Carbon hexoxide
| |
| Names | |
|---|---|
| IUPAC name
pentaoxan-6-one
| |
| Identifiers | |
3D model (
JSmol ) |
|
PubChem CID
|
|
| |
| |
| Properties | |
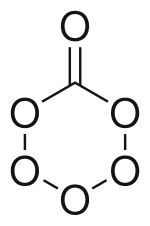
| CO6 | |
| Molar mass | 108.005 g·mol−1 |
| Related compounds | |
Related compounds
|
Carbon pentoxide Carbon tetroxide Carbon hexasulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Carbon hexoxide or carbon hexaoxide is an
Shape
The molecule that has been observed has a Cs symmetry. The ring is not a flat hexagon but puckered with slightly different side lengths and angles (120°) from the regular hexagon. Going around the ring starting at the carbon to oxygen bond the interatomic distances are C–O: 1.362 Å O–O 1.491 Å, O–O 1.391 Å, O–O 1.391 Å, O–O 1.491 Å, and O–C 1.362 Å. The angles between the bonds are: O–C–O 120.4 °, C–O–O 115.7°, O–O–O 105.9°, and the opposite from carbon O–O–O 104.1°. For the double carbon to oxygen bond, the length is 1.185 Å and the angle from the single bonds is 119.6°.[1]
Formation
In an experiment, carbon hexoxide was formed by irradiating solid carbon dioxide with electrons at an energy of 5000 eV at 10 K in a vacuum. The reaction proceeds by breaking
- O
The atomic oxygen then reacts with carbon dioxide to form
Properties
Carbon hexoxide is stable up to 60 K.[1] Vibrational infrared wavenumbers include the most prominent ν1 = 1876 cm−1 for the most common isotopologue 12C16O6.[1]
Other isomers
Other possible isomers of carbon hexoxide are the C2 form with a five and three membered ring, and the D2d with two four membered rings. The D2d O3CO3 isomer has a calculated C–O bond length of 1.391 Å, and an O–O length of 1.469 Å. The O–C–O bond angle is 94.1°. However these two isomers have not been observed.[2]
The equivalent carbon hexasulfide is also known from inert gas matrix study. It has C2 symmetry with the same atomic arrangement as the hexoxide.[3]
