) K[2] (30.978(15) °C), 7.3773(30) MPa[2] (72.808(30) atm) | ||
| 194.6855(30) K (−78.4645(30) °C) at 1 atm (0.101325 MPa) | |||
| 1.45 g/L at 25 °C (77 °F), 100 kPa (0.99 atm) | |||
| Vapor pressure | 5.7292(30) MPa, 56.54(30) atm (20 °C (293.15 K)) | ||
| Acidity (pKa) | Carbonic acid: pKa1 = 3.6 pKa1(apparent) = 6.35 pKa2 = 10.33 | ||
| −20.5·10−6 cm3/mol | |||
Thermal conductivity
|
0.01662 W·m−1·K−1 (300 K (27 °C; 80 °F))[3] | ||
Refractive index (nD)
|
1.00045 | ||
| Viscosity |
| ||
| 0 D | |||
| Structure | |||
| Trigonal | |||
Linear
| |||
| Thermochemistry | |||
Heat capacity (C)
|
37.135 J/(K·mol) | ||
Std molar
entropy (S⦵298) |
214 J·mol−1·K−1 | ||
Std enthalpy of (ΔfH⦵298)formation |
−393.5 kJ·mol−1 | ||
| Pharmacology | |||
| V03AN02 (WHO) | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
90,000 ppm (162,000 mg/m3) (human, 5 min)[6] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 5000 ppm (9000 mg/m3)[5] | ||
REL (Recommended)
|
TWA 5000 ppm (9000 mg/m3), ST 30,000 ppm (54,000 mg/m3)[5] | ||
IDLH (Immediate danger) |
40,000 ppm (72,000 mg/m3)[5] | ||
| Safety data sheet (SDS) | Sigma-Aldrich | ||
| Related compounds | |||
Other anions
|
|||
Other cations
|
| ||
| See Oxocarbon | |||
Related compounds
|
|||
| Supplementary data page | |||
| Carbon dioxide (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Carbon dioxide is a chemical compound with the chemical formula CO2. It is made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature, and as the source of available carbon in the carbon cycle, atmospheric CO2 is the primary carbon source for life on Earth. In the air, carbon dioxide is transparent to visible light but absorbs infrared radiation, acting as a greenhouse gas. Carbon dioxide is soluble in water and is found in groundwater, lakes, ice caps, and seawater. When carbon dioxide dissolves in water, it forms carbonate and mainly bicarbonate (HCO−3), which causes ocean acidification as atmospheric CO2 levels increase.[9]
It is a
Its concentration in Earth's pre-industrial atmosphere since late in the Precambrian was regulated by organisms and geological phenomena. Plants, algae and cyanobacteria use energy from sunlight to synthesize carbohydrates from carbon dioxide and water in a process called photosynthesis, which produces oxygen as a waste product.[14] In turn, oxygen is consumed and CO2 is released as waste by all aerobic organisms when they metabolize organic compounds to produce energy by respiration.[15] CO2 is released from organic materials when they decay or combust, such as in forest fires. Since plants require CO2 for photosynthesis, and humans and animals depend on plants for food, CO2 is necessary for the survival of life on earth.
Carbon dioxide is 53% more dense than dry air, but is long lived and thoroughly mixes in the atmosphere. About half of excess CO2 emissions to the atmosphere are absorbed by
CO2 is a versatile industrial material, used, for example, as an inert gas in welding and
Chemical and physical properties
Structure, bonding and molecular vibrations
The

As a linear triatomic molecule, CO2 has four vibrational modes as shown in the diagram. In the symmetric and the antisymmetric stretching modes, the atoms move along the axis of the molecule. There are two bending modes, which are degenerate, meaning that they have the same frequency and same energy, because of the symmetry of the molecule. When a molecule touches a surface or touches another molecule, the two bending modes can differ in frequency because the interaction is different for the two modes. Some of the vibrational modes are observed in the infrared (IR) spectrum: the antisymmetric stretching mode at wavenumber 2349 cm−1 (wavelength 4.25 μm) and the degenerate pair of bending modes at 667 cm−1 (wavelength 15 μm). The symmetric stretching mode does not create an electric dipole so is not observed in IR spectroscopy, but it is detected in Raman spectroscopy at 1388 cm−1 (wavelength 7.2 μm).[20]
In the gas phase, carbon dioxide molecules undergo significant vibrational motions and do not keep a fixed structure. However, in a Coulomb explosion imaging experiment, an instantaneous image of the molecular structure can be deduced. Such an experiment[21] has been performed for carbon dioxide. The result of this experiment, and the conclusion of theoretical calculations[22] based on an ab initio potential energy surface of the molecule, is that none of the molecules in the gas phase are ever exactly linear. This counter-intuitive result is trivially due to the fact that the nuclear motion volume element vanishes for linear geometries.[22] This is so for all molecules except diatomic molecules.
In aqueous solution
Carbon dioxide is
- CO2 + H2O ⇌ H2CO3
The hydration equilibrium constant of carbonic acid is, at 25 °C:
Hence, the majority of the carbon dioxide is not converted into carbonic acid, but remains as CO2 molecules, not affecting the pH.
The relative concentrations of CO2, H2CO3, and the deprotonated forms HCO−3 (bicarbonate) and CO2−3(carbonate) depend on the pH. As shown in a Bjerrum plot, in neutral or slightly alkaline water (pH > 6.5), the bicarbonate form predominates (>50%) becoming the most prevalent (>95%) at the pH of seawater. In very alkaline water (pH > 10.4), the predominant (>50%) form is carbonate. The oceans, being mildly alkaline with typical pH = 8.2–8.5, contain about 120 mg of bicarbonate per liter.
Being
- H2CO3 ⇌ HCO−3 + H+
- Ka1 = 2.5 × 10−4 mol/L; pKa1 = 3.6 at 25 °C.[19]
This is the true first acid dissociation constant, defined as
where the denominator includes only covalently bound H2CO3 and does not include hydrated CO2(aq). The much smaller and often-quoted value near 4.16 × 10−7 (or pKa1 = 6.38) is an apparent value calculated on the (incorrect) assumption that all dissolved CO2 is present as carbonic acid, so that
Since most of the dissolved CO2 remains as CO2 molecules, Ka1(apparent) has a much larger denominator and a much smaller value than the true Ka1.[23]
The bicarbonate ion is an
- HCO−3 ⇌ CO2−3 + H+
- Ka2 = 4.69 × 10−11 mol/L; pKa2 = 10.329
In organisms, carbonic acid production is catalysed by the enzyme known as carbonic anhydrase.
In addition to altering its acidity, the presence of carbon dioxide in water also affects its electrical properties.
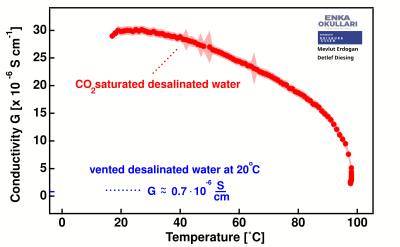
When carbon dioxide dissolves in desalinated water, the electrical conductivity increases significantly from below 1 μS/cm to nearly 30 μS/cm. When heated, the water begins to gradually lose the conductivity induced by the presence of , especially noticeable as temperatures exceed 30°C.
The temperature dependence of the electrical conductivity of fully deionized water without CO2 saturation is comparably low in relation to these data.
Chemical reactions of CO2
CO2 is a potent
- MR + CO2 → RCO2M
- where M = aryl.
In metal carbon dioxide complexes, CO2 serves as a ligand, which can facilitate the conversion of CO2 to other chemicals.[25]
The reduction of CO2 to CO is ordinarily a difficult and slow reaction:
- CO2 + 2 e− + 2 H+ → CO + H2O
The
- n CO2 + n H2O → (CH2O)n + n O2
Physical properties

Carbon dioxide is colorless. At low concentrations, the gas is odorless; however, at sufficiently high concentrations, it has a sharp, acidic odor.
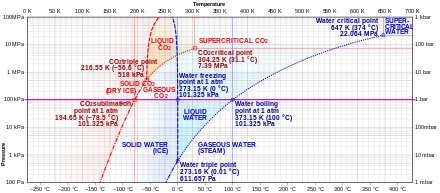
Carbon dioxide has no liquid state at pressures below 0.51795(10)

At temperatures and pressures above the critical point, carbon dioxide behaves as a supercritical fluid known as supercritical carbon dioxide.
Table of thermal and physical properties of saturated liquid carbon dioxide:[29][30]
| Temperature (°C) | Density (kg/m3) | Specific heat (kJ/(kg⋅K))
|
Kinematic viscosity (m2/s)
|
Thermal conductivity (W/(m⋅K))
|
Thermal diffusivity (m2/s) | Prandtl Number
|
Bulk modulus (K^-1)[clarification needed] |
| −50 | 1156.34 | 1.84 | 1.19 × 10−7 | 0.0855 | 4.02 × 10−8 | 2.96 | |
| −40 | 1117.77 | 1.88 | 1.18 × 10−7 | 0.1011 | 4.81 × 10−8 | 2.46 | |
| −30 | 1076.76 | 1.97 | 1.17 × 10−7 | 0.1116 | 5.27 × 10−8 | 2.22 | |
| −20 | 1032.39 | 2.05 | 1.15 × 10−7 | 0.1151 | 5.45 × 10−8 | 2.12 | |
| −10 | 983.38 | 2.18 | 1.13 × 10−7 | 0.1099 | 5.13 × 10−8 | 2.2 | |
| 0 | 926.99 | 2.47 | 1.08 × 10−7 | 0.1045 | 4.58 × 10−8 | 2.38 | |
| 10 | 860.03 | 3.14 | 1.01 × 10−7 | 0.0971 | 3.61 × 10−8 | 2.8 | |
| 20 | 772.57 | 5 | 9.10 × 10−8 | 0.0872 | 2.22 × 10−8 | 4.1 | 1.40 × 10−2[clarification needed] |
| 30 | 597.81 | 36.4 | 8.00 × 10−8 | 0.0703 | 0.279 × 10−8 | 28.7 |
Table of thermal and physical properties of carbon dioxide (CO2) at atmospheric pressure:[29][30]
| Temperature (K) | Density (kg/m3) | Specific heat (kJ/(kg⋅°C)) | Dynamic viscosity (kg/(m⋅s))
|
Kinematic viscosity (m2/s) | Thermal conductivity (W/(m⋅°C)) | Thermal diffusivity (m2/s) | Prandtl Number |
| 220 | 2.4733 | 0.783 | 1.11 × 10−5 | 4.49 × 10−6 | 0.010805 | 5.92 × 10−6 | 0.818 |
| 250 | 2.1657 | 0.804 | 1.26 × 10−5 | 5.81 × 10−6 | 0.012884 | 7.40 × 10−6 | 0.793 |
| 300 | 1.7973 | 0.871 | 1.50 × 10−5 | 8.32 × 10−6 | 0.016572 | 1.06 × 10−5 | 0.77 |
| 350 | 1.5362 | 0.9 | 1.72 × 10−5 | 1.12 × 10−5 | 0.02047 | 1.48 × 10−5 | 0.755 |
| 400 | 1.3424 | 0.942 | 1.93 × 10−5 | 1.44 × 10−5 | 0.02461 | 1.95 × 10−5 | 0.738 |
| 450 | 1.1918 | 0.98 | 2.13 × 10−5 | 1.79 × 10−5 | 0.02897 | 2.48 × 10−5 | 0.721 |
| 500 | 1.0732 | 1.013 | 2.33 × 10−5 | 2.17 × 10−5 | 0.03352 | 3.08 × 10−5 | 0.702 |
| 550 | 0.9739 | 1.047 | 2.51 × 10−5 | 2.57 × 10−5 | 0.03821 | 3.75 × 10−5 | 0.685 |
| 600 | 0.8938 | 1.076 | 2.68 × 10−5 | 3.00 × 10−5 | 0.04311 | 4.48 × 10−5 | 0.668 |
| 650 | 0.8143 | 1.1 | 2.88 × 10−5 | 3.54 × 10−5 | 0.0445 | 4.97 × 10−5 | 0.712 |
| 700 | 0.7564 | 1.13 | 3.05 × 10−5 | 4.03 × 10−5 | 0.0481 | 5.63 × 10−5 | 0.717 |
| 750 | 0.7057 | 1.15 | 3.21 × 10−5 | 4.55 × 10−5 | 0.0517 | 6.37 × 10−5 | 0.714 |
| 800 | 0.6614 | 1.17 | 3.37 × 10−5 | 5.10 × 10−5 | 0.0551 | 7.12 × 10−5 | 0.716 |
Biological role
Carbon dioxide is an end product of
Photosynthesis and carbon fixation
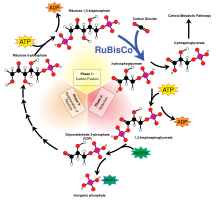
RuBisCO is thought to be the single most abundant protein on Earth.[31]
Phototrophs use the products of their photosynthesis as internal food sources and as raw material for the biosynthesis of more complex organic molecules, such as polysaccharides, nucleic acids, and proteins. These are used for their own growth, and also as the basis of the food chains and webs that feed other organisms, including animals such as ourselves. Some important phototrophs, the coccolithophores synthesise hard calcium carbonate scales.[32] A globally significant species of coccolithophore is Emiliania huxleyi whose calcite scales have formed the basis of many sedimentary rocks such as limestone, where what was previously atmospheric carbon can remain fixed for geological timescales.
Plants can grow as much as 50% faster in concentrations of 1,000 ppm CO2 when compared with ambient conditions, though this assumes no change in climate and no limitation on other nutrients.[33] Elevated CO2 levels cause increased growth reflected in the harvestable yield of crops, with wheat, rice and soybean all showing increases in yield of 12–14% under elevated CO2 in FACE experiments.[34][35]
Increased atmospheric CO2 concentrations result in fewer stomata developing on plants
The concentration of secondary
Plants also emit CO2 during respiration, and so the majority of plants and algae, which use
Toxicity
Carbon dioxide content in fresh air (averaged between sea-level and 10 kPa level, i.e., about 30 km (19 mi) altitude) varies between 0.036% (360 ppm) and 0.041% (412 ppm), depending on the location.[46]
CO2 is an asphyxiant gas and not classified as toxic or harmful in accordance with Globally Harmonized System of Classification and Labelling of Chemicals standards of United Nations Economic Commission for Europe by using the OECD Guidelines for the Testing of Chemicals. In concentrations up to 1% (10,000 ppm), it will make some people feel drowsy and give the lungs a stuffy feeling.[45] Concentrations of 7% to 10% (70,000 to 100,000 ppm) may cause suffocation, even in the presence of sufficient oxygen, manifesting as dizziness, headache, visual and hearing dysfunction, and unconsciousness within a few minutes to an hour.[47] The physiological effects of acute carbon dioxide exposure are grouped together under the term hypercapnia, a subset of asphyxiation.
Because it is heavier than air, in locations where the gas seeps from the ground (due to sub-surface volcanic or geothermal activity) in relatively high concentrations, without the dispersing effects of wind, it can collect in sheltered/pocketed locations below average ground level, causing animals located therein to be suffocated. Carrion feeders attracted to the carcasses are then also killed. Children have been killed in the same way near the city of Goma by CO2 emissions from the nearby volcano Mount Nyiragongo.[48] The Swahili term for this phenomenon is mazuku.

Adaptation to increased concentrations of CO2 occurs in humans, including modified breathing and kidney bicarbonate production, in order to balance the effects of blood acidification (acidosis). Several studies suggested that 2.0 percent inspired concentrations could be used for closed air spaces (e.g. a submarine) since the adaptation is physiological and reversible, as deterioration in performance or in normal physical activity does not happen at this level of exposure for five days.[49][50] Yet, other studies show a decrease in cognitive function even at much lower levels.[51][52] Also, with ongoing respiratory acidosis, adaptation or compensatory mechanisms will be unable to reverse the condition.
Below 1%
There are few studies of the health effects of long-term continuous CO2 exposure on humans and animals at levels below 1%. Occupational CO2 exposure limits have been set in the United States at 0.5% (5000 ppm) for an eight-hour period.[53] At this CO2 concentration, International Space Station crew experienced headaches, lethargy, mental slowness, emotional irritation, and sleep disruption.[54] Studies in animals at 0.5% CO2 have demonstrated kidney calcification and bone loss after eight weeks of exposure.[55] A study of humans exposed in 2.5 hour sessions demonstrated significant negative effects on cognitive abilities at concentrations as low as 0.1% (1000 ppm) CO2 likely due to CO2 induced increases in cerebral blood flow.[51] Another study observed a decline in basic activity level and information usage at 1000 ppm, when compared to 500 ppm.[52]
However a review of the literature found that a reliable subset of studies on the phenomenon of carbon dioxide induced cognitive impairment to only show a small effect on high-level decision making (for concentrations below 5000 ppm). Most of the studies were confounded by inadequate study designs, environmental comfort, uncertainties in exposure doses and differing cognitive assessments used.[56] Similarly a study on the effects of the concentration of CO2 in motorcycle helmets has been criticized for having dubious methodology in not noting the self-reports of motorcycle riders and taking measurements using mannequins. Further when normal motorcycle conditions were achieved (such as highway or city speeds) or the visor was raised the concentration of CO2 declined to safe levels (0.2%).[57][58]
| Concentration | Note |
|---|---|
| 280 ppm | Pre-industrial levels |
| 421 ppm | Current (May 2022) levels |
| 700 ppm | ASHRAE recommendation[59] |
| 1000 ppm | Cognitive impairment, Canada's long term exposure limit[60] |
| 1000-2000 ppm | Drowsiness[61] |
| 2000-5000 ppm | Headaches, sleepiness; poor concentration, loss of attention, slight nausea also possible[61] |
| 5000 ppm | USA 8h exposure limit[53] |
Ventilation

Poor ventilation is one of the main causes of excessive CO2 concentrations in closed spaces, leading to poor indoor air quality. Carbon dioxide differential above outdoor concentrations at steady state conditions (when the occupancy and ventilation system operation are sufficiently long that CO2 concentration has stabilized) are sometimes used to estimate ventilation rates per person.[citation needed] Higher CO2 concentrations are associated with occupant health, comfort and performance degradation.[62][63] ASHRAE Standard 62.1–2007 ventilation rates may result in indoor concentrations up to 2,100 ppm above ambient outdoor conditions. Thus if the outdoor concentration is 400 ppm, indoor concentrations may reach 2,500 ppm with ventilation rates that meet this industry consensus standard. Concentrations in poorly ventilated spaces can be found even higher than this (range of 3,000 or 4,000 ppm).
Miners, who are particularly vulnerable to gas exposure due to insufficient ventilation, referred to mixtures of carbon dioxide and nitrogen as "
In February 2020, three people died from suffocation at a party in Moscow when dry ice (frozen CO2) was added to a swimming pool to cool it down.[64] A similar accident occurred in 2018 when a woman died from CO2 fumes emanating from the large amount of dry ice she was transporting in her car.[65]
Indoor air
Humans spend more and more time in a confined atmosphere (around 80-90% of the time in a building or vehicle). According to the French Agency for Food, Environmental and Occupational Health & Safety (ANSES) and various actors in France, the CO2 rate in the indoor air of buildings (linked to human or animal occupancy and the presence of combustion installations), weighted by air renewal, is “usually between about 350 and 2,500 ppm”.[66]
In homes, schools, nurseries and offices, there are no systematic relationships between the levels of CO2 and other pollutants, and indoor CO2 is statistically not a good predictor of pollutants linked to outdoor road (or air, etc.) traffic.[67] CO2 is the parameter that changes the fastest (with hygrometry and oxygen levels when humans or animals are gathered in a closed or poorly ventilated room). In poor countries, many open hearths are sources of CO2 and CO emitted directly into the living environment.[68]
Outdoor areas with elevated concentrations
Local concentrations of carbon dioxide can reach high values near strong sources, especially those that are isolated by surrounding terrain. At the Bossoleto hot spring near Rapolano Terme in Tuscany, Italy, situated in a bowl-shaped depression about 100 m (330 ft) in diameter, concentrations of CO2 rise to above 75% overnight, sufficient to kill insects and small animals. After sunrise the gas is dispersed by convection.[69] High concentrations of CO2 produced by disturbance of deep lake water saturated with CO2 are thought to have caused 37 fatalities at Lake Monoun, Cameroon in 1984 and 1700 casualties at Lake Nyos, Cameroon in 1986.[70]
Human physiology
Content
| Blood compartment | ( kPa )
|
( mm Hg )
|
|---|---|---|
| Venous blood carbon dioxide | 5.5–6.8 | 41–51[71] |
| Alveolar pulmonary gas pressures |
4.8 | 36 |
Arterial blood carbon dioxide
|
4.7–6.0 | 35–45[71] |
The body produces approximately 2.3 pounds (1.0 kg) of carbon dioxide per day per person,
Transport in the blood
CO2 is carried in blood in three different ways. Exact percentages vary between arterial and venous blood.
- Majority (about 70% to 80%) is converted to bicarbonate ions HCO−3 by the enzyme carbonic anhydrase in the red blood cells,[74] by the reaction:
- CO2 + H2O → H2CO3 → H+ + HCO−3
- 5–10% is dissolved in blood plasma[74]
- 5–10% is bound to hemoglobin as carbamino compounds[74]
Regulation of respiration
Carbon dioxide is one of the mediators of local
Bicarbonate ions are crucial for regulating blood pH. A person's breathing rate influences the level of CO2 in their blood. Breathing that is too slow or shallow causes respiratory acidosis, while breathing that is too rapid leads to hyperventilation, which can cause respiratory alkalosis.[76]
Although the body requires oxygen for metabolism, low oxygen levels normally do not stimulate breathing. Rather, breathing is stimulated by higher carbon dioxide levels. As a result, breathing low-pressure air or a gas mixture with no oxygen at all (such as pure nitrogen) can lead to loss of consciousness without ever experiencing
The respiratory centers try to maintain an arterial CO2 pressure of 40
Concentrations and role in the environment
Atmosphere

In
Carbon dioxide is a greenhouse gas. It absorbs and emits
The increase in atmospheric concentrations of CO2 and other long-lived greenhouse gases such as methane increase the absorption and emission of infrared radiation by the atmosphere. This has led to a rise in average global temperature and ocean acidification. Another direct effect is the CO2 fertilization effect. The increase in atmospheric concentrations of CO2 causes a range of further effects of climate change on the environment and human living conditions.
The present atmospheric concentration of CO2 is the highest for 14 million years.[86] Concentrations of CO2 in the atmosphere were as high as 4,000 ppm during the Cambrian period about 500 million years ago, and as low as 180 ppm during the Quaternary glaciation of the last two million years.[79] Reconstructed temperature records for the last 420 million years indicate that atmospheric CO2 concentrations peaked at approximately 2,000 ppm. This peak happened during the Devonian period (400 million years ago). Another peak occurred in the Triassic period (220–200 million years ago).[87]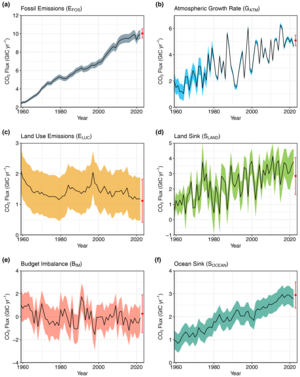
Oceans
Ocean acidification
Carbon dioxide dissolves in the ocean to form carbonic acid (H2CO3), bicarbonate (HCO−3), and carbonate (CO2−3). There is about fifty times as much carbon dioxide dissolved in the oceans as exists in the atmosphere. The oceans act as an enormous carbon sink, and have taken up about a third of CO2 emitted by human activity.[89]
Changes in ocean chemistry can have extensive direct and indirect effects on organisms and their habitats. One of the most important repercussions of increasing ocean acidity relates to the production of shells out of
Very little of the extra carbon dioxide that is added into the ocean remains as dissolved carbon dioxide. The majority dissociates into additional bicarbonate and free hydrogen ions. The increase in hydrogen is larger than the increase in bicarbonate,[95] creating an imbalance in the reaction:
- HCO−3 ⇌ CO2−3 + H+
To maintain chemical equilibrium, some of the carbonate ions already in the ocean combine with some of the hydrogen ions to make further bicarbonate. Thus the ocean's concentration of carbonate ions is reduced, removing an essential building block for marine organisms to build shells, or calcify:
- Ca2+ + CO2−3 ⇌ CaCO3
Hydrothermal vents
Carbon dioxide is also introduced into the oceans through hydrothermal vents. The Champagne hydrothermal vent, found at the Northwest Eifuku volcano in the Mariana Trench, produces almost pure liquid carbon dioxide, one of only two known sites in the world as of 2004, the other being in the Okinawa Trough.[96] The finding of a submarine lake of liquid carbon dioxide in the Okinawa Trough was reported in 2006.[97]
Production
Biological processes
Carbon dioxide is a by-product of the
, also known as alcohol, as follows:- C6H12O6 → 2 CO2 + 2 CH3CH2OH
All aerobic organisms produce CO2 when they oxidize carbohydrates, fatty acids, and proteins. The large number of reactions involved are exceedingly complex and not described easily. Refer to cellular respiration, anaerobic respiration and photosynthesis. The equation for the respiration of glucose and other monosaccharides is:
- C6H12O6 + 6 O2 → 6 CO2 + 6 H2O
Industrial processes
Carbon dioxide can be obtained by distillation from air, but the method is inefficient. Industrially, carbon dioxide is predominantly an unrecovered waste product, produced by several methods which may be practiced at various scales.[100]
Combustion
The combustion of all carbon-based fuels, such as methane (natural gas), petroleum distillates (gasoline, diesel, kerosene, propane), coal, wood and generic organic matter produces carbon dioxide and, except in the case of pure carbon, water. As an example, the chemical reaction between methane and oxygen:
- CH4 + 2 O2 → CO2 + 2 H2O
Iron is reduced from its oxides with coke in a blast furnace, producing pig iron and carbon dioxide:[101]
- Fe2O3 + 3 CO → 3 CO2 + 2 Fe
By-product from hydrogen production
Carbon dioxide is a byproduct of the industrial production of hydrogen by
Thermal decomposition of limestone
It is produced by thermal decomposition of limestone, CaCO3 by heating (
- CaCO3 → CaO + CO2
Acids liberate CO2 from most metal carbonates. Consequently, it may be obtained directly from natural carbon dioxide
- CaCO3 + 2 HCl → CaCl2 + H2CO3
The carbonic acid (H2CO3) then decomposes to water and CO2:
- H2CO3 → CO2 + H2O
Such reactions are accompanied by foaming or bubbling, or both, as the gas is released. They have widespread uses in industry because they can be used to neutralize waste acid streams.
Commercial uses
Carbon dioxide is used by the food industry, the oil industry, and the chemical industry.[100] The compound has varied commercial uses but one of its greatest uses as a chemical is in the production of carbonated beverages; it provides the sparkle in carbonated beverages such as soda water, beer and sparkling wine.
Precursor to chemicals
This section needs expansion. You can help by adding to it. (July 2014) |
In the chemical industry, carbon dioxide is mainly consumed as an ingredient in the production of urea, with a smaller fraction being used to produce methanol and a range of other products.[104] Some carboxylic acid derivatives such as sodium salicylate are prepared using CO2 by the Kolbe–Schmitt reaction.[105]
In addition to conventional processes using CO2 for chemical production, electrochemical methods are also being explored at a research level. In particular, the use of renewable energy for production of fuels from CO2 (such as methanol) is attractive as this could result in fuels that could be easily transported and used within conventional combustion technologies but have no net CO2 emissions.[106]
Agriculture
Plants require carbon dioxide to conduct photosynthesis. The atmospheres of greenhouses may (if of large size, must) be enriched with additional CO2 to sustain and increase the rate of plant growth.
Foods

Carbon dioxide is a
A candy called Pop Rocks is pressurized with carbon dioxide gas[114] at about 4,000 kPa (40 bar; 580 psi). When placed in the mouth, it dissolves (just like other hard candy) and releases the gas bubbles with an audible pop.
Beverages
Carbon dioxide is used to produce
The taste of soda water (and related taste sensations in other carbonated beverages) is an effect of the dissolved carbon dioxide rather than the bursting bubbles of the gas.
Winemaking

Carbon dioxide in the form of
Carbon dioxide is sometimes used to top up wine bottles or other
Stunning animals
Carbon dioxide is often used to "stun" animals before slaughter.[117] "Stunning" may be a misnomer, as the animals are not knocked out immediately and may suffer distress.[118][119]
Inert gas
Carbon dioxide is one of the most commonly used compressed gases for pneumatic (pressurized gas) systems in portable pressure tools. Carbon dioxide is also used as an atmosphere for
Carbon dioxide is used in many consumer products that require pressurized gas because it is inexpensive and nonflammable, and because it undergoes a phase transition from gas to liquid at room temperature at an attainable pressure of approximately 60
.Fire extinguisher

Carbon dioxide can be used to extinguish flames by flooding the environment around the flame with the gas. It does not itself react to extinguish the flame, but starves the flame of oxygen by displacing it. Some
Carbon dioxide has also been widely used as an extinguishing agent in fixed fire-protection systems for local application of specific hazards and total flooding of a protected space.[122] International Maritime Organization standards recognize carbon dioxide systems for fire protection of ship holds and engine rooms. Carbon dioxide-based fire-protection systems have been linked to several deaths, because it can cause suffocation in sufficiently high concentrations. A review of CO2 systems identified 51 incidents between 1975 and the date of the report (2000), causing 72 deaths and 145 injuries.[123]
Supercritical CO2 as solvent
Liquid carbon dioxide is a good
Medical and pharmacological uses
In medicine, up to 5% carbon dioxide (130 times atmospheric concentration) is added to oxygen for stimulation of breathing after apnea and to stabilize the O2/CO2 balance in blood.
Carbon dioxide can be mixed with up to 50% oxygen, forming an inhalable gas; this is known as Carbogen and has a variety of medical and research uses.
Another medical use are the mofette, dry spas that use carbon dioxide from post-volcanic discharge for therapeutic purposes.
Energy
Supercritical CO2 is used as the working fluid in the Allam power cycle engine.
Fossil fuel recovery
Carbon dioxide is used in
In enhanced coal bed methane recovery, carbon dioxide would be pumped into the coal seam to displace methane, as opposed to current methods which primarily rely on the removal of water (to reduce pressure) to make the coal seam release its trapped methane.[126]
Bio transformation into fuel
It has been proposed that CO2 from power generation be bubbled into ponds to stimulate growth of
Researchers have developed an
Refrigerant
Liquid and solid carbon dioxide are important refrigerants, especially in the food industry, where they are employed during the transportation and storage of ice cream and other frozen foods. Solid carbon dioxide is called "dry ice" and is used for small shipments where refrigeration equipment is not practical. Solid carbon dioxide is always below −78.5 °C (−109.3 °F) at regular atmospheric pressure, regardless of the air temperature.
Liquid carbon dioxide (industry nomenclature R744 or R-744) was used as a refrigerant prior to the use of
Minor uses

Carbon dioxide is the lasing medium in a carbon-dioxide laser, which is one of the earliest type of lasers.
Carbon dioxide can be used as a means of controlling the pH of swimming pools,[135] by continuously adding gas to the water, thus keeping the pH from rising. Among the advantages of this is the avoidance of handling (more hazardous) acids. Similarly, it is also used in the maintaining reef aquaria, where it is commonly used in calcium reactors to temporarily lower the pH of water being passed over calcium carbonate in order to allow the calcium carbonate to dissolve into the water more freely, where it is used by some corals to build their skeleton.
Used as the primary coolant in the British
Carbon dioxide induction is commonly used for the euthanasia of laboratory research animals. Methods to administer CO2 include placing animals directly into a closed, prefilled chamber containing CO2, or exposure to a gradually increasing concentration of CO2. The American Veterinary Medical Association's 2020 guidelines for carbon dioxide induction state that a displacement rate of 30–70% of the chamber or cage volume per minute is optimal for the humane euthanasia of small rodents.[136]: 5, 31 Percentages of CO2 vary for different species, based on identified optimal percentages to minimize distress.[136]: 22
Carbon dioxide is also used in several related cleaning and surface-preparation techniques.
History of discovery

Carbon dioxide was the first gas to be described as a discrete substance. In about 1640,
The properties of carbon dioxide were further studied in the 1750s by the Scottish physician Joseph Black. He found that limestone (calcium carbonate) could be heated or treated with acids to yield a gas he called "fixed air". He observed that the fixed air was denser than air and supported neither flame nor animal life. Black also found that when bubbled through limewater (a saturated aqueous solution of calcium hydroxide), it would precipitate calcium carbonate. He used this phenomenon to illustrate that carbon dioxide is produced by animal respiration and microbial fermentation. In 1772, English chemist Joseph Priestley published a paper entitled Impregnating Water with Fixed Air in which he described a process of dripping sulfuric acid (or oil of vitriol as Priestley knew it) on chalk in order to produce carbon dioxide, and forcing the gas to dissolve by agitating a bowl of water in contact with the gas.[139]
Carbon dioxide was first liquefied (at elevated pressures) in 1823 by Humphry Davy and Michael Faraday.[140] The earliest description of solid carbon dioxide (dry ice) was given by the French inventor Adrien-Jean-Pierre Thilorier, who in 1835 opened a pressurized container of liquid carbon dioxide, only to find that the cooling produced by the rapid evaporation of the liquid yielded a "snow" of solid CO2.[141][142]
Carbon dioxide in combination with nitrogen was known from earlier times as
See also
- Arterial blood gas test – A test of blood taken from an artery that measures the amounts of certain dissolved gases
- Bosch reaction – forms elemental carbon from CO2 and hydrogen using a metallic catalyst
- Carbon dioxide removal – Removal of atmospheric carbon dioxide through human activity (from the atmosphere)
- List of least carbon efficient power stations
- List of countries by carbon dioxide emissions
- Meromictic lake – Permanently stratified lake with layers of water that do not intermix
- Gilbert Plass – Canadian physicist (1920–2004) (early work on CO2 and climate change)
- Sabatier reaction – Methanation process of carbon dioxide with hydrogen
- NASA's Orbiting Carbon Observatory 2 – NASA climate satellite
- Greenhouse Gases Observing Satellite – Earth observation satellite
- Soil gas – Gases in the air space between soil components
Notes
References
- ^ a b c "Carbon Dioxide" (PDF). Air Products. Archived from the original (PDF) on 29 July 2020. Retrieved 28 April 2017.
- ^ doi:10.1063/1.555991.
- ^ Touloukian YS, Liley PE, Saxena SC (1970). "Thermophysical properties of matter - the TPRC data series". Thermal Conductivity - Nonmetallic Liquids and Gases. 3. Data book.
- .
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0103". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Carbon dioxide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "Safety Data Sheet – Carbon Dioxide Gas – version 0.03 11/11" (PDF). AirGas.com. 12 February 2018. Archived (PDF) from the original on 4 August 2018. Retrieved 4 August 2018.
- ^ "Carbon dioxide, refrigerated liquid" (PDF). Praxair. p. 9. Archived from the original (PDF) on 29 July 2018. Retrieved 26 July 2018.
- ISBN 978-0-309-15359-1. Archivedfrom the original on 5 February 2016. Retrieved 29 February 2016.
- ^ "CO2 Gas Concentration Defined". CO2 Meter. 18 November 2022. Retrieved 5 September 2023.
- ISBN 9781107618763. Retrieved 9 November 2020.
- ^ "Carbon dioxide now more than 50% higher than pre-industrial levels | National Oceanic and Atmospheric Administration". www.noaa.gov. 3 June 2022. Retrieved 14 June 2022.
- ^ IPCC (2022) Summary for policy makers in Climate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, Cambridge, United Kingdom and New York, NY, US
- ISBN 978-0-7872-0460-0.
- ^ "Food Factories". www.legacyproject.org. Archived from the original on 12 August 2017. Retrieved 10 October 2011.
- IPCC (2021). "Summary for Policymakers" (PDF). Climate Change 2021: The Physical Science Basis. p. 20. Archived(PDF) from the original on 10 October 2022.
- ^ Myles, Allen (September 2020). "The Oxford Principles for Net Zero Aligned Carbon Offsetting" (PDF). Archived (PDF) from the original on 2 October 2020. Retrieved 10 December 2021.
- ^ ISBN 978-3-527-31558-1. Archivedfrom the original on 21 March 2020. Retrieved 3 December 2019.
- ^ ISBN 978-0-08-037941-8.
- ISBN 978-0-7167-8759-4.
- S2CID 250782825.
- ^ hdl:2142/107329.
- ISBN 978-0-07-032760-3.
- S2CID 216557447.
- ISBN 978-3-527-32475-0.
- (PDF) from the original on 19 April 2021. Retrieved 6 December 2019.
- ^ "Gases – Densities". Engineering Toolbox. Archived from the original on 2 March 2006. Retrieved 21 November 2020.
- S2CID 4363092.
- ^ ISBN 9780072406559.
- ^ ISBN 9780471457282.
- PMID 15067115.
(Rubisco) is the most prevalent enzyme on this planet, accounting for 30–50% of total soluble protein in the chloroplast
- OCLC 845654016.
- ^ Blom TJ, Straver WA, Ingratta FJ, Khosla S, Brown W (December 2002). "Carbon Dioxide In Greenhouses". Archived from the original on 29 April 2019. Retrieved 12 June 2007.
- S2CID 19200429. Archived from the original(PDF) on 19 July 2011.
- (PDF) from the original on 20 October 2016. Retrieved 27 October 2017.
- .
- S2CID 33415877.
- S2CID 16074723.
- S2CID 52262618.
- .
- PMID 15587703.
- ^ "Global Environment Division Greenhouse Gas Assessment Handbook – A Practical Guidance Document for the Assessment of Project-level Greenhouse Gas Emissions". World Bank. Archived from the original on 3 June 2016. Retrieved 10 November 2007.
- S2CID 4424430.
- S2CID 1779934.
- ^ a b Friedman D. "Toxicity of Carbon Dioxide Gas Exposure, CO2 Poisoning Symptoms, Carbon Dioxide Exposure Limits, and Links to Toxic Gas Testing Procedures". InspectAPedia. Archived from the original on 28 September 2009.
- ^ "CarbonTracker CT2011_oi (Graphical map of CO2)". esrl.noaa.gov. Archived from the original on 13 February 2021. Retrieved 20 April 2007.
- ^ "Carbon Dioxide as a Fire Suppressant: Examining the Risks". U.S. Environmental Protection Agency. Archived from the original on 2 October 2015.
- ^ "Volcano Under the City". A NOVA Production by Bonne Pioche and Greenspace for WGBH/Boston. Public Broadcasting System. 1 November 2005. Archived from the original on 5 April 2011..
- ^ Glatte Jr HA, Motsay GJ, Welch BE (1967). Carbon Dioxide Tolerance Studies (Report). Brooks AFB, TX School of Aerospace Medicine Technical Report. SAM-TR-67-77. Archived from the original on 9 May 2008. Retrieved 2 May 2008.
{{cite report}}: CS1 maint: unfit URL (link) - ^ Lambertsen CJ (1971). Carbon Dioxide Tolerance and Toxicity (Report). IFEM Report. Environmental Biomedical Stress Data Center, Institute for Environmental Medicine, University of Pennsylvania Medical Center. No. 2-71. Archived from the original on 24 July 2011. Retrieved 2 May 2008.
{{cite report}}: CS1 maint: unfit URL (link) - ^ PMID 23008272. Archived from the original(PDF) on 5 March 2016. Retrieved 11 December 2014.
- ^ PMID 26502459.
- ^ a b "Exposure Limits for Carbon Dioxide Gas – CO2 Limits". InspectAPedia.com. Archived from the original on 16 September 2018. Retrieved 19 October 2014.
- ^ Law J, Watkins S, Alexander D (2010). In-Flight Carbon Dioxide Exposures and Related Symptoms: Associations, Susceptibility and Operational Implications (PDF) (Report). NASA Technical Report. TP–2010–216126. Archived from the original (PDF) on 27 June 2011. Retrieved 26 August 2014.
- PMID 505623. Archived from the originalon 19 October 2014. Retrieved 19 October 2014.
- S2CID 219915861.
- ^ Kaplan L (4 June 2019). "Ask the doc: Does my helmet make me stupid? - RevZilla". www.revzilla.com. Archived from the original on 22 May 2021. Retrieved 22 May 2021.
- PMID 15893291.
- (PDF) from the original on 26 October 2022. Retrieved 10 August 2023.
- ^ Canada, Health (19 March 2021). "Carbon dioxide in your home". www.canada.ca. Retrieved 10 August 2023.
- ^ a b "Typical CO2 Levels at Home Tester". CO2 Meter. 8 August 2023. Retrieved 10 August 2023.
- PMID 26502459.
- ^ Romm J (26 October 2015). "Exclusive: Elevated CO2 Levels Directly Affect Human Cognition, New Harvard Study Shows". ThinkProgress. Archived from the original on 9 October 2019. Retrieved 14 October 2019.
- ^ "Three die in dry-ice incident at Moscow pool party". BBC News. 29 February 2020. Archived from the original on 29 February 2020.
The victims were connected to Instagram influencer Yekaterina Didenko.
- ^ Rettner R (2 August 2018). "A Woman Died from Dry Ice Fumes. Here's How It Can Happen". Live Science. Archived from the original on 22 May 2021. Retrieved 22 May 2021.
- ^ Concentrations de CO2 dans l'air intérieur et effets sur la santé (PDF) (Report) (in French). ANSES. July 2013. p. 294.
- S2CID 111182451.
- ^ Cetin, Mehmet; Sevik, Hakan (2016). "INDOOR QUALITY ANALYSIS OF CO2 FOR KASTAMONU UNIVERSITY" (PDF). Conference of the International Journal of Arts & Sciences. 9 (3): 71.
- ISBN 978-0-521-58203-2.
- ISBN 978-0-521-58203-2.
- ^ a b "ABG (Arterial Blood Gas)". Brookside Associates. Archived from the original on 12 August 2017. Retrieved 2 January 2017.
- ^ "How much carbon dioxide do humans contribute through breathing?". EPA.gov. Archived from the original on 2 February 2011. Retrieved 30 April 2009.
- ISBN 978-0-7645-7419-1.
- PMID 21521758.
- PMID 29494063. Retrieved 20 August 2022.
- PMID 9765173.
- ^ a b "Carbon dioxide now more than 50% higher than pre-industrial levels | National Oceanic and Atmospheric Administration". www.noaa.gov. 3 June 2022. Archived from the original on 5 June 2022. Retrieved 14 June 2022.
- ^ ISBN 9781107618763. Archivedfrom the original on 14 March 2023. Retrieved 14 March 2023.
- NOAA Global Monitoring Laboratory/Earth System Research Laboratories. Archivedfrom the original on 27 November 2020. Retrieved 18 December 2020.
- S2CID 19674607.
- ^ IPCC (2022) Summary for policy makers Archived 12 March 2023 at the Wayback Machine in Climate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 2 August 2022 at the Wayback Machine, Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
- .
- ISBN 978-0-7167-8759-4.
- ^ "Carbon Dioxide Absorbs and Re-emits Infrared Radiation". UCAR Center for Science Education. 2012. Archived from the original on 21 September 2017. Retrieved 9 September 2017.
- ^ AHMED, Issam. "Current carbon dioxide levels last seen 14 million years ago". phys.org. Retrieved 8 February 2024.
- ^ "Climate and CO2 in the Atmosphere". Archived from the original on 6 October 2018. Retrieved 10 October 2007.
- ..
- ^ Doney SC, Levine NM (29 November 2006). "How Long Can the Ocean Slow Global Warming?". Oceanus. Archived from the original on 4 January 2008. Retrieved 21 November 2007.
- S2CID 255431338.
Figure 1f
- ^ ISBN 0-85403-617-2.
- PMID 31819102.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - S2CID 214271838.
- S2CID 221369828.
- .
- Bibcode:2004AGUFM.V43F..08L.
- on 19 October 2018.
- ^ "Collecting and using biogas from landfills". U.S. Energy Information Administration. 11 January 2017. Archived from the original on 11 July 2018. Retrieved 22 November 2015.
- ^ "Facts About Landfill Gas" (PDF). U.S. Environmental Protection Agency. January 2000. Archived (PDF) from the original on 23 September 2015. Retrieved 4 September 2015.
- ^ ISBN 978-0-471-23896-6.
- ^
Strassburger J (1969). Blast Furnace Theory and Practice. New York: American Institute of Mining, Metallurgical, and Petroleum Engineers. ISBN 978-0-677-10420-1.
- ISBN 3527306730.
- ^ "CO2 shortage: Food industry calls for government action". BBC. 21 June 2018. Archived from the original on 23 May 2021. Retrieved 24 June 2018.
- ^ "IPCC Special Report on Carbon dioxide Capture and Storage" (PDF). The Intergovernmental Panel on Climate Change. Archived from the original (PDF) on 24 September 2015. Retrieved 4 September 2015.
- ISBN 978-0-205-05838-9.
- PMID 25309898.
- ^ Whiting D, Roll M, Vickerman L (August 2010). "Plant Growth Factors: Photosynthesis, Respiration, and Transpiration". CMG GardenNotes. Colorado Master Gardener Program. Archived from the original on 2 September 2014. Retrieved 10 October 2011.
- ^ Waggoner PE (February 1994). "Carbon dioxide". How Much Land Can Ten Billion People Spare for Nature?. Archived from the original on 12 October 2011. Retrieved 10 October 2011.
- S2CID 9845813.
- ISBN 978-3-319-46707-8, retrieved 10 April 2024
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". Archived from the original on 7 October 2010. Retrieved 27 October 2011.
- ^ US Food and Drug Administration: "Food Additive Status List". Food and Drug Administration. Archived from the original on 4 November 2017. Retrieved 13 June 2015.
- ^ Australia New Zealand Food Standards Code"Standard 1.2.4 – Labelling of ingredients". 8 September 2011. Archived from the original on 19 January 2012. Retrieved 27 October 2011.
- ISBN 978-0-9847670-1-4. Archivedfrom the original on 15 August 2021. Retrieved 9 November 2020.
- ]
- ^ "Scientists Discover Protein Receptor For Carbonation Taste". ScienceDaily. 16 October 2009. Archived from the original on 29 March 2020. Retrieved 29 March 2020.
- ^ Coghlan A (3 February 2018). "A more humane way of slaughtering chickens might get EU approval". New Scientist. Archived from the original on 24 June 2018. Retrieved 24 June 2018.
- ^ "What is CO2 stunning?". RSPCA. Archived from the original on 9 April 2014.
- ^ Campbell A (10 March 2018). "Humane execution and the fear of the tumbril". New Scientist. Archived from the original on 24 June 2018. Retrieved 24 June 2018.
- S2CID 32065173.
- ^ "Types of Fire Extinguishers". The Fire Safety Advice Centre. Archived from the original on 28 June 2021. Retrieved 28 June 2021.
- ^ National Fire Protection Association Code 12.
- ^ Carbon Dioxide as a Fire Suppressant: Examining the Risks, US EPA. 2000.
- ^ "Appendix A: CO2 for use in enhanced oil recovery (EOR)". Accelerating the uptake of CCS: industrial use of captured carbon dioxide. 20 December 2011. Archived from the original on 28 April 2017. Retrieved 2 January 2017.
{{cite book}}:|website=ignored (help) - ^ Austell JM (2005). "CO2 for Enhanced Oil Recovery Needs – Enhanced Fiscal Incentives". Exploration & Production: The Oil & Gas Review. Archived from the original on 7 February 2012. Retrieved 28 September 2007.
- ^ "Enhanced coal bed methane recovery". ETH Zurich. 31 August 2006. Archived from the original on 6 July 2011.
- ^ Clayton M (11 January 2006). "Algae – like a breath mint for smokestacks". The Christian Science Monitor. Archived from the original on 14 September 2008. Retrieved 11 October 2007.
- S2CID 1492698.
- S2CID 247160910.
- PMID 35058361.
- ^ "Clean Way To Turn CO2 Into Fuel Inspired by Nature". Applied Sciences from Technology Networks. 1 March 2022. Retrieved 2 March 2022.
- ^ Pearson, S. Forbes. "Refrigerants Past, Present and Future" (PDF). R744. Archived from the original (PDF) on 13 July 2018. Retrieved 30 March 2021.
- ^ "The Coca-Cola Company Announces Adoption of HFC-Free Insulation in Refrigeration Units to Combat Global Warming". The Coca-Cola Company. 5 June 2006. Archived from the original on 1 November 2013. Retrieved 11 October 2007.
- ^ "Modine reinforces its CO2 research efforts". R744.com. 28 June 2007. Archived from the original on 10 February 2008.
- ^ TCE, the Chemical Engineer. Institution of Chemical Engineers. 1990. Archived from the original on 17 August 2021. Retrieved 2 June 2020.
- ^ a b "AVMA guidelines for the euthanasia of animals: 2020 Edition" (PDF). American Veterinary Medical Association. 2020. Archived (PDF) from the original on 1 February 2014. Retrieved 13 August 2021.
- from the original on 17 March 2020. Retrieved 6 December 2019.
- ISBN 978-0-306-47277-0.
- from the original on 7 June 2010. Retrieved 11 October 2007.
- JSTOR 107649.
- ^ Thilorier AJ (1835). "Solidification de l'Acide carbonique". Comptes Rendus. 1: 194–196. Archived from the original on 2 September 2017. Retrieved 1 September 2017.
- from the original on 2 May 2016. Retrieved 15 November 2015.
- JSTOR 115391.



![{\displaystyle K_{\mathrm {h} }={\frac {{\ce {[H2CO3]}}}{{\ce {[CO2_{(aq)}]}}}}=1.70\times 10^{-3}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/7b01be634ec0c3d83cbf3aa2c71bbd51b9ce0e26)
![{\displaystyle K_{\mathrm {a1} }={\frac {{\ce {[HCO3- ][H+]}}}{{\ce {[H2CO3]}}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/f6013fac05dba06751345e4d824c525ed2c43b12)
![{\displaystyle K_{\mathrm {a1} }{\rm {(apparent)}}={\frac {{\ce {[HCO3- ][H+]}}}{{\ce {[H2CO3] + [CO2_{(aq)}]}}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/3ae2fa07440c037d6054ce4e7ef95c15c59264e7)
