Carbonate
 Carbonate anion
| |
anion
| |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Carbonate | |
| Systematic IUPAC name
Trioxidocarbonate[1]: 127 | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| CO2−3 | |
| Molar mass | 60.008 g·mol−1 |
Conjugate acid
|
Bicarbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
A carbonate is a salt of carbonic acid, H2CO3,[2] characterized by the presence of the carbonate ion, a polyatomic ion with the formula CO2−3. The word "carbonate" may also refer to a carbonate ester, an organic compound containing the carbonate group O=C(−O−)2.
The term is also used as a verb, to describe carbonation: the process of raising the concentrations of carbonate and bicarbonate ions in water to produce carbonated water and other carbonated beverages – either by the addition of carbon dioxide gas under pressure or by dissolving carbonate or bicarbonate salts into the water.
In
Structure and bonding
The carbonate ion is the simplest
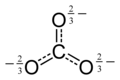
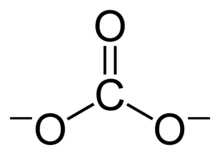
The Lewis structure of the carbonate ion has two (long) single bonds to negative oxygen atoms, and one short double bond to a neutral oxygen atom.
This structure is incompatible with the observed symmetry of the ion, which implies that the three bonds are the same length and that the three oxygen atoms are equivalent. As in the case of the
among three structures:This resonance can be summarized by a model with fractional bonds and
Chemical properties

Metal carbonates generally decompose on heating, liberating carbon dioxide leaving behind an oxide of the metal.[2] This process is called calcination, after calx, the Latin name of quicklime or calcium oxide, CaO, which is obtained by roasting limestone in a lime kiln:
- CaCO3 → CaO + CO2
As illustrated by its affinity for Ca2+, carbonate is a ligand for many metal cations. Transition metal carbonate and bicarbonate complexes feature metal ions covalently bonded to carbonate in a variety of bonding modes.
Lithium, sodium, potassium, rubidium, caesium, and ammonium carbonates are water-soluble salts, but carbonates of 2+ and 3+ ions are often poorly soluble in water. Of the insoluble metal carbonates, CaCO3 is important because, in the form of scale, it accumulates in and impedes flow through pipes. Hard water has the source of this material.
Acidification of carbonates generally liberates carbon dioxide:
- CaCO3 + 2 HCl → CaCl2 + CO2 + H2O
Thus, scale can be removed with acid.
In solution the equilibrium between carbonate, bicarbonate, carbon dioxide and carbonic acid is sensitive to pH, temperature, and pressure. Although di- and trivalent carbonates have low solubility, bicarbonate salts are far more soluble. This difference is related to the disparate lattice energies of solids composed of mono- vs dianions, as well as mono- vs dications.
In aqueous solution, carbonate, bicarbonate, carbon dioxide, and carbonic acid participate in a dynamic equilibrium. In strongly basic conditions, the carbonate ion predominates, while in weakly basic conditions, the bicarbonate ion is prevalent. In more acid conditions, aqueous carbon dioxide, CO2(aq), is the main form, which, with water, H2O, is in equilibrium with carbonic acid – the equilibrium lies strongly towards carbon dioxide. Thus sodium carbonate is basic, sodium bicarbonate is weakly basic, while carbon dioxide itself is a weak acid.
Organic carbonates
In organic chemistry a carbonate can also refer to a functional group within a larger molecule that contains a carbon atom bound to three oxygen atoms, one of which is double bonded. These compounds are also known as organocarbonates or carbonate esters, and have the general formula R−O−C(=O)−O−R′, or RR′CO3. Important organocarbonates include dimethyl carbonate, the cyclic compounds ethylene carbonate and propylene carbonate, and the phosgene replacement, triphosgene.
Buffer
Three reversible reactions control the pH balance of blood and act as a buffer to stabilise it in the range 7.37–7.43:[9][10]
- H+ + HCO−3 ⇌ H2CO3
- H2CO3 ⇌ CO2(aq) + H2O
- CO2(aq) ⇌ CO2(g)
Exhaled CO2(g) depletes CO2(aq), which in turn consumes H2CO3, causing the equilibrium of the first reaction to try to restore the level of carbonic acid by reacting bicarbonate with a hydrogen ion, an example of
Crucially, a similar buffer operates in the oceans. It is a major factor in climate change and the long-term carbon cycle, due to the large number of marine organisms (especially coral) which are made of calcium carbonate. Increased solubility of carbonate through increased temperatures results in lower production of marine calcite and increased concentration of atmospheric carbon dioxide. This, in turn, increases Earth temperature. The amount of CO2−3 available is on a geological scale and substantial quantities may eventually be redissolved into the sea and released to the atmosphere, increasing CO2 levels even more.[12]
Carbonate salts
- Carbonate overview:
Presence outside Earth
It is generally thought that the presence of carbonates in rock is strong evidence for the presence of liquid water. Recent observations of the planetary nebula NGC 6302 show evidence for carbonates in space,[13] where aqueous alteration similar to that on Earth is unlikely. Other minerals have been proposed which would fit the observations.
Until recently
See also
- Cap carbonates
- Orthocarbonic acid, H4CO4, or C(OH)4, a hypothetic unstable molecule
- Oxalate
- Peroxocarbonate
- Sodium percarbonate
References
- ^ a b Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica (11th ed.). Cambridge University Press.
- S2CID 245455194.
- ISSN 0010-938X.
- S2CID 258197818.
- S2CID 234180559.
- ^ Anodic generation of hydrogen peroxide in continuous flow, DOI: 10.1039/D2GC02575B (Paper) Green Chem., 2022, 24, 7931-7940
- ^ Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005 (PDF), IUPAC, p. 137, archived (PDF) from the original on 2017-05-18
- ^ "Chemical of the Week -- Biological Buffers". Archived from the original on 2011-07-21. Retrieved 2010-09-05.
- ^ Acid–Base Regulation and Disorders at Merck Manual of Diagnosis and Therapy Professional Edition
- ISBN 978-1-292-09493-9.
- IPCC (2019). "Summary for Policymakers"(PDF). IPCC Special Report on the Ocean and Cryosphere in a Changing Climate. pp. 3–35.
- ^ Kemper, F., Molster, F.J., Jager, C. and Waters, L.B.F.M. (2001) The mineral composition and spatial distribution of the dust ejecta of NGC 6302. Astronomy & Astrophysics 394, 679–690.
- (PDF) from the original on 2017-09-22.
- hdl:1893/17165. Archived(PDF) from the original on 2017-08-08.