Chagas disease
| Chagas disease | |
|---|---|
| Other names | American trypanosomiasis |
kissing bugs[1] | |
| Diagnostic method | Finding the parasite, its DNA, or antibodies in the blood[2] |
| Prevention | Eliminating kissing bugs and avoiding their bites[1] |
| Medication | Benznidazole, nifurtimox[1] |
| Frequency | 6.5 million (2019)[3] |
| Deaths | 9,490 (2019)[3] |
Chagas disease, also known as American trypanosomiasis, is a
T. cruzi is commonly spread to humans and other mammals by the kissing bug's bite wound and infected feces.
Prevention focuses on eliminating kissing bugs and avoiding their bites.
It is estimated that 6.5 million people, mostly in Mexico, Central America and South America, have Chagas disease as of 2019,[1][3] resulting in approximately 9,490 annual deaths.[3] Most people with the disease are poor,[12] and most do not realize they are infected.[13] Large-scale population migrations have carried Chagas disease to new regions, which include the United States and many European countries.[1] The disease affects more than 150 types of animals.[14]
The disease was first described in 1909 by Brazilian physician
Signs and symptoms

Chagas disease occurs in two stages: an acute stage, which develops one to two weeks after the insect bite, and a chronic stage, which develops over many years.[2][4][16] The acute stage is often symptom-free.[2] When present, the symptoms are typically minor and not specific to any particular disease.[4] Signs and symptoms include fever, malaise, headache, and enlargement of the liver, spleen, and lymph nodes.[1][2][4] Sometimes, people develop a swollen nodule at the site of infection, which is called "Romaña's sign" if it is on the eyelid, or a "chagoma" if it is elsewhere on the skin.[4][17] In rare cases (less than 1–5%), infected individuals develop severe acute disease, which can involve inflammation of the heart muscle, fluid accumulation around the heart, and inflammation of the brain and surrounding tissues, and may be life-threatening. The acute phase typically lasts four to eight weeks and resolves without treatment.[2]
Unless treated with
The most common long-term manifestation is
Also common in chronic Chagas disease is damage to the digestive system, which affects 10–21% of people.
Signs and symptoms differ for people infected with T. cruzi through less common routes. People infected through ingestion of parasites tend to develop severe disease within three weeks of consumption, with symptoms including fever,
Cause
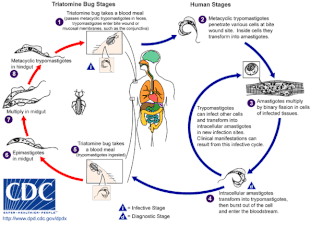
Chagas disease is caused by infection with the protozoan parasite T. cruzi, which is typically introduced into humans through the bite of triatomine bugs, also called "kissing bugs".[4] When the insect defecates at the bite site, motile T. cruzi forms called trypomastigotes enter the bloodstream and invade various host cells.[5] Inside a host cell, the parasite transforms into a replicative form called an amastigote, which undergoes several rounds of replication.[5] The replicated amastigotes transform back into trypomastigotes, which burst the host cell and are released into the bloodstream.[2] Trypomastigotes then disseminate throughout the body to various tissues, where they invade cells and replicate.[2] Over many years, cycles of parasite replication and immune response can severely damage these tissues, particularly the heart and digestive tract.[2]
Transmission

T. cruzi can be transmitted by various triatomine bugs in the genera
In addition to classical vector spread, Chagas disease can be transmitted through consumption of food or drink contaminated with triatomine insects or their feces.[22] Since heating or drying kills the parasites, drinks and especially fruit juices are the most frequent source of infection.[22] This oral route of transmission has been implicated in several outbreaks, where it led to unusually severe symptoms, likely due to infection with a higher parasite load than from the bite of a triatomine bug—[6][22]a single crushed triatomine in a food or beverage harboring T cruzi can contain about 600,000 metacyclic trypomastigotes, while triatomine fecal matter contains 3,000-4,000 per µL.[23]
T. cruzi can be transmitted independent of the triatomine bug during blood transfusion, following organ transplantation, or across the placenta during pregnancy.[2] Transfusion with the blood of an infected donor infects the recipient 10–25% of the time.[2] To prevent this, blood donations are screened for T. cruzi in many countries with endemic Chagas disease, as well as the United States.[6] Similarly, transplantation of solid organs from an infected donor can transmit T. cruzi to the recipient.[2] This is especially true for heart transplant, which transmits T. cruzi 75–100% of the time, and less so for transplantation of the liver (0–29%) or a kidney (0–19%).[2] An infected mother can pass T. cruzi to her child through the placenta; this occurs in up to 15% of births by infected mothers.[24] As of 2019, 22.5% of new infections occurred through congenital transmission.[25]
Pathophysiology

In the acute phase of the disease, signs and symptoms are caused directly by the replication of T. cruzi and the
During chronic Chagas disease, long-term organ damage develops over years due to continued replication of the parasite and damage from the immune system. Early in the course of the disease, T. cruzi is found frequently in the striated muscle fibers of the heart.[26] As disease progresses, the heart becomes generally enlarged, with substantial regions of cardiac muscle fiber replaced by scar tissue and fat.[26] Areas of active inflammation are scattered throughout the heart, with each housing inflammatory immune cells, typically macrophages and T cells.[26] Late in the disease, parasites are rarely detected in the heart, and may be present at only very low levels.[26]
In the heart, colon, and esophagus, chronic disease leads to a massive loss of nerve endings.[18] In the heart, this may contribute to arrythmias and other cardiac dysfunction.[18] In the colon and esophagus, loss of nervous system control is the major driver of organ dysfunction.[18] Loss of nerves impairs the movement of food through the digestive tract, which can lead to blockage of the esophagus or colon and restriction of their blood supply.[18]
Diagnosis

The presence of T. cruzi in the blood is diagnostic of Chagas disease. During the acute phase of infection, it can be detected by
Alternatively, T. cruzi DNA can be detected by polymerase chain reaction (PCR). In acute and congenital Chagas disease, PCR is more sensitive than microscopy,[25] and it is more reliable than antibody-based tests for the diagnosis of congenital disease because it is not affected by transfer of antibodies against T. cruzi from a mother to her baby (passive immunity).[28] PCR is also used to monitor T. cruzi levels in organ transplant recipients and immunosuppressed people, which allows infection or reactivation to be detected at an early stage.[2][4][25]
In chronic Chagas disease, the concentration of parasites in the blood is too low to be reliably detected by microscopy or PCR,[2] so the diagnosis is usually made using serological tests, which detect immunoglobulin G antibodies against T. cruzi in the blood.[6] Two positive serology results, using different test methods, are required to confirm the diagnosis.[4] If the test results are inconclusive, additional testing methods such as Western blot can be used.[2]
Various rapid diagnostic tests for Chagas disease are available. These tests are easily transported and can be performed by people without special training.[29] They are useful for screening large numbers of people and testing people who cannot access healthcare facilities, but their sensitivity is relatively low,[2] and it is recommended that a second method is used to confirm a positive result.[29][30]
T. cruzi parasites can be grown from blood samples by blood culture, xenodiagnosis, or by inoculating animals with the person's blood. In the blood culture method, the person's red blood cells are separated from the plasma and added to a specialized growth medium to encourage multiplication of the parasite. It can take up to six months to obtain the result. Xenodiagnosis involves feeding the blood to triatomine insects, then examining their feces for the parasite 30 to 60 days later.[29] These methods are not routinely used, as they are slow and have low sensitivity.[31][29]
Prevention
Efforts to prevent Chagas disease have largely focused on
Blood transfusion was formerly the second-most common mode of transmission for Chagas disease.[33] T. cruzi can survive in refrigerated stored blood, and can survive freezing and thawing, allowing it to persist in whole blood, packed red blood cells, granulocytes, cryoprecipitate, and platelets.[33] The development and implementation of blood bank screening tests has dramatically reduced the risk of infection during blood transfusion.[33] Nearly all blood donations in Latin American countries undergo Chagas screening.[33] Widespread screening is also common in non-endemic nations with significant populations of immigrants from endemic areas, including the United Kingdom (implemented in 1999), Spain (2005), the United States (2007), France and Sweden (2009), Switzerland (2012), and Belgium (2013).[34] Serological tests, typically ELISAs, are used to detect antibodies against T. cruzi proteins in donor blood.[33]
Other modes of transmission have been targeted by Chagas disease prevention programs. Treating T. cruzi-infected mothers during pregnancy reduces the risk of congenital transmission of the infection.[24] To this end, many countries in Latin America have implemented routine screening of pregnant women and infants for T. cruzi infection, and the World Health Organization recommends screening all children born to infected mothers to prevent congenital infection from developing into chronic disease.[1][35] Similarly to blood transfusions, many countries with endemic Chagas disease screen organs for transplantation with serological tests.[2]
There is no vaccine against Chagas disease.[8] Several experimental vaccines have been tested in animals infected with T. cruzi and were able to reduce parasite numbers in the blood and heart,[36] but no vaccine candidates had undergone clinical trials in humans as of 2016.[37]
Management

Chagas disease is managed using
Benznidazole is usually considered the
Complications
In the chronic stage, treatment involves managing the clinical manifestations of the disease. The treatment of Chagas cardiomyopathy is similar to that of other forms of heart disease.
Mild gastrointestinal disease may be treated symptomatically, such as by using
Epidemiology

In 2019, an estimated 6.5 million people worldwide had Chagas disease, with approximately 173,000 new infections and 9,490 deaths each year.
The endemic area of Chagas disease stretches from the southern United States to northern Chile and Argentina, with Bolivia (6.1%), Argentina (3.6%), and Paraguay (2.1%) exhibiting the highest prevalence of the disease.[2] Within continental Latin America, Chagas disease is endemic to 21 countries: Argentina, Belize, Bolivia, Brazil, Chile, Colombia, Costa Rica, Ecuador, El Salvador, French Guiana, Guatemala, Guyana, Honduras, Mexico, Nicaragua, Panama, Paraguay, Peru, Suriname, Uruguay, and Venezuela.[1][2] In endemic areas, due largely to vector control efforts and screening of blood donations, annual infections and deaths have fallen by 67% and more than 73% respectively from their peaks in the 1980s to 2010.[2][44] Transmission by insect vector and blood transfusion has been completely interrupted in Uruguay (1997), Chile (1999), and Brazil (2006),[44] and in Argentina, vectorial transmission had been interrupted in 13 of the 19 endemic provinces as of 2001.[45] During Venezuela's humanitarian crisis, vectorial transmission has begun occurring in areas where it had previously been interrupted, and Chagas disease seroprevalence rates have increased.[46] Transmission rates have also risen in the Gran Chaco region due to insecticide resistance and in the Amazon basin due to oral transmission.[2]
While the rate of vector-transmitted Chagas disease has declined throughout most of Latin America, the rate of orally transmitted disease has risen, possibly due to increasing urbanization and deforestation bringing people into closer contact with triatomines and altering the distribution of triatomine species.[22][47][48] Orally transmitted Chagas disease is of particular concern in Venezuela, where 16 outbreaks have been recorded between 2007 and 2018.[46]
Chagas exists in two different ecological zones. In the
Non-endemic countries
Though Chagas is traditionally considered a disease of rural Latin America, international migration has dispersed those with the disease to numerous non-endemic countries, primarily in North America and Europe.[1][34] As of 2020, approximately 300,000 infected people are living in the United States,[52] and in 2018 it was estimated that 30,000 to 40,000 people in the United States had Chagas cardiomyopathy.[18] The vast majority of cases in the United States occur in immigrants from Latin America,[18][25] but local transmission is possible. Eleven triatomine species are native to the United States, and some southern states have persistent cycles of disease transmission between insect vectors and animal reservoirs,[2][25] which include woodrats, possums, raccoons, armadillos and skunks.[53] However, locally acquired infection is very rare: only 28 cases were documented from 1955 to 2015.[2][52] As of 2013, the cost of treatment in the United States was estimated to be US$900 million annually (global cost $7 billion), which included hospitalization and medical devices such as pacemakers.[43]
Chagas disease affected approximately 68,000 to 123,000 people in Europe as of 2019.[54] Spain, which has a high rate of immigration from Latin America, has the highest prevalence of the disease. It is estimated that 50,000 to 70,000 people in Spain are living with Chagas disease, accounting for the majority of European cases.[55] The prevalence varies widely within European countries due to differing immigration patterns.[54] Italy has the second highest prevalence, followed by the Netherlands, the United Kingdom, and Germany.[55]
History

T. cruzi likely circulated in South American mammals long before the arrival of humans on the continent.
The formal description of Chagas disease was made by
In the 1930s, Salvador Mazza rekindled Chagas disease research, describing over a thousand cases in Argentina's Chaco Province.[56] In Argentina, the disease is known as mal de Chagas-Mazza in his honor.[61] Serological tests for Chagas disease were introduced in the 1940s, demonstrating that infection with T. cruzi was widespread across Latin America.[56] This, combined with successes eliminating the malaria vector through insecticide use, spurred the creation of public health campaigns focused on treating houses with insecticides to eradicate triatomine bugs.[32][56] The 1950s saw the discovery that treating blood with crystal violet could eradicate the parasite, leading to its widespread use in transfusion screening programs in Latin America.[56] Large-scale control programs began to take form in the 1960s, first in São Paulo, then various locations in Argentina, then national-level programs across Latin America.[62] These programs received a major boost in the 1980s with the introduction of pyrethroid insecticides, which did not leave stains or odors after application and were longer-lasting and more cost-effective.[56][62] Regional bodies dedicated to controlling Chagas disease arose through support of the Pan American Health Organization, with the Initiative of the Southern Cone for the Elimination of Chagas Diseases launching in 1991, followed by the Initiative of the Andean countries (1997), Initiative of the Central American countries (1997), and the Initiative of the Amazon countries (2004).[32]
Research
Treatments
A number of experimental vaccines have been tested in animals. In addition to
Diagnostic tests
As of 2018, standard diagnostic tests for Chagas disease were limited in their ability to measure the effectiveness of antiparasitic treatment, as serological tests may remain positive for years after T. cruzi is eliminated from the body, and PCR may give false-negative results when the parasite concentration in the blood is low. Several potential biomarkers of treatment response are under investigation, such as immunoassays against specific T. cruzi antigens, flow cytometry testing to detect antibodies against different life stages of T. cruzi, and markers of physiological changes caused by the parasite, such as alterations in coagulation and lipid metabolism.[18]
Another research area is the use of biomarkers to predict the progression of chronic disease. Serum levels of
T. cruzi shed acute-phase antigen (SAPA), which can be detected in blood using ELISA or Western blot,[24] has been used as an indicator of early acute and congenital infection.[65] An assay for T. cruzi antigens in urine has been developed to diagnose congenital disease.[24]
See also
- Drugs for Neglected Diseases Initiative
- Chagas: Time to Treat campaign
- Association for the Promotion of Independent Disease Control in Developing Countries
References
- ^ a b c d e f g h i j k l m n o p q "Chagas disease (American trypanosomiasis)". World Health Organization. 17 April 2019. Archived from the original on 20 January 2020. Retrieved 9 March 2020.
- ^ S2CID 4514617.
- ^ a b c d e "Global Health Metrics: Chagas disease—Level 3 cause" (PDF). The Lancet. 2020. Archived (PDF) from the original on 18 March 2022.
- ^ PMID 26222561.
- ^ a b c "DPDx – Trypanosomiasis, American. Fact Sheet". Centers for Disease Control (CDC). 30 April 2019. Archived from the original on 5 September 2019. Retrieved 10 March 2020.
- ^ PMID 31006555.
- ^ "Prevention of Chagas disease". World Health Organization. Archived from the original on 11 February 2018. Retrieved 24 February 2018.
- ^ PMID 35239945.
- PMID 37576826.
- ^ PMID 31513763.
- ^ S2CID 207815975.
- S2CID 34941921.
- ISBN 9781402062421.
- S2CID 5068417.
- ^ "Neglected Tropical Diseases". cdc.gov. 6 June 2011. Archived from the original on 4 December 2014. Retrieved 28 November 2014.
- ^ a b c d e f g h i j k l Despommier DD, Griffin DO, Gwadz RW, Hotez PJ, Knirsch CA (2019). "American Trypanosomiasis". Parasitic Diseases (7 ed.). New York: Parasites Without Borders. pp. 71–84. Retrieved 26 March 2020.
- ^ "Chagas Disease – Detailed Fact Sheet". www.cdc.gov. CDC-Centers for Disease Control and Prevention. Archived from the original on 16 August 2019. Retrieved 15 April 2020.
- ^ PMID 30354432.
- S2CID 73446292.
- ^ S2CID 203357705.
- ISBN 9780851990347.
- ^ PMID 27253136.
- ^ "Oral Transmission of Chagas Disease Has Severe Effects (with section 'Seven Things to Know About Orally Transmitted Chagas Disease')". Medscape. 1 March 2024.
- ^ (PDF) from the original on 5 March 2020.
- ^ PMID 31776135.
- ^ PMID 30355152.
- ISBN 978-1-118-81733-9.
- S2CID 3513900.
- ^ S2CID 203376369.
- PMID 31150377.
- ^ a b Kirchhoff OV (26 April 2019). "Chagas disease (American Trypanosomiasis)". eMedicine. Archived from the original on 12 March 2020. Retrieved 8 April 2020.
- ^ PMID 26003952.
- ^ PMID 26513769.
- ^ PMID 31312626.
- PMID 24626257.
- PMID 30703092.
- PMID 27026146.
- ^ PMID 31862616.
- ^ WHO. (13 April 2022). "Chagas disease (also known as American trypanosomiasis". Fact sheets.
- ^ S2CID 203457516.
- PMID 26715535.
- ^ World Health Organization. "Disease burden and mortality estimates". Archived from the original on 10 February 2020. Retrieved 27 April 2020.
- ^ PMID 23395248.
- ^ .
- ^ "The Southern Cone Initiative: an update". Special Programme for Research and Training in Tropical Diseases (TDR) (Press release). WHO. 2004. Archived from the original on 22 September 2009. Retrieved 29 August 2008.
- ^ S2CID 73475841.
- S2CID 239445211.
- PMID 28662038.
- S2CID 29869370.
- ^ a b c d e f Wilkowsky SE (9 March 2022). "Trypanosomiasis in Animals - Circulatory System". Merck Veterinary Manual. Archived from the original on 28 March 2022. Retrieved 28 March 2022.
- ^ ISBN 978-0-123-84876-5.
- ^ PMID 32078594.
- PMID 24808250.
- ^ S2CID 203406723.
- ^ PMID 32127121.
- ^ PMID 25011546.
- ^ S2CID 36106351.
- ^ Chagas C (1909). "Neue Trypanosomen". Vorläufige Mitteilung Arch Schiff Tropenhyg. 13: 120–2.
- ISSN 0074-0276. (in Portuguese with German full translation as "Ueber eine neue Trypanosomiasis des Menschen.")
- PMID 19185367.
- ^ "Enfermedad de Chagas – Mazza" (in Spanish). Asociación Lucha Contra el Mal de Chagas. Archived from the original on 16 September 2011. Retrieved 15 September 2011.
- ^ PMID 26407516.
- S2CID 57772417.
- PMID 31351074.
- ^ PMID 28325368.
External links
- Chagas disease at Curlie
- Chagas information at the U.S. Centers for Disease Control
- Chagas information from the Drugs for Neglected Diseases initiative
- Chagas disease information for travellers from the International Association for Medical Assistance to Travellers
