Cerium(IV) oxide

| |
| |
| Names | |
|---|---|
| IUPAC name
Cerium(IV) oxide
| |
| Other names
Ceric oxide,
Ceria, Cerium dioxide | |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.013.774 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CeO2 | |
| Molar mass | 172.115 g/mol |
| Appearance | white or pale yellow solid, slightly hygroscopic |
| Density | 7.215 g/cm3 |
| Melting point | 2,400 °C (4,350 °F; 2,670 K) |
| Boiling point | 3,500 °C (6,330 °F; 3,770 K) |
| insoluble | |
| +26.0·10−6 cm3/mol | |
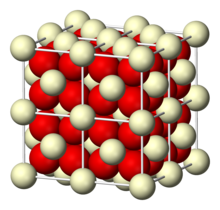
| Structure | |
| cubic crystal system, cF12 (fluorite)[1] | |
| Fm3m, #225 | |
a = 5.41 Å [2], b = 5.41 Å, c = 5.41 Å α = 90°, β = 90°, γ = 90°
| |
| Ce, 8, cubic O, 4, tetrahedral | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds
|
Cerium(III) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cerium(IV) oxide, also known as ceric oxide, ceric dioxide, ceria, cerium oxide or cerium dioxide, is an
Production
Cerium(IV) oxide is formed by the calcination of cerium oxalate or cerium hydroxide.
Cerium also forms cerium(III) oxide, Ce
2O
3, which is unstable and will oxidize to cerium(IV) oxide.[4]
Structure and defect behavior
Cerium oxide adopts the
has been shown to predict the equilibrium non-stoichiometry x over a wide range of oxygen partial pressures (103–10−4 Pa) and temperatures (1000–1900 °C).[7]
The non-stoichiometric form has a blue to black color, and exhibits both ionic and electronic conduction with ionic being the most significant at temperatures > 500 °C.[8]
The number of oxygen vacancies is frequently measured by using X-ray photoelectron spectroscopy to compare the ratio of Ce3+
to Ce4+
.
Defect chemistry
In the most stable fluorite phase of ceria, it exhibits several defects depending on partial pressure of oxygen or stress state of the material.[9][10][11][12]
The primary defects of concern are oxygen vacancies and small
The presence of oxygen vacancies at terminating ceria planes governs the energetics of ceria interactions with adsorbate molecules, and its wettability. Controlling such surface interactions is key to harnessing ceria in catalytic applications.[13]
Natural occurrence
Cerium(IV) oxide occurs naturally as the mineral
Applications
Cerium has two main applications, which are listed below.
The principal industrial application of ceria is for polishing, especially
In its other main application, CeO2 is used to decolorize glass. It functions by converting green-tinted ferrous impurities to nearly colorless ferric oxides.[3]
Other niche and emerging applications
Catalysis
CeO2 has attracted much attention in the area of
The interconvertibility of CeOx materials is the basis of the use of ceria for an oxidation catalyst. One small but illustrative use is its use in the walls of
Building on its distinct surface interactions, ceria finds further use as a sensor in catalytic converters in automotive applications, controlling the air-exhaust ratio to reduce NOx and carbon monoxide emissions.[25]
Energy & fuels
Due to the significant
Thermochemically, the cerium(IV) oxide–cerium(III) oxide cycle or CeO2/Ce2O3 cycle is a two-step water splitting process that has been used for hydrogen production.[28] Because it leverages the oxygen vacancies between systems, this allows ceria in water to form hydroxyl (OH) groups.[29] The hydroxyl groups can then be released as oxygen oxidizes, thus providing a source of clean energy.
Optics
Cerium oxide has found use in infrared filters and as a replacement for thorium dioxide in incandescent mantles[30]
Welding
Cerium oxide is used as an addition to tungsten electrodes for Gas Tungsten Arc Welding. It provides advantages over pure Tungsten electrodes such as reducing electrode consumption rate and easier arc starting & stability. Ceria electrodes were first introduced in the US market in 1987, and are useful in AC, DC Electrode Positive, and DC Electrode Negative.
Safety aspects
Cerium oxide nanoparticles (nanoceria) have been investigated for their antibacterial and antioxidant activity.[31][32][33][34]
Nanoceria is a prospective replacement of zinc oxide and titanium dioxide in sunscreens, as it has lower photocatalytic activity.[35]
See also
References
- ISBN 0-07-049439-8
- ^ E. A. Kümmerle and G. Heger, “The Structures of C-Ce2O3+δ, Ce7O12, and Ce11O20,” Journal of Solid State Chemistry, vol. 147, no. 2, pp. 485–500, 1999.
- ^ ISBN 978-3527306732..
- ^ "Standard Thermodynamic Properties of Chemical Substances" (PDF). Archived from the original (PDF) on October 29, 2013.
- ^ DFT study of Cerium Oxide Surfaces Applied surface science 2019 vol 478
- ^ Defects and Defect Processes in Nonmetallic Solids By William Hayes, A. M. Stoneham Courier Dover Publications, 2004.
- hdl:2262/76279.
- ISBN 978-3-527-31156-9.
- S2CID 95469920.
- .
- PMID 35528557.
- S2CID 204754299.
- S2CID 118895100.
- ^ a b "Cerianite-(Ce)". www.mindat.org. Retrieved 2020-11-12.
- ^ a b "List of Minerals". www.ima-mineralogy.org. 2011-03-21. Retrieved 2020-11-12.
- ^ Burke, Ernst (2008). "The use of suffixes in mineral names" (PDF). Elements. 4 (2): 96.
- S2CID 41920305.
- ^ "Properties of Common Abrasives (Boston Museum of Fine Arts)" (PDF).
- ^ "Ceric oxide - CAMEO". cameo.mfa.org.
- ^ Ruosi Peng; et a. (2018). "Size effect of Pt nanoparticles on the catalytic oxidation of toluene over Pt/CeO2 catalysts". Applied Catalysis B: Environmental. 220.
- PMID 27120134.
- PMID 23651311.
- .
- ISBN 978-0-08-037941-8.
- .
- ^ "Mixed conductors". Max Planck institute for solid state research. Retrieved 16 September 2016.
- .
- ^ "Hydrogen production from solar thermochemical water splitting cycles". SolarPACES. Archived from the original on August 30, 2009.
- ^ "New discoveries made on the role of Cerium Oxide in Hydrogen production". Ceric. 2018-07-01. Retrieved 2022-09-22.
- ^ "Cerium dioxide". DaNa. Archived from the original on 2013-03-02.
- PMID 29234605.
- PMID 20617106.
- PMID 29234605.
- PMID 22717232.
- PMID 20926307.


