Cephalopod
| Cephalopoda Temporal range:
| |
|---|---|

| |
| Extant and extinct cephalopods; clockwise from top-left: Orthosphynctes, Clarkeiteuthis conocauda, and common cuttlefish (Sepia officinalis)
| |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Mollusca |
| Subphylum: | Conchifera |
| Class: | Cephalopoda Cuvier, 1797 |
| Subclasses | |
| |
A cephalopod
Cephalopods became dominant during the
Distribution
There are over 800
Cephalopods are found in all the oceans of Earth. None of them can tolerate fresh water, but the brief squid, Lolliguncula brevis, found in Chesapeake Bay, is a notable partial exception in that it tolerates brackish water.[6] Cephalopods are thought to be unable to live in fresh water due to multiple biochemical constraints, and in their >400 million year existence have never ventured into fully freshwater habitats.[7]
Cephalopods occupy most of the depth of the ocean, from the abyssal plains to the sea surface, and have also been found in the hadal zone.[8] Their diversity is greatest near the equator (~40 species retrieved in nets at 11°N by a diversity study) and decreases towards the poles (~5 species captured at 60°N).[9]
Biology
Nervous system and behavior
Cephalopods are widely regarded as the most intelligent of the
The brain is protected in a
Many cephalopods are social creatures; when isolated from their own kind, some species have been observed shoaling with fish.[15]
Some cephalopods are able to fly through the air for distances of up to 50 metres (160 ft). While cephalopods are not particularly aerodynamic, they achieve these impressive ranges by jet-propulsion; water continues to be expelled from the funnel while the organism is in the air.[16] The animals spread their fins and tentacles to form wings and actively control lift force with body posture.[17] One species, Todarodes pacificus, has been observed spreading tentacles in a flat fan shape with a mucus film between the individual tentacles,[17][18] while another, Sepioteuthis sepioidea, has been observed putting the tentacles in a circular arrangement.[19]
Senses
Cephalopods have advanced vision, can detect gravity with statocysts, and have a variety of chemical sense organs.[9]: 34 Octopuses use their arms to explore their environment and can use them for depth perception.[9]
Vision

Most cephalopods rely on vision to detect predators and prey and to communicate with one another.[20] Consequently, cephalopod vision is acute: training experiments have shown that the common octopus can distinguish the brightness, size, shape, and horizontal or vertical orientation of objects. The morphological construction gives cephalopod eyes the same performance as shark eyes; however, their construction differs, as cephalopods lack a cornea and have an everted retina.[20] Cephalopods' eyes are also sensitive to the plane of polarization of light.[21] Unlike many other cephalopods,

All octopuses
In 2015, a novel mechanism for spectral discrimination in cephalopods was described. This relies on the exploitation of chromatic aberration (wavelength-dependence of focal length). Numerical modeling shows that chromatic aberration can yield useful chromatic information through the dependence of image acuity on accommodation. The unusual off-axis slit and annular pupil shapes in cephalopods enhance this ability by acting as prisms which are scattering white light in all directions.[29][30]
Photoreception
In 2015, molecular evidence was published indicating that cephalopod chromatophores are photosensitive; reverse transcription polymerase chain reactions (RT-PCR) revealed transcripts encoding rhodopsin and retinochrome within the retinas and skin of the longfin inshore squid (Doryteuthis pealeii), and the common cuttlefish (Sepia officinalis) and broadclub cuttlefish (Sepia latimanus). The authors claim this is the first evidence that cephalopod dermal tissues may possess the required combination of molecules to respond to light.[31]
Hearing
Some squids have been shown to detect sound using their statocysts,[32] but, in general, cephalopods are deaf.
Use of light
Most cephalopods possess an assemblage of skin components that interact with light. These may include iridophores, leucophores, chromatophores and (in some species) photophores. Chromatophores are colored pigment cells that expand and contract in accordance to produce color and pattern which they can use in a startling array of fashions.[9][31] As well as providing camouflage with their background, some cephalopods bioluminesce, shining light downwards to disguise their shadows from any predators that may lurk below.[9] The bioluminescence is produced by bacterial symbionts; the host cephalopod is able to detect the light produced by these organisms.[33] Bioluminescence may also be used to entice prey, and some species use colorful displays to impress mates, startle predators, or even communicate with one another.[9]
Coloration
Cephalopods can change their colors and patterns in milliseconds, whether for
Cephalopods can use chromatophores like a muscle, which is why they can change their skin hue as rapidly as they do. Coloration is typically stronger in near-shore species than those living in the open ocean, whose functions tend to be restricted to disruptive camouflage.[9]: 2 These chromatophores are found throughout the body of the octopus, however, they are controlled by the same part of the brain that controls elongation during jet propulsion to reduce drag. As such, jetting octopuses can turn pale because the brain is unable to achieve both controlling elongation and controlling the chromatophores.[37] Most octopuses mimic select structures in their field of view rather than becoming a composite color of their full background.[38]
Evidence of original coloration has been detected in cephalopod fossils dating as far back as the Silurian; these orthoconic individuals bore concentric stripes, which are thought to have served as camouflage.[39] Devonian cephalopods bear more complex color patterns, of unknown function.[40]
Chromatophores
Coleoids, a shell-less subclass of cephalopods (squid, cuttlefish, and octopuses), have complex pigment containing cells called chromatophores which are capable of producing rapidly changing color patterns. These cells store pigment within an elastic sac which produces the color seen from these cells. Coleoids can change the shape of this sac, called the cytoelastic sacculus, which then causes changes in the translucency and opacity of the cell. By rapidly changing multiple chromatophores of different colors, cephalopods are able to change the color of their skin at astonishing speeds, an adaptation that is especially notable in an organism that sees in black and white. Chromatophores are known to only contain three pigments, red, yellow, and brown, which cannot create the full color spectrum.[41] However, cephalopods also have cells called iridophores, thin, layered protein cells that reflect light in ways that can produce colors chromatophores cannot.[42] The mechanism of iridophore control is unknown, but chromatophores are under the control of neural pathways, allowing the cephalopod to coordinate elaborate displays. Together, chromatophores and iridophores are able to produce a large range of colors and pattern displays.
Adaptive value
Cephalopods utilize chromatophores' color changing ability in order to camouflage themselves. Chromatophores allow Coleoids to blend into many different environments, from coral reefs to the sandy sea floor. The color change of chromatophores works in concert with papillae, epithelial tissue which grows and deforms through hydrostatic motion to change skin texture. Chromatophores are able to perform two types of camouflage, mimicry and color matching. Mimicry is when an organism changes its appearance to appear like a different organism. The squid Sepioteuthis sepioide has been documented changing its appearance to appear as the non threatening herbivorous parrotfish to approach unaware prey. The octopus Thaumoctopus mimicus is known to mimic a number of different venomous organisms it cohabitates with to deter predators.[43] While background matching, a cephalopod changes its appearance to resemble its surroundings, hiding from its predators or concealing itself from prey. The ability to both mimic other organisms and match the appearance of their surroundings is notable given that cephalopods' vision is monochromatic.
Cephalopods also use their fine control of body coloration and patterning to perform complex signaling displays for both conspecific and intraspecific communication. Coloration is used in concert with locomotion and texture to send signals to other organisms. Intraspecifically this can serve as a warning display to potential predators. For example, when the octopus Callistoctopus macropus is threatened, it will turn a bright red brown color speckled with white dots as a high contrast display to startle predators. Conspecifically, color change is used for both mating displays and social communication. Cuttlefish have intricate mating displays from males to females. There is also male to male signaling that occurs during competition over mates, all of which are the product of chromatophore coloration displays.
Origin
There are two hypotheses about the evolution of color change in cephalopods. One hypothesis is that the ability to change color may have evolved for social, sexual, and signaling functions. Another explanation is that it first evolved because of selective pressures encouraging predator avoidance and stealth hunting.
For color change to have evolved as the result of social selection the environment of cephalopods' ancestors would have to fit a number of criteria. One, there would need to be some kind of mating ritual that involved signaling. Two, they would have to experience demonstrably high levels of sexual selection. And three, the ancestor would need to communicate using sexual signals that are visible to a conspecific receiver. For color change to have evolved as the result of natural selection different parameters would have to be met. For one, you would need some phenotypic diversity in body patterning among the population. The species would also need to cohabitate with predators which rely on vision for prey identification. These predators should have a high range of visual sensitivity, detecting not just motion or contrast but also colors. The habitats they occupy would also need to display a diversity of backgrounds.[44] Experiments done in Dwarf chameleons testing these hypotheses showed that chameleon taxa with greater capacity for color change had more visually conspicuous social signals but did not come from more visually diverse habitats, suggesting that color change ability likely evolved to facilitate social signaling, while camouflage is a useful byproduct.[45] Because camouflage is used for multiple adaptive purposes in cephalopods, color change could have evolved for one use and the other developed later, or it evolved to regulate trade offs within both.
Convergent evolution
Color change is widespread in ectotherms including anoles, frogs, mollusks, many fish, insects, and spiders.[46] The mechanism behind this color change can be either morphological or physiological. Morphological change is the result of a change in the density of pigment containing cells and tends to change over longer periods of time. Physiological change, the kind observed in cephalopod lineages, is typically the result of the movement of pigment within the chromatophore, changing where different pigments are localized within the cell. This physiological change typically occurs on much shorter timescales compared to morphological change. Cephalopods have a rare form of physiological color change which utilizes neural control of muscles to change the morphology of their chromatophores. This neural control of chromatophores has evolved convergently in both cephalopods and teleosts fishes.[47]
Ink
With the exception of the
The ink sac of cephalopods has led to a common name of "inkfish",[49] formerly the pen-and-ink fish.[50]
Circulatory system
Cephalopods are the only molluscs with a closed circulatory system.
Like most molluscs, cephalopods use hemocyanin, a copper-containing protein, rather than hemoglobin, to transport oxygen. As a result, their blood is colorless when deoxygenated and turns blue when bonded to oxygen.[52] In oxygen-rich environments and in acidic water, hemoglobin is more efficient, but in environments with little oxygen and in low temperatures, hemocyanin has the upper hand.[53][54][55] The hemocyanin molecule is much larger than the hemoglobin molecule, allowing it to bond with 96 O2 or CO2 molecules, instead of the hemoglobin's just four. But unlike hemoglobin, which are attached in millions on the surface of a single red blood cell, hemocyanin molecules float freely in the bloodstream.[56]
Respiration
Cephalopods exchange gases with the seawater by forcing water through their gills, which are attached to the roof of the organism.[57]: 488 [58] Water enters the mantle cavity on the outside of the gills, and the entrance of the mantle cavity closes. When the mantle contracts, water is forced through the gills, which lie between the mantle cavity and the funnel. The water's expulsion through the funnel can be used to power jet propulsion. If respiration is used concurrently with jet propulsion, large losses in speed or oxygen generation can be expected.[59][60] The gills, which are much more efficient than those of other mollusks, are attached to the ventral surface of the mantle cavity.[58] There is a trade-off with gill size regarding lifestyle. To achieve fast speeds, gills need to be small – water will be passed through them quickly when energy is needed, compensating for their small size. However, organisms which spend most of their time moving slowly along the bottom do not naturally pass much water through their cavity for locomotion; thus they have larger gills, along with complex systems to ensure that water is constantly washing through their gills, even when the organism is stationary.[57] The water flow is controlled by contractions of the radial and circular mantle cavity muscles.[61]
The gills of cephalopods are supported by a skeleton of robust fibrous proteins; the lack of mucopolysaccharides distinguishes this matrix from cartilage.[62][63] The gills are also thought to be involved in excretion, with NH4+ being swapped with K+ from the seawater.[58]
Locomotion and buoyancy

While most cephalopods can move by
Cephalopods employ a similar method of propulsion despite their increasing size (as they grow) changing the dynamics of the water in which they find themselves. Thus their paralarvae do not extensively use their fins (which are less efficient at low Reynolds numbers) and primarily use their jets to propel themselves upwards, whereas large adult cephalopods tend to swim less efficiently and with more reliance on their fins.[66]

Early cephalopods are thought to have produced jets by drawing their body into their shells, as Nautilus does today.
Some octopus species are also able to walk along the seabed. Squids and cuttlefish can move short distances in any direction by rippling of a flap of muscle around the mantle.
While most cephalopods float (i.e. are neutrally buoyant or nearly so; in fact most cephalopods are about 2–3% denser than seawater[15]), they achieve this in different ways.[64] Some, such as Nautilus, allow gas to diffuse into the gap between the mantle and the shell; others allow purer water to ooze from their kidneys, forcing out denser salt water from the body cavity;[64] others, like some fish, accumulate oils in the liver;[64] and some octopuses have a gelatinous body with lighter chloride ions replacing sulfate in the body chemistry.[64]
Squids are the primary sufferers of negative buoyancy in cephalopods. The negative buoyancy means that some squids, especially those whose habitat depths are rather shallow, have to actively regulate their vertical positions. This means that they must expend energy, often through jetting or undulations, in order to maintain the same depth. As such, the cost of transport of many squids are quite high. That being said, squid and other cephalopod that dwell in deep waters tend to be more neutrally buoyant which removes the need to regulate depth and increases their locomotory efficiency.[75][59]
The Macrotritopus defilippi, or the sand-dwelling octopus, was seen mimicking both the coloration and the swimming movements of the sand-dwelling flounder Bothus lunatus to avoid predators. The octopuses were able to flatten their bodies and put their arms back to appear the same as the flounders as well as move with the same speed and movements.[76]
Females of two species, Ocythoe tuberculata and Haliphron atlanticus, have evolved a true swim bladder.[77]
Octopus vs. squid locomotion
Two of the categories of cephalopods, octopus and squid, are vastly different in their movements despite being of the same class. Octopuses are generally not seen as active swimmers; they are often found scavenging the sea floor instead of swimming long distances through the water. Squids, on the other hand, can be found to travel vast distances, with some moving as much as 2000 km in 2.5 months at an average pace of 0.9 body lengths per second.[78] There is a major reason for the difference in movement type and efficiency: anatomy.
Both octopuses and squids have mantles (referenced above) which function towards respiration and locomotion in the form of jetting. The composition of these mantles differs between the two families, however. In octopuses, the mantle is made up of three muscle types: longitudinal, radial, and circular. The longitudinal muscles run parallel to the length of the octopus and they are used in order to keep the mantle the same length throughout the jetting process. Given that they are muscles, it can be noted that this means the octopus must actively flex the longitudinal muscles during jetting in order to keep the mantle at a constant length. The radial muscles run perpendicular to the longitudinal muscles and are used to thicken and thin the wall of the mantle. Finally, the circular muscles are used as the main activators in jetting. They are muscle bands that surround the mantle and expand/contract the cavity. All three muscle types work in unison to produce a jet as a propulsion mechanism.[78]
Squids do not have the longitudinal muscles that octopus do. Instead, they have a tunic.[78] This tunic is made of layers of collagen and it surrounds the top and the bottom of the mantle. Because they are made of collagen and not muscle, the tunics are rigid bodies that are much stronger than the muscle counterparts. This provides the squids some advantages for jet propulsion swimming. The stiffness means that there is no necessary muscle flexing to keep the mantle the same size. In addition, tunics take up only 1% of the squid mantle's wall thickness, whereas the longitudinal muscle fibers take up to 20% of the mantle wall thickness in octopuses.[78] Also because of the rigidity of the tunic, the radial muscles in squid can contract more forcefully.
The mantle is not the only place where squids have collagen. Collagen fibers are located throughout the other muscle fibers in the mantle. These collagen fibers act as elastics and are sometimes named "collagen springs".[78] As the name implies, these fibers act as springs. When the radial and circular muscles in the mantle contract, they reach a point where the contraction is no longer efficient to the forward motion of the creature. In such cases, the excess contraction is stored in the collagen which then efficiently begins or aids in the expansion of the mantle at the end of the jet. In some tests, the collagen has been shown to be able to begin raising mantle pressure up to 50ms before muscle activity is initiated.[78]
These anatomical differences between squid and octopuses can help explain why squid can be found swimming comparably to fish while octopuses usually rely on other forms of locomotion on the sea floor such as bipedal walking, crawling, and non-jetting swimming.[79]
Shell
Females of the octopus genus Argonauta secrete a specialized paper-thin egg case in which they reside, and this is popularly regarded as a "shell", although it is not attached to the body of the animal and has a separate evolutionary origin.
The largest group of shelled cephalopods, the
The deposition of carbonate, leading to a mineralized shell, appears to be related to the acidity of the organic shell matrix (see Mollusc shell); shell-forming cephalopods have an acidic matrix, whereas the gladius of squid has a basic matrix.[89] The basic arrangement of the cephalopod outer wall is: an outer (spherulitic) prismatic layer, a laminar (nacreous) layer and an inner prismatic layer. The thickness of every layer depends on the taxa.[90] In modern cephalopods, the Ca carbonate is aragonite. As for other mollusc shells or coral skeletons, the smallest visible units are irregular rounded granules.[91]
Head appendages
Cephalopods, as the name implies, have muscular appendages extending from their heads and surrounding their mouths. These are used in feeding, mobility, and even reproduction. In
The tentacle consists of a thick central nerve cord (which must be thick to allow each sucker to be controlled independently)[93] surrounded by circular and radial muscles. Because the volume of the tentacle remains constant, contracting the circular muscles decreases the radius and permits the rapid increase in length. Typically a 70% lengthening is achieved by decreasing the width by 23%.[1]: 227 The shorter arms lack this capability.
The size of the tentacle is related to the size of the buccal cavity; larger, stronger tentacles can hold prey as small bites are taken from it; with more numerous, smaller tentacles, prey is swallowed whole, so the mouth cavity must be larger.[94]
Externally shelled
Feeding

All living cephalopods have a two-part beak;[9]: 7 most have a radula, although it is reduced in most octopus and absent altogether in Spirula.[9]: 7 [95]: 110 They feed by capturing prey with their tentacles, drawing it into their mouth and taking bites from it.[22] They have a mixture of toxic digestive juices, some of which are manufactured by symbiotic algae, which they eject from their salivary glands onto their captured prey held in their mouths. These juices separate the flesh of their prey from the bone or shell.[22] The salivary gland has a small tooth at its end which can be poked into an organism to digest it from within.[22]
The digestive gland itself is rather short.[22] It has four elements, with food passing through the crop, stomach and caecum before entering the intestine. Most digestion, as well as the absorption of nutrients, occurs in the digestive gland, sometimes called the liver. Nutrients and waste materials are exchanged between the gut and the digestive gland through a pair of connections linking the gland to the junction of the stomach and caecum.[22] Cells in the digestive gland directly release pigmented excretory chemicals into the lumen of the gut, which are then bound with mucus passed through the anus as long dark strings, ejected with the aid of exhaled water from the funnel.[22] Cephalopods tend to concentrate ingested heavy metals in their body tissue.[96] However, octopus arms use a family of cephalopod-specific chemotactile receptors (CRs) to be their "taste by touch" system.[97]
Radula

The cephalopod radula consists of multiple symmetrical rows of up to nine teeth[98] – thirteen in fossil classes.[99] The organ is reduced or even vestigial in certain octopus species and is absent in Spirula.[99] The teeth may be homodont (i.e. similar in form across a row), heterodont (otherwise), or ctenodont (comb-like).[99] Their height, width and number of cusps is variable between species.[99] The pattern of teeth repeats, but each row may not be identical to the last; in the octopus, for instance, the sequence repeats every five rows.[99]: 79
Cephalopod radulae are known from fossil deposits dating back to the Ordovician.[100] They are usually preserved within the cephalopod's body chamber, commonly in conjunction with the mandibles; but this need not always be the case;[101] many radulae are preserved in a range of settings in the Mason Creek.[102] Radulae are usually difficult to detect, even when they are preserved in fossils, as the rock must weather and crack in exactly the right fashion to expose them; for instance, radulae have only been found in nine of the 43 ammonite genera,[103][clarification needed] and they are rarer still in non-ammonoid forms: only three pre-Mesozoic species possess one.[100]
Excretory system
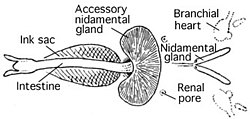
Most cephalopods possess a single pair of large
Nautilus, unusually, possesses four nephridia, none of which are connected to the pericardial cavities.
The incorporation of
Reproduction and life cycle




Cephalopods are a diverse group of species, but share common life history traits, for example, they have a rapid growth rate and short life spans.[107] Stearns (1992) suggested that in order to produce the largest possible number of viable offspring, spawning events depend on the ecological environmental factors of the organism. The majority of cephalopods do not provide parental care to their offspring, except, for example, octopus, which helps this organism increase the survival rate of their offspring.[107] Marine species' life cycles are affected by various environmental conditions.[108] The development of a cephalopod embryo can be greatly affected by temperature, oxygen saturation, pollution, light intensity, and salinity.[107] These factors are important to the rate of embryonic development and the success of hatching of the embryos. Food availability also plays an important role in the reproductive cycle of cephalopods. A limitation of food influences the timing of spawning along with their function and growth.[108] Spawning time and spawning vary among marine species; it's correlated with temperature, though cephalopods in shallow water spawn in cold months so that the offspring would hatch at warmer temperatures. Breeding can last from several days to a month.[107]
Sexual maturity
Cephalopods that are sexually mature and of adult size begin spawning and reproducing. After the transfer of genetic material to the following generation, the adult cephalopods in most species then die.[107] Sexual maturation in male and female cephalopods can be observed internally by the enlargement of gonads and accessory glands.[109] Mating would be a poor indicator of sexual maturation in females; they can receive sperm when not fully reproductively mature and store them until they are ready to fertilize the eggs.[108] Males are more aggressive in their pre-mating competition when in the presence of immature females than when competing for a sexually mature female.[110] Most cephalopod males develop a hectocotylus, an arm tip which is capable of transferring their spermatozoa into the female mantle cavity. Though not all species use a hectocotylus; for example, the adult nautilus releases a spadix.[111] Some male squids, mainly deep-water species, have instead evolved a penis longer than their own body length, the longest penis in any free-living animals. It is assumed these males simply attach a spermatophore anywhere on a female's body.[112] An indication of sexual maturity of females is the development of brachial photophores to attract mates.[113]
Fertilization
Cephalopods are not
Male–male competition
Most cephalopods engage in aggressive sex: a protein in the male capsule sheath stimulates this behavior. They also engage in male–male aggression, where larger males tend to win the interactions.[107] When a female is near, the males charge one another continuously and flail their arms. If neither male backs away, the arms extend to the back, exposing the mouth, followed by the biting of arm tips.[122] During mate competition males also participate in a technique called flushing. This technique is used by the second male attempting to mate with a female. Flushing removes spermatophores in the buccal cavity that was placed there by the first mate by forcing water into the cavity.[107] Another behavior that males engage in is sneaker mating or mimicry – smaller males adjust their behavior to that of a female in order to reduce aggression. By using this technique, they are able to fertilize the eggs while the larger male is distracted by a different male.[122] During this process, the sneaker males quickly insert drop-like sperm into the seminal receptacle.[123]
Mate choice
Mate choice is seen in cuttlefish species, where females prefer some males over others, though characteristics of the preferred males are unknown.[107] A hypothesis states that females reject males by olfactory cues rather than visual cues.[107] Several cephalopod species are polyandrous – accepting and storing multiple male spermatophores, which has been identified by DNA fingerprinting.[114] Females are no longer receptive to mating attempts when holding their eggs in their arms. Females can store sperm in two places (1) the buccal cavity where recently mated males place their spermatophores, and (2) the internal sperm-storage receptacles where sperm packages from previous males are stored.[107] Spermatophore storage results in sperm competition; which states that the female controls which mate fertilizes the eggs. In order to reduce this sort of competition, males develop agonistic behaviors like mate guarding and flushing.[107] The Hapalochlaena lunulata, or the blue-ringed octopus, readily mates with both males and females.[124]
Sexual dimorphism
In a variety of marine organisms, it is seen that females are larger in size compared to the males in some closely related species. In some lineages, such as the blanket octopus, males become structurally smaller and smaller resembling a term, "dwarfism" dwarf males usually occurs at low densities.[125] The blanket octopus male is an example of sexual-evolutionary dwarfism; females grow 10,000 to 40,000 times larger than the males and the sex ratio between males and females can be distinguished right after hatching of the eggs.[125]

Embryology
Cephalopod eggs span a large range of sizes, from 1 to 30 mm in diameter.
The funnel of cephalopods develops on the top of their head, whereas the mouth develops on the opposite surface.
The shells develop from the ectoderm as an organic framework which is subsequently mineralized.[80] In Sepia, which has an internal shell, the ectoderm forms an invagination whose pore is sealed off before this organic framework is deposited.[80]
Development
Right: A mature
The length of time before hatching is highly variable; smaller eggs in warmer waters are the fastest to hatch, and newborns can emerge after as little as a few days. Larger eggs in colder waters can develop for over a year before hatching.[126]
The process from spawning to hatching follows a similar trajectory in all species, the main variable being the amount of yolk available to the young and when it is absorbed by the embryo.[126]
Unlike most other molluscs, cephalopods do not have a morphologically distinct
Growth in juveniles is usually
Evolution
The traditional view of cephalopod evolution holds that they evolved in the Late Cambrian from a
Possible early Cambrian remains have been found in the Avalon Peninsula, matching genetic data for a pre-Cambrian origin.[2]
However, some morphological evidence is difficult to reconcile with this view, and the redescription of Nectocaris pteryx, which did not have a shell and appeared to possess jet propulsion in the manner of "derived" cephalopods, complicated the question of the order in which cephalopod features developed – provided Nectocaris is a cephalopod at all.[134]
Early cephalopods were likely predators near the top of the food chain.
In the Early Palaeozoic, their range was far more restricted than today; they were mainly constrained to sublittoral regions of shallow shelves of the low latitudes, and usually occurred in association with thrombolites.[138] A more pelagic habit was gradually adopted as the Ordovician progressed.[138] Deep-water cephalopods, whilst rare, have been found in the Lower Ordovician – but only in high-latitude waters.[138] The mid-Ordovician saw the first cephalopods with septa strong enough to cope with the pressures associated with deeper water, and could inhabit depths greater than 100–200 m.[136] The direction of shell coiling would prove to be crucial to the future success of the lineages; endogastric coiling would only permit large size to be attained with a straight shell, whereas exogastric coiling – initially rather rare – permitted the spirals familiar from the fossil record to develop, with their corresponding large size and diversity.[139] (Endogastric means the shell is curved so as the ventral or lower side is longitudinally concave (belly in); exogastric means the shell is curved so as the ventral side is longitudinally convex (belly out) allowing the funnel to be pointed backward beneath the shell.)[139]
The ancestors of coleoids (including most modern cephalopods) and the ancestors of the modern nautilus, had diverged by the Floian Age of the Early Ordovician Period, over 470 million years ago.[138][140] The Bactritida, a Devonian–Triassic group of orthocones, are widely held to be paraphyletic without the coleoids and ammonoids, that is, the latter groups arose from within the Bactritida.[141]: 393 An increase in the diversity of the coleoids and ammonoids is observed around the start of the Devonian period and corresponds with a profound increase in fish diversity. This could represent the origin of the two derived groups.[141]
Unlike most modern cephalopods, most ancient varieties had protective shells. These shells at first were conical but later developed into curved nautiloid shapes seen in modern nautilus species. Competitive pressure from fish is thought to have forced the shelled forms into deeper water, which provided an evolutionary pressure towards shell loss and gave rise to the modern coleoids, a change which led to greater metabolic costs associated with the loss of buoyancy, but which allowed them to recolonize shallow waters.[132]: 36 However, some of the straight-shelled nautiloids evolved into belemnites, out of which some evolved into squid and cuttlefish.[verification needed] The loss of the shell may also have resulted from evolutionary pressure to increase maneuverability, resulting in a more fish-like habit.[1]: 289
There has been debate on the
Genetics
The sequencing of a full Cephalopod genome has remained challenging to researchers due to the length and repetition of their DNA.[145] The characteristics of Cephalopod genomes were initially hypothesized to be the result of entire genome duplications. Following the full sequencing of a California two-spot octopus, the genome showed similar patterns to other marine invertebrates with significant additions to the genome assumed to be unique to Cephalopods. No evidence of full genome duplication was found.[146]
Within the California two-spot octopus genome there are substantial replications of two gene families. Significantly, the expanded gene families were only previously known to exhibit replicative behaviour within vertebrates.[146] The first gene family was identified as the Protocadherins which are attributed to neuron development. Protocadherins function as cell adhesion molecules, essential for synaptic specificity. The mechanism for Protocadherin gene family replication in vertebrates is attributed to complex splicing, or cutting and pasting, from a locus. Following the sequencing of the California two-spot octopus, researchers found that the Prorocadherin gene family in Cephalopods has expanded in the genome due to tandem gene duplication. The different replication mechanisms for Protocadherin genes indicate an independent evolution of Protocadherin gene expansion in vertebrates and invertebrates.[146] Analysis of individual Cephalopod Protocadherin genes indicate independent evolution between species of Cephalopod. A species of shore squid Doryteuthis pealeii with expanded Protocadherin gene families differ significantly from those of the California two-spot octopus suggesting gene expansion did not occur before speciation within Cephalopods. Despite different mechanisms for gene expansion, the two-spot octopus Protocadherin genes were more similar to vertebrates than squid, suggesting a convergent evolution mechanism. The second gene family known as C2H2 are small proteins that function as zinc transcription factors. C2H2 are understood to moderate DNA, RNA and protein functions within the cell.[145]
The sequenced California two spot octopus genome also showed a significant presence of transposable elements as well as transposon expression. Although the role of transposable elements in marine vertebrates is still relatively unknown, significant expression of transposons in nervous system tissues have been observed.[147] In a study conducted on vertebrates, the expression of transposons during development in the fruitfly Drosophila melanogaster activated genomic diversity between neurons.[148] This diversity has been linked to increased memory and learning in mammals. The connection between transposons and increased neuron capability may provide insight into the observed intelligence, memory and function of Cephalopods.[147]
Using long-read sequencing, researchers have decoded the cephalopod genomes and discovered they have been churned and scrambled. The genes were compared to those of thousands of other species and while blocks of three or more genes co-occurred between squid and octopus, the blocks of genes were not found together in any other animals'. Many of the groupings were in the nervous tissue, suggesting the course they adapted their intelligence.[149][150]
Phylogeny
The approximate consensus of extant cephalopod phylogeny, after Whalen & Landman (2022), is shown in the cladogram.[151] Mineralized taxa are in bold.
| Cephalopoda |
| |||||||||||||||||||||||||||||||||||||||||||||
The internal phylogeny of the cephalopods is difficult to constrain; many molecular techniques have been adopted, but the results produced are conflicting.
Molecular estimates for clade divergence vary. One 'statistically robust' estimate has Nautilus diverging from Octopus at 415 ± 24 million years ago.[155]
Taxonomy




The classification presented here, for recent cephalopods, follows largely from Current Classification of Recent Cephalopoda (May 2001), for fossil cephalopods takes from Arkell et al. 1957, Teichert and Moore 1964, Teichert 1988, and others. The three subclasses are traditional, corresponding to the three orders of cephalopods recognized by Bather.[156]
Class Cephalopoda († indicates
- Subclass Nautiloidea: Fundamental ectocochliate cephalopods that provided the source for the Ammonoidea and Coleoidea.
- Order † Plectronocerida: the ancestral cephalopods from the Cambrian Period
- Order † Ellesmerocerida (500 to 470 Ma)
- Order † Endocerida (485 to 430 Ma)
- Order † Actinocerida (480 to 312 Ma)
- Order † Discosorida (482 to 392 Ma)
- Order † Pseudorthocerida (432 to 272 Ma)
- Order † Tarphycerida (485 to 386 Ma)
- Order † Oncocerida (478.5 to 324 Ma)
- Order Nautilida (extant; 410.5 Ma to present)
- Order † Orthocerida (482.5 to 211.5 Ma)
- Order † Ascocerida (478 to 412 Ma)
- Order † Bactritida (418.1 to 260.5 Ma)
- Subclass † Ammonoidea: Ammonites (479 to 66 Ma)
- Order † Goniatitida (388.5 to 252Ma)
- Order † Ceratitida (254 to 200 Ma)
- Order † Ammonitida (215 to 66 Ma)
- Order †
- Subclass Coleoidea (410.0 Ma-Rec)
- Cohort † Belemnoidea: Belemnites and kin
- Genus † Jeletzkya
- Order † Aulacocerida (265 to 183 Ma)
- Order † Phragmoteuthida (189.6 to 183 Ma)
- Order † Hematitida (339.4 to 318.1 Ma)
- Order † Belemnitida (339.4 to 66 Ma)
- Genus † Belemnoteuthis (189.6 to 183Ma)
- Cohort Neocoleoidea
- Superorder Decapodiformes (also known as Decabrachia or Decembranchiata)
- Order Spirulida: Ram's horn squid
- Order Sepiida: cuttlefish
- Order Sepiolida: pygmy, bobtail and bottletail squid
- Order Idiosepida
- Order Oegopsida: neritic squid
- Order Myopsida: coastal squid
- Order Bathyteuthida
- Superorder Octopodiformes (also known as Vampyropoda)
- Family † Trachyteuthididae
- Order Vampyromorphida: Vampire squid
- Order Octopoda: octopus
- Family †
- Superorder † Palaeoteuthomorpha
- Order † Boletzkyida
- Superorder Decapodiformes (also known as Decabrachia or Decembranchiata)
- Cohort † Belemnoidea: Belemnites and kin
Other classifications differ, primarily in how the various decapod orders are related, and whether they should be orders or families.
Suprafamilial classification of the Treatise
This is the older classification that combines those found in parts K and L of the Treatise on Invertebrate Paleontology, which forms the basis for and is retained in large part by classifications that have come later.
Nautiloids in general (Teichert and Moore, 1964) sequence as given.
- Subclass † Endoceratoidea. Not used by Flower, e.g. Flower and Kummel 1950, interjocerids included in the Endocerida.
- Order † Endocerida
- Order † Intejocerida
- Subclass † Actinoceratoidea Not used by Flower, ibid
- Order † Actinocerida
- Subclass Nautiloidea Nautiloidea in the restricted sense.
- Order † Ellesmerocerida Plectronocerida subsequently split off as separate order.
- Order † Orthocerida Includes orthocerids and pseudorthocerids
- Order † Ascocerida
- Order † Oncocerida
- Order † Discosorida
- Order † Tarphycerida
- Order † Barrandeocerida A polyphyletic group now included in the Tarphycerida
- Order Nautilida
- Subclass † Bactritoidea
- Order † Bactritida
Paleozoic Ammonoidea (Miller, Furnish and Schindewolf, 1957)
- Suborder † Anarcestina
- Suborder † Clymeniina
- Suborder † Goniatitina
- Suborder † Prolecanitina
- Suborder †
Mesozoic Ammonoidea (Arkel et al., 1957)
- Suborder † Ceratitina
- Suborder † Phylloceratina
- Suborder † Lytoceratina
- Suborder † Ammonitina
Subsequent revisions include the establishment of three Upper Cambrian orders, the Plectronocerida, Protactinocerida, and Yanhecerida; separation of the pseudorthocerids as the Pseudorthocerida, and elevating orthoceratid as the Subclass Orthoceratoidea.
Shevyrev classification
Shevyrev (2005) suggested a division into eight subclasses, mostly comprising the more diverse and numerous fossil forms,[157][158] although this classification has been criticized as arbitrary, lacking evidence, and based on misinterpretations of other papers.[159]


Class Cephalopoda
- Subclass † Ellesmeroceratoidea
- Order † Plectronocerida (501 to 490 Ma)
- Order † Protactinocerida
- Order † Yanhecerida
- Order † Ellesmerocerida (500 to 470 Ma)
- Subclass † Endoceratoidea (485 to 430 Ma)
- Order † Endocerida (485 to 430 Ma)
- Order † Intejocerida (485 to 480 Ma)
- Subclass † Actinoceratoidea
- Order † Actinocerida (480 to 312 Ma)
- Subclass Nautiloidea (490.0 Ma- Rec)
- Order † Basslerocerida (490 to 480 Ma)
- Order † Tarphycerida (485 to 386 Ma)
- Order † Lituitida (485 to 480 Ma)
- Order † Discosorida (482 to 392 Ma)
- Order † Oncocerida (478.5 to 324 Ma)
- Order Nautilida (410.5 Ma-Rec)
- Subclass † Orthoceratoidea (482.5 to 211.5 Ma)
- Order † Orthocerida (482.5 to 211.5 Ma)
- Order † Ascocerida (478 to 412 Ma)
- Order † Dissidocerida (479 to 457.5 Ma)
- Order † Bajkalocerida
- Subclass † Bactritoidea (422 to 252Ma)
- Subclass † Ammonoidea (410 to 66 Ma)
- Subclass Coleoidea (410.0 Ma-rec)[160]
Cladistic classification
Another recent system divides all cephalopods into two
The coleoids, despite some doubts,[1]: 289 appear from molecular data to be monophyletic.[163]
In culture
Ancient seafaring people were aware of cephalopods, as evidenced by such artworks as a stone carving found in the archaeological recovery from Bronze Age
The Kraken are legendary sea monsters of giant proportions said to dwell off the coasts of Norway and Greenland, usually portrayed in art as giant cephalopods attacking ships. Linnaeus included it in the first edition of his 1735 Systema Naturae.[166][167] In a Hawaiian creation myth that says the present cosmos is the last of a series which arose in stages from the ruins of the previous universe, the octopus is the lone survivor of the previous, alien universe.[168] The Akkorokamui is a gigantic tentacled monster from Ainu folklore.[169]
A battle with an octopus plays a significant role in Victor Hugo's book Travailleurs de la mer (Toilers of the Sea), relating to his time in exile on Guernsey.[170] Ian Fleming's 1966 short story collection Octopussy and The Living Daylights, and the 1983 James Bond film were partly inspired by Hugo's book.[171]
Japanese erotic art,
Its many arms that emanate from a common center means that the octopus is sometimes used to symbolize a powerful and manipulative organization.[175]
See also
- Cephalopod size
- Cephalopod eye
- Cephalopod intelligence
- Pain in cephalopods
- Kraken
- List of nautiloids
- List of ammonites
References
- ^ ISBN 0-12-728702-7
- ^ PMID 33758350.
- ISBN 978-1-138-33293-5.
- ^ "Welcome to CephBase". CephBase. Archived from the original on 12 January 2016. Retrieved 29 January 2016.
- ^ ISBN 0-12-728702-7
- .
- ^ "Are there any freshwater cephalopods?". ABC Science. 16 January 2013.
- ^ Cephalopods Observed At Record-Shattering Oceanic Depths
- ^ ISBN 978-0-19-852761-9.
- ISBN 978-94-007-7413-1.
- ISBN 978-3-7643-5076-5.
- PMID 31972515.
- ISBN 9780073383071.
- PMID 14077488.
- ^ S2CID 85088231.
- .
- ^ S2CID 84388744.
- ^ "Scientists Unravel Mystery of Flying Squid". Ocean Views. National Geographic. 20 February 2013.
- ^ Jabr, Ferris (2 August 2010). "Fact or Fiction: Can a Squid Fly out of Water?". Scientific American.
- ^ S2CID 2881223.
- doi:10.17161/to.v0i0.4226. Archived from the originalon 2016-08-22. Retrieved 2013-05-10.(subscription required)
- ^ ISBN 978-0-632-06048-1.
- ^ ISBN 978-0-521-64583-6.
- PMID 27629028.
- ^ Hanlon and Messenger, 68.
- PMID 20392722.
- PMID 9317205.
- S2CID 25707481.
- bioRxiv 10.1101/017756.
- ^ "Octopus Eyes Are Crazier Than We Imagined". Gizmodo. 2016-07-06. Archived from the original on 2023-04-30.
- ^ S2CID 25431963.
- ^ "The cephalopods can hear you". BBC News. 2009-06-15. Retrieved 2010-04-28.
- PMID 19509343.
- Encyclopædia Britannica 2006 Ultimate Reference Suite DVD.
- (PDF) from the original on 2016-08-09.
- PMC 8101055.
- S2CID 26862414.
- PMID 22649542.
- S2CID 54043278.
- S2CID 55851070.
- ^ Hanlon, R. et al. (2011) Rapid adaptive camouflage in cephalopods. in Animal Camouflage: Mechanisms and Function. Cambridge University Press, Cambridge, UK. pp145-161.
- ^ Cloney, R. A., & Brocco, S. L. (1983). Chromatophore organs, reflector cells, iridocytes and leucophores in cephalopods. American Zoologist, 23(3), 581–592. https://doi.org/10.1093/icb/23.3.581
- ^ Norman, M. D., Finn, J., & Tregenza, T. (2001). Dynamic mimicry in an Indo–Malayan octopus. Proceedings of the Royal Society of London. Series B: Biological Sciences, 268(1478), 1755–1758. https://doi.org/10.1098/rspb.2001.1708
- ^ Stevens, M., & Merilaita, S. (Eds.). (2011). Animal camouflage: Mechanisms and function. Cambridge University Press.
- ^ Stuart-Fox, D., & Moussalli, A. (2008). Selection for social signalling drives the evolution of chameleon colour change. PLoS Biology, 6(1), e25. https://doi.org/10.1371/journal.pbio.0060025
- ^ Bagnara, J. T., & Hadley, M. E. (1973). Chromatophores and color change: The comparative physiology of animal pigmentation. Prentice-Hall.
- ^ Demski, L. S. (1992). Chromatophore systems in teleosts and cephalopods: A levels oriented analysis of convergent systems. Brain, Behavior and Evolution, 40(2–3), 141–156. https://doi.org/10.1159/000113909
- ISBN 978-0-521-64583-6.
- ^ "inkfish". Merriam-Webster. Retrieved 1 February 2018.
- ^ Bickerdyke, John (1895). Sea Fishing. London: Longmans, Green, and Co. p. 114.
the common squid or calamary (Loligo vulgaris). It is sometimes called the pen-and-ink fish, on account of its ink bag, and the delicate elongated shell which is found within it.
- PMID 7373208.
- S2CID 33290596.
- ^ How many hearts does an octopus have? - New Scientist
- ^ Why Do Octopuses Have Three Hearts? - Science ABC
- ^ "Where Would We Be Without Blood? - Illinois Science Council". Archived from the original on 2023-04-25. Retrieved 2022-07-07.
- ^ Why is Octopus Blood Blue? | Science | Wilstar.com
- ^ ISBN 978-0-306-43513-3.
- ^ S2CID 20214206.
- ^ a b c Wells, M. J.; O'Dor, R. K. (1991-09-01). "Jet Propulsion and the Evolution of the Cephalopods". Bulletin of Marine Science. 49 (1–2): 419–432.
- PMID 4443736.
- (PDF) from the original on 2009-03-04.
- PMID 18722759.
- ^ See also http://tolweb.org/articles/?article_id=4200 Archived 2010-06-16 at the Wayback Machine
- ^ ISBN 0-12-728702-7
- ^ PMID 10952883.
- ^ PMID 21669828.
- S2CID 84822175.
- .
- ^ Campbell, Reece & Mitchell (1999), p. 612.
- S2CID 85770435.
- ^ a b Wells, Martin J.; O'Dor, R. K. (July 1991). "Jet Propulsion and the Evolution of the Cephalopods". Bulletin of Marine Science. 49 (1): 419–432(14).
- .
- ^ .
- .
- PMID 28581868.
- S2CID 12935620.
- ^ "The argonaut shell: Gas-mediated buoyancy control in a pelagic octopus".
- ^ JSTOR 24967551.
- S2CID 26862414.
- ^ S2CID 22241391.
- .
- ISBN 978-1420006391.
- .
- .
- ^ von Boletzky, Sigurd (July 1991). "The terminal spine of sepiolid hatchlings: its development and functional morphology (Mollusca, Cephalopoda)". Bulletin of Marine Science. 49: 107–112.
- ^ .
- S2CID 85026080.
- PMID 19665573.
- S2CID 83400480.
- OCLC 972899981.
- S2CID 126900936.
- S2CID 34858474.
- ^ Anatomy of the Common Squid. 1912.
- .
- ISBN 0-12-728702-7
- ^ C.Michael Hogan. 2011. Celtic Sea. eds. P.Saundry & C.Cleveland. Encyclopedia of Earth. National Council for Science and the Environment. Washington DC.
- .
- ^ "Cephalopod radula". Tree of Life web project.
- ^ .
- ^ .
- ISBN 978-1-4020-6461-6.
- ^ Richardson & ... (1977). Fossils of the Mason Creek.
- S2CID 206530342.
- ^ ISBN 978-0-03-056747-6.
- S2CID 87142440.
- ^ .
- ^ a b c d e f g h i j k Vidal, Erica A. G. Advances in Cephalopod Science: Biology, Ecology, Cultivation and Fisheries.
- ^ S2CID 41577834.
- ^ doi:10.2960/j.v12.a7.
- S2CID 85256742.
- S2CID 85899974.
- ^ Observation of penis elongation in Onykia ingens: implications for spermatophore transfer in deep-water squid
- S2CID 145374345.
- ^ .
- ^ Rare Deep-Sea Video Shows a Squid Mom Carrying Its Eggs For Safety
- ^ Cephalopod Behaviour
- ^ Structure and properties of the egg mass of the ommastrephid squid Todarodes pacificus
- ^ Deep-sea squid mom carries dazzling pearl-like string of eggs
- ^ Vampire squid reproductive strategy is unique among coleoid cephalopods
- ^ Individually unique, fixed stripe configurations of Octopus chierchiae allow for photoidentification in long-term studies
- S2CID 8693207.
- ^ S2CID 85748957.
- S2CID 43094931.
- S2CID 32899443.
- ^ a b Fairbairn, D. (2013). "Blanket Octopus: Drifting Females and Dwarf Males". Odd couples: Extraordinary differences between the sexes in the animal kingdom. Princeton University Press. pp. 104–115.
- ^ PMID 12846042.
- ^ S2CID 13109195.
- ISBN 978-0-306-43513-3.
- doi:10.1071/MF03147.
- ^ Lemche, H.; Wingstrand, K. G. (1959). "The anatomy of Neopilina galatheae Lemche, 1957 (Mollusca, Tryblidiacea)" (Link to free full text + plates). Galathea Report. 3: 9–73.
- ^ Wingstrand, K. G. (1985). "On the anatomy and relationships of Recent Monoplacophora". Galathea Report. 16: 7–94. Archived from the original (Link to free full text + plates) on 2016-03-03. Retrieved 2009-03-23.
- ^ ISBN 9780470995310.
- .
- S2CID 205055896.
- ISBN 978-81-322-3658-0.
- ^ .
- ^ Dzik, J. (1981). "Origin of the Cephalopoda" (PDF). Acta Palaeontologica Polonica. 26 (2): 161–191. Archived (PDF) from the original on 2008-12-16.
- ^ PMID 19789709.
- ^ S2CID 128629737.
- .
- ^ .
- ^ a b Tanabe, K. (2008). Cephalopods – Present and Past. Tokyo: Tokai University Press.[page needed]
- S2CID 33835096.
- S2CID 13109195.
- ^ PMID 29962956.
- ^ PMID 26268193.
- ^ ISBN 978-1-118-50015-6.
- PMID 25005482.
- ^ Caroline B. Albertin et al. May 4th, 2022 "Genome and transcriptome mechanisms driving cephalopod evolution" Nature Communications
- ^ Hannah Schimdbour et al. April 21, 2022 "Emergence of novel cephalopod gene regulation and expression through large-scale genome reorganization" Nature Communications
- PMID 35260548.
- PMID 15935706.
- S2CID 84743000.
- PMID 10958852.
- S2CID 4389953.
- ^ Bather, F.A. (1888b). "Professor Blake and Shell-Growth in Cephalopoda". Annals and Magazine of Natural History. 6. 1 (6): 421–426. .
- ^ Shevyrev, A. A. (2005). "The Cephalopod Macrosystem: A Historical Review, the Present State of Knowledge, and Unsolved Problems: 1. Major Features and Overall Classification of Cephalopod Mollusks". Paleontological Journal. 39 (6): 606–614. Translated from Paleontologicheskii Zhurnal No. 6, 2005, 33–42.
- ^
Shevyrev, A. A. (2006). "The cephalopod macrosystem; a historical review, the present state of knowledge, and unsolved problems; 2, Classification of nautiloid cephalopods". Paleontological Journal. 40 (1): 46–54. S2CID 84616115.
- ^ Kroger, B. "Peer review in the Russian 'Paleontological Journal'". Archived from the original on 2009-08-31.
- ^ Bather, F.A. (1888a). "Shell-growth in Cephalopoda (Siphonopoda)". Annals and Magazine of Natural History. 6. 1 (4): 298–310. .
- ^ Berthold, Thomas; Engeser, Theo (1987). "Phylogenetic analysis and systematization of the Cephalopoda (Mollusca)". Verhandlungen Naturwissenschaftlichen Vereins in Hamburg. 29: 187–220.
- ^ Engeser, Theo (1997). "Fossil Nautiloidea Page". Archived from the original on 2006-09-25.
- S2CID 85975284.
- ^ Hogan, C. Michael (22 December 2007). "Knossos fieldnotes". The Modern Antiquarian.
- ISBN 978-0-19-988773-6.
- ^ "Caroli Linnaei Systema naturae sistens regna tria naturae". google.com.
- ^ Smedley, Edward; Rose, Hugh James; Rose, Henry John (1845). Encyclopaedia Metropolitana, Or, Universal Dictionary of Knowledge: Comprising the Twofold Advantage of a Philosophical and an Alphabetical Arrangement, with Appropriate Engravings. B. Fellowes. pp. 255–.
- ^ Dixon, Roland Burrage (1916). Oceanic. The Mythology of All Races. Vol. 9. Marshall Jones Company. pp. 2–.
- ^ Batchelor, John (1901). The Ainu and Their Folklore. London: The Religious Tract Society.
- ^ Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica (11th ed.). Cambridge University Press.
- .
- ISBN 978-951-8965-54-4.
- ISBN 978-90-74822-66-4.
- )
- ^ Smith, S. (26 February 2010). "Why Mark Zuckerberg Octopus Cartoon Evokes 'Nazi Propaganda,' German Paper Apologizes". iMediaEthics. Retrieved 31 May 2017.
Further reading
- Barskov, I. S.; Boiko, M. S.; Konovalova, V. A.; Leonova, T. B.; Nikolaeva, S. V. (2008). "Cephalopods in the marine ecosystems of the Paleozoic". Paleontological Journal. 42 (11): 1167–1284. S2CID 83608661. A comprehensive overview of Paleozoic cephalopods.
- Campbell, Neil A.; Reece, Jane B.; Mitchell, Lawrence G. (1999). Biology, fifth edition. Menlo Park, California: Addison Wesley Longman, Inc. ISBN 978-0-8053-6566-5.
- Felley, J., Vecchione, M., Roper, C. F. E., Sweeney, M. & Christensen, T., 2001–2003: Current Classification of Recent Cephalopoda. National Museum of Natural History: Department of Systematic Biology: Invertebrate Zoology: Cephalopods
- Hanlon, Roger; Vecchione, Mike; Allcock, Louise (2018). Octopus, Squid, and Cuttlefish: A Visual, Scientific Guide to the Oceans' Most Advanced Invertebrates. University of Chicago Press. ISBN 978-0226459561.
- N. Joan Abbott, Roddy Williamson, Linda Maddock. Cephalopod Neurobiology. Oxford University Press, 1995. ISBN 0-19-854790-0
- Marion Nixon & ISBN 0-19-852761-6
- Hanlon, Roger T. & John B. Messenger. Cephalopod Behaviour. Cambridge University Press, 1996. ISBN 0-521-42083-0
- Martin Stevens & Sami Merilaita. Animal camouflage: mechanisms and function. Cambridge University Press, 2011. ISBN 0-521-19911-5
- Rodhouse, P. G.; Nigmatullin, Ch. M. (1996). "Role as Consumers". Philosophical Transactions of the Royal Society B: Biological Sciences. 351 (1343): 1003–1022. .
- Classification key to modern cephalopods: ftp://ftp.fao.org/docrep/fao/009/a0150e/a0150e03.pdf[permanent dead link]
External links
Cephalopoda.