Cephalosporin
| Cephalosporin | |
|---|---|
Penicillin binding proteins | |
| Clinical data | |
| Drugs.com | Drug Classes |
| External links | |
| MeSH | D002511 |
| Legal status | |
| In Wikidata | |
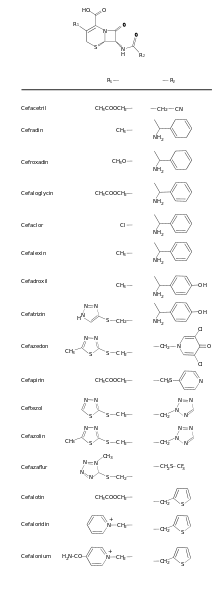
The cephalosporins (sg.
Together with cephamycins, they constitute a subgroup of β-lactam antibiotics called cephems. Cephalosporins were discovered in 1945, and first sold in 1964.[4]
Discovery
The
Structure
Cephalosporin contains a 6-membered dihydrothiazine ring. Substitutions at position 3 generally affect pharmacology; substitutions at position 7 affect antibacterial activity, but these cases are not always true.[6]
Medical uses
Cephalosporins can be indicated for the
The antibiotic may be used for patients who are allergic to penicillin due to the different
Side effects
Common
Allergic hypersensitivity
The commonly quoted figure of 10% of patients with allergic hypersensitivity to
The contraindication, however, should be viewed in the light of recent epidemiological work suggesting, for many second-generation (or later) cephalosporins, the cross-reactivity rate with penicillin is much lower, having no significantly increased risk of reactivity over the first generation based on the studies examined.[10][12] The British National Formulary previously issued blanket warnings of 10% cross-reactivity, but, since the September 2008 edition, suggests, in the absence of suitable alternatives, oral cefixime or cefuroxime and injectable cefotaxime, ceftazidime, and ceftriaxone can be used with caution, but the use of cefaclor, cefadroxil, cefalexin, and cefradine should be avoided.[13] A 2012 literature review similarly finds that the risk is negligible with third- and fourth-generation cephalosporins. The risk with first-generation cephalosporins having similar R1 sidechains was also found to be overestimated, with the real value closer to 1%.[14]
MTT side chain
Several cephalosporins are associated with
Mechanism of action
Cephalosporins are
Resistance
Classification
The cephalosporin nucleus can be modified to gain different properties. Cephalosporins are sometimes grouped into "generations" by their antimicrobial properties.[citation needed]
The first cephalosporins were designated first-generation cephalosporins, whereas, later, more extended-spectrum cephalosporins were classified as second-generation cephalosporins. Each newer generation has significantly greater Gram-negative antimicrobial properties than the preceding generation, in most cases with decreased activity against Gram-positive organisms. Fourth-generation cephalosporins, however, have true broad-spectrum activity.[23]
The classification of cephalosporins into "generations" is commonly practised, although the exact categorization is often imprecise. For example, the fourth generation of cephalosporins is not recognized as such in Japan.[citation needed] In Japan, cefaclor is classed as a first-generation cephalosporin, though in the United States it is a second-generation one; and cefbuperazone, cefminox, and cefotetan are classed as second-generation cephalosporins.
First generation
Cefalotin, cefazolin, cefalexin, cefapirin, cefradine, and cefadroxil are drugs belonging to this group.
Second generation
Cefoxitin, cefuroxime, cefaclor, cefprozil, and cefmetazole are classed as second-generation cephems.
Third generation
Ceftazidime, ceftriaxone, and cefotaxime are classed as third-generation cephalosporins. Flomoxef and latamoxef are in a new, related class called oxacephems.[24]
Fourth generation
Drugs included in this group are cefepime and cefpirome.
Further generations
Some state that cephalosporins can be divided into five or even six generations, although the usefulness of this organization system is of limited clinical relevance.[25]
Naming
Most first-generation cephalosporins were originally spelled "ceph-" in English-speaking countries. This continues to be the preferred spelling in the United States, Australia, and New Zealand, while European countries (including the United Kingdom) have adopted the
Activity
There exist bacteria which cannot be treated with cephalosporins of generations first through fourth:[26]
- Listeria spp.
- Atypicals (including Mycoplasma and Chlamydia)
- MRSA
- Enterococci
Fifth-generation cephalosporins (e.g. ceftaroline) are effective against MRSA, Listeria spp., and Enterococcus faecalis.[27][26]
Overview table
| Name | Description | |||||
|---|---|---|---|---|---|---|
| Common | Alternate name or spelling | Brand | ||||
| (#) = noncephalosporins similar to generation # | H, anaerobe
| |||||
| 1 | Cefalexin | cephalexin | Keflex | H V | Gram-positive: Activity against penicillinase-producing, methicillin-susceptible staphylococci and streptococci (though they are not the drugs of choice for such infections). No activity against methicillin-resistant staphylococci or enterococci.[citation needed]
Gram-negative: Activity against Proteus mirabilis, some Escherichia coli, and Klebsiella pneumoniae ("PEcK"), but have no activity against Bacteroides fragilis, Pseudomonas, Acinetobacter, Enterobacter, indole-positive Proteus, or Serratia.[citation needed] | |
| Cefadroxil | cefadroxyl | Duricef | H | |||
| Cefazolin | cephazolin | Ancef, Kefzol | H | |||
| Cefapirin | cephapirin | Cefadryl | V | |||
| Cefacetrile | cephacetrile | |||||
| Cefaloglycin | cephaloglycin | |||||
| Cefalonium | cephalonium | |||||
Cefaloridine |
cephaloradine | |||||
| Cefalotin | cephalothin | Keflin | ||||
| Cefatrizine | ||||||
| Cefazaflur | ||||||
| Cefazedone | ||||||
| Cefradine | cephradine | Velosef | ||||
| Cefroxadine | ||||||
| Ceftezole | ||||||
| 2 | Cefuroxime | Altacef, Zefu, Zinnat, Zinacef, Ceftin, Biofuroksym,[28] Xorimax | H | Gram-positive: Less than first-generation.[citation needed]
Gram-negative: Greater than first-generation: HEN | ||
| Cefprozil | cefproxil | Cefzil | H | |||
| Cefaclor | Ceclor, Distaclor, Keflor, Raniclor | H | ||||
| Cefonicid | Monocid | |||||
| Cefuzonam | ||||||
| Cefamandole | W | |||||
| (2) | Cefoxitin | Mefoxin | H | An | Cephamycins sometimes grouped with second-generation cephalosporins | |
| Cefotetan | Cefotan | H | An | |||
| Cefmetazole | Zefazone | An | ||||
| Cefminox | ||||||
| Cefbuperazone | ||||||
| Cefotiam | Pansporin | |||||
| Loracarbef | Lorabid | The carbacephem analog of cefaclor | ||||
| 3 | Cefdinir | Sefdin, Zinir, Omnicef, Kefnir | H | Gram-positive: Some members of this group (in particular, those available in an oral formulation, and those with antipseudomonal activity) have decreased activity against gram-positive organisms.
Activity against staphylococci and streptococci is less with the third-generation compounds than with the first- and second-generation compounds.[29] Gram-negative: Third-generation cephalosporins have a broad spectrum of activity and further increased activity against gram-negative organisms. They may be particularly useful in treating hospital-acquired infections, although increasing levels of extended-spectrum beta-lactamases are reducing the clinical utility of this class of antibiotics. They are also able to penetrate the central nervous system, making them useful against meningitis caused by pneumococci, meningococci, H. influenzae, and susceptible E. coli, Klebsiella, and penicillin-resistant N. gonorrhoeae. Since August 2012, the third-generation cephalosporin, ceftriaxone, is the only recommended treatment for gonorrhea in the United States (in addition to azithromycin or doxycycline for concurrent Chlamydia treatment). Cefixime is no longer recommended as a first-line treatment due to evidence of decreasing susceptibility.[30]
| ||
| Ceftriaxone | Rocephin | H | ||||
| Ceftazidime | Meezat, Fortum, Fortaz | H | P | |||
| Cefixime | Fixx, Zifi, Suprax | H | ||||
| Cefpodoxime | Vantin, PECEF, Simplicef | H V | ||||
| Ceftiofur | Naxcel, Excenel | H V | ||||
| Cefotaxime | Claforan | H | ||||
| Ceftizoxime | Cefizox | H | ||||
| Cefditoren | Zostom-O | H | ||||
| Ceftibuten | Cedax | H | ||||
| Cefovecin | Convenia | V | ||||
| Cefdaloxime | ||||||
| Cefcapene | ||||||
| Cefetamet | ||||||
| Cefmenoxime | ||||||
| Cefodizime | ||||||
| Cefpimizole | ||||||
| Cefteram | ||||||
| Ceftiolene | ||||||
| Cefoperazone | Cefobid | W[31] | P | |||
| (3) | Latamoxef | moxalactam | W[31] | An oxacephem sometimes grouped with third-generation cephalosporins | ||
| 4 | Cefepime | Maxipime | H | P | Gram-positive: They are extended-spectrum agents with similar activity against Gram-positive organisms as first-generation cephalosporins.[citation needed]
Gram-negative: Fourth-generation cephalosporins are zwitterions that can penetrate the outer membrane of Gram-negative bacteria.[32] They also have a greater resistance to β-lactamases than the third-generation cephalosporins. Many can cross the blood–brain barrier and are effective in meningitis. They are also used against Pseudomonas aeruginosa.[citation needed] Cefiderocol has been called a fourth-generation cephalosporin by only one source as of November 2019.[33] | |
| Cefiderocol | Fetroja | H | ||||
| Cefquinome | V | |||||
Cefclidine |
||||||
| Cefluprenam | ||||||
| Cefoselis | ||||||
| Cefozopran | ||||||
| Cefpirome | Cefrom | |||||
| (4) | Flomoxef | An oxacephem sometimes grouped with fourth-generation cephalosporins | ||||
| 5 | Ceftaroline |
H | MR | |||
Ceftolozane |
Zerbaxa | H | ||||
| Ceftobiprole | MR | |||||
| ? | Cefaloram | These cephems have progressed far enough to be named, but have not been assigned to a particular generation. Nitrocefin is a chromogenic cephalosporin substrate, and is used for detection of β-lactamases.[citation needed] | ||||
| Cefaparole | ||||||
| Cefcanel | ||||||
| Cefedrolor | ||||||
| Cefempidone | ||||||
| Cefetrizole | ||||||
| Cefivitril | ||||||
| Cefmatilen | ||||||
| Cefmepidium | ||||||
| Cefoxazole | ||||||
| Cefrotil | ||||||
| Cefsumide | ||||||
| Ceftioxide | ||||||
| Cefuracetime | ||||||
| Nitrocefin | ||||||
History
Cephalosporin compounds were first isolated from cultures of
References
- ^ "cephalosporin". Merriam-Webster.com Dictionary.
- OxfordDictionaries.com. Archived from the originalon 7 July 2012. Retrieved 20 January 2016.
- ^ "cephalosporin" at Dorland's Medical Dictionary
- ISBN 9780191039621.
- Wikidata Q29581637.
- ^ Prince, A. "Cephalosporins and vancomycin" (PDF). Columbia University. Archived from the original (PDF) on 19 August 2019. Retrieved 15 October 2022.
- ^ a b "Cephalosporins – Infectious Diseases". Merck Manuals Professional Edition. Retrieved 15 May 2019.
- PMID 31424895.
- PMID 1201975.
- ^ PMID 17991982.
- ^ Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006.[page needed]
- PMID 16451776.
- ISBN 978-0-85369-778-7.
- PMID 21742459.
- PMID 3593530.
- S2CID 30591177.
- ISBN 978-0-07-143763-9.
- PMID 24670024.
- PMID 31871085.
- PMID 5219821.
- ^ "Cephalosporin spectrum of resistance". Retrieved 1 July 2012.
- PMID 29712652.
- ^ "Cephalosporins – Infectious Diseases – Merck Manuals Professional Edition". Merck Manuals Professional Edition. Retrieved 14 June 2018.
- ISBN 978-3-642-75619-1.
- ^ "Case Based Pediatrics Chapter".
- ^ PMID 31855361, retrieved 2 June 2023
- PMID 21785568.
- ^ Jędrzejczyk, Tadeusz. "Internetowa Encyklopedia Leków". leki.med.pl. Archived from the original on 7 October 2007. Retrieved 3 March 2007.
- ISBN 978-0-19-512528-3.
- PMID 22874837.
- ^ )
- ISBN 978-0-7817-7815-2. Retrieved 8 September 2010.
- ^ "CHEBI:140376 – cefiderocol". ebi.ac.uk. EMBL-EBI. Retrieved 22 November 2019.
- PMID 18225983.
- PMID 17613382.
- PMID 20001879.
- PMID 17145788.
- PMID 18701284.
- PMID 21321149.
- PMID 23274659.
- S2CID 44694926.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - ISBN 978-1-4822-2973-8.[page needed]


