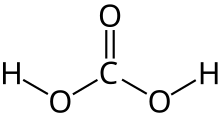
Carbonic acid
| |

| |
| Names | |
|---|---|
| IUPAC name
Carbonic acid[1]
| |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.133.015 |
| EC Number |
|
| 25554 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| H 2CO 3 | |
| Appearance | Colorless gas |
| Melting point | −53 °C (−63 °F; 220 K)[2] (sublimes) |
| Boiling point | 127 °C (261 °F; 400 K) (decomposes) |
| Reacts to form carbon dioxide and water | |
| Acidity (pKa) | pKa1 = 3.6 at 25 °C pKa2 = 10.329 |
Conjugate base
|
Bicarbonate, carbonate |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Structure | |
| monoclinic | |
| p21/c, No. 14 | |
| - | |
a = 5.392 Å, b = 6.661 Å, c = 5.690 Å α = 90°, β = 92.66°, γ = 90°[3] (D
2CO 3 at 1.85 GPa, 298 K) | |
Lattice volume (V)
|
204.12 Å3 |
Formula units (Z)
|
4 formula per cell |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Carbonic acid is a chemical compound with the chemical formula H2CO3. The molecule rapidly converts to water and carbon dioxide in the presence of water. However, in the absence of water, it is (contrary to popular belief) quite stable at room temperature.[4][5] The interconversion of carbon dioxide and carbonic acid is related to the breathing cycle of animals and the acidification of natural waters.[3]
In biochemistry and physiology, the name "carbonic acid" is sometimes applied to aqueous solutions of carbon dioxide. These chemical species play an important role in the bicarbonate buffer system, used to maintain acid–base homeostasis.[6]
Terminology in biochemical literature
In chemistry, the term "carbonic acid" strictly refers to the chemical compound with the formula H
2CO
3. Some biochemistry literature effaces the distinction between carbonic acid and carbon dioxide dissolved in extracellular fluid.
In
Anhydrous carbonic acid
At ambient temperatures, pure carbonic acid is a stable gas.
Carbonic acid
At low temperatures and
According to
In aqueous solution
In even a slight presence of water, carbonic acid dehydrates to carbon dioxide and water, which then catalyzes further decomposition.[5] For this reason, carbon dioxide can be considered the carbonic acid anhydride.
The
In biological solutions
In the presence of the enzyme carbonic anhydrase, equilibrium is instead reached rapidly, and the following reaction takes precedence:[13]
When the created carbon dioxide exceeds its solubility, gas evolves and a third equilibrium
The two reactions can be combined for the equilibrium in solution:
Under high CO2 partial pressure
In the
Significant amounts of molecular H
2CO
3 exist in aqueous solutions subjected to pressures of multiple
Relationship to bicarbonate and carbonate
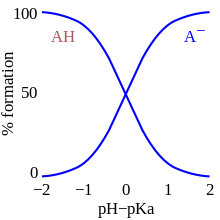
Carbonic acid is the formal
To interpret these numbers, note that two chemical species in an acid equilibrium are equiconcentrated when pK = pH. In particular, the extracellular fluid (cytosol) in biological systems exhibits pH ≈ 7.2, so that carbonic acid will be almost 50%-dissociated at equilibrium.
Ocean acidification
The
It has been estimated that the increase in dissolved carbon dioxide has already caused the ocean's average surface pH to decrease by about 0.1 from pre-industrial levels.Further reading
- Popular Science MonthlyVolume 59, July 1901
- Welch, M. J.; Lifton, J. F.; Seck, J. A. (1969). "Tracer studies with radioactive oxygen-15. Exchange between carbon dioxide and water". .
- Jolly, W. L. (1991). Modern Inorganic Chemistry (2nd ed.). McGraw-Hill. ISBN 978-0-07-112651-9.
- Moore, M. H.; Khanna, R. (1991). "Infrared and Mass Spectral Studies of Proton Irradiated H2O+CO2 Ice: Evidence for Carbonic Acid Ice: Evidence for Carbonic Acid". .
- W. Hage, K. R. Liedl; Liedl, E.; Hallbrucker, A; Mayer, E (1998). "Carbonic Acid in the Gas Phase and Its Astrophysical Relevance". PMID 9478889.
- Hage, W.; Hallbrucker, A.; Mayer, E. (1995). "A Polymorph of Carbonic Acid and Its Possible Astrophysical Relevance". .
References
- ISBN 978-0-85404-182-4.
- ^ PMID 9478889.
- ^ ISSN 2304-6740.
- ISBN 978-0-08-037941-8.
- ^ PMID 10760883.
- ^ Acid-Base Physiology 2.1 – Acid-Base Balance by Kerry Brandis.
- ^ S2CID 240250750.
- ^ S2CID 17094010.
- .
- ^ PMID 17944463.
- OCLC 56834315.
- .
- PMID 9336012.
- ISSN 1365-3075.
- PMID 26813580.
- S2CID 195791860.
- PMID 27580525.
- IUPAC (2006). "Stability constants" (database).
- PMID 26862781.
- S2CID 4417880.
- from the original on 6 July 2008. Retrieved 22 June 2021.




![{\displaystyle {\begin{aligned}{\ce {HCO3^{-}{}+ H+{}<=> CO2(soln){}+ H2O}}&&K_{3}={\frac {[{\ce {H+}}][{\ce {HCO3^-}}]}{[{\ce {CO2(soln)}}]}}\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a3efe827e57da672cf1c617cebeddb8cf04baa81)
![{\displaystyle {\begin{aligned}{\ce {CO3^{2-}{}+ H+{}<=> HCO3^-}}&&\beta _{1}={\frac {[{\ce {HCO3^-}}]}{[{\ce {H+}}][{\ce {CO3^{2-}}}]}}\\{\ce {CO3^{2-}{}+ 2H+{}<=> H2CO3}}&&\beta _{2}={\frac {[{\ce {H2CO3}}]}{[{\ce {H+}}]^{2}[{\ce {CO3^{2-}}}]}}\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/09f8d396f1143f199cf1136f1ad60c7ce873b848)

