Fusion protein

Fusion proteins or chimeric (kī-ˈmir-ik) proteins (literally, made of parts from different sources) are proteins created through the joining of two or more
Functions
Some fusion proteins combine whole peptides and therefore contain all
Many whole gene fusions are fully functional and can still act to replace the original peptides. Some, however, experience interactions between the two proteins that can modify their functions. Beyond these effects, some gene fusions may cause regulatory changes that alter when and where these genes act. For partial gene fusions, the shuffling of different active sites and binding domains have the potential to result in new proteins with novel functions.
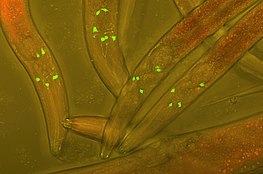
Fluorescent protein tags
The fusion of
Chimeric protein drugs
The purpose of creating fusion proteins in drug development is to impart properties from each of the "parent" proteins to the resulting chimeric protein. Several chimeric protein drugs are currently available for medical use.
Many chimeric protein drugs are
In addition to chimeric and humanized antibodies, there are other pharmaceutical purposes for the creation of chimeric constructs.
- IL-1 receptors to treat cryopyrin-associated periodic syndromes (CAPS).[2]
- psoriasis vulgaris.[2]
- immune thrombocytopenia.[2]
- Abatacept/Belatacept: Interferes with T-cell co-stimulation to treat autoimmune disorders like rheumatoid arthritis, psoriatic arthritis, and psoriasis.[2]
- Denileukin-diftitox: Treats cutaneous lymphoma.[2]
Recombinant technology

A recombinant fusion protein is a
If the two entities are proteins, often linker (or "spacer") peptides are also added, which make it more likely that the proteins fold independently and behave as expected. Especially in the case where the linkers enable
Recombinant functionality
Novel recombinant technologies have made it possible to improve fusion protein design for use in fields as diverse as biodetection, paper and food industries, and biopharmaceuticals. Recent improvements have involved the fusion of single peptides or protein fragments to regions of existing proteins, such as N and C termini, and are known to increase the following properties:[4]
- tertiary and quaternary structure of the target protein.[4]
- Molecular chaperones that are able to aid in protein folding may be added, thereby better segregating hydrophobic and hydrophilic interactions in the solute to increase protein solubility.[4]
- Thermostability: Singular peptides or protein fragments are typically added to reduce flexibility of either the N or C terminus of the target protein, which reinforces thermostability and stabilizes pH range.[4]
- Enzyme activity: Fusion that involves the introduction of hydrogen bonds may be used to expand overall enzyme activity.[4]
- Expression levels: Addition of numerous fusion fragments, such as maltose binding protein (MBP) or small ubiquitin-like molecule (SUMO), serve to enhance enzyme expression and secretion of the target protein.[4]
- Immobilization: PHA synthase, an enzyme that allows for the immobilization of proteins of interest, is an important fusion tag in industrial research.[4]
- Crystal quality: Crystal quality can be improved by adding covalent links between proteins, aiding in structure determination techniques.[4]
Recombinant protein design
The earliest applications of recombinant protein design can be documented in the use of single peptide tags for purification of proteins in affinity chromatography. Since then, a variety of fusion protein design techniques have been developed for applications as diverse as fluorescent protein tags to recombinant fusion protein drugs. Three commonly used design techniques include tandem fusion, domain insertion, and post-translational conjugation.[5]
Tandem fusion
The proteins of interest are simply connected end-to-end via fusion of N or C termini between the proteins. This provides a flexible bridge structure allowing enough space between fusion partners to ensure proper folding. However, the N or C termini of the peptide are often crucial components in obtaining the desired folding pattern for the recombinant protein, making simple end-to-end conjoining of domains ineffective in this case. For this reason, a protein linker is often needed to maintain the functionality of the protein domains of interest.[5]
Domain insertion
This technique involves the fusion of consecutive protein domains by encoding desired structures into a single polypeptide chain, but sometimes may require insertion of a domain within another domain. This technique is typically regarding as more difficult to carry out than tandem fusion, due to difficulty finding an appropriate ligation site in the gene of interest.[5]
Post-translational conjugation
This technique fuses protein domains following ribosomal translation of the proteins of interest, in contrast to genetic fusion prior to translation used in other recombinant technologies.[5]
Protein linkers

Protein linkers aid fusion protein design by providing appropriate spacing between domains, supporting correct protein folding in the case that N or C termini interactions are crucial to folding. Commonly, protein linkers permit important domain interactions, reinforce stability, and reduce steric hindrance, making them preferred for use in fusion protein design even when N and C termini can be fused. Three major types of linkers are flexible, rigid, and in vivo cleavable.[5][6]
- Flexible linkers may consist of many small glycine residues, giving them the ability curl into a dynamic, adaptable shape.[6]
- Rigid linkers may be formed of large, cyclic proline residues, which can be helpful when highly specific spacing between domains must be maintained.[6]
- In vivo cleavable linkers are unique in that they are designed to allow the release of one or more fused domains under certain reaction conditions, such as a specific pH gradient, or when coming in contact with another biomolecule in the cell.[6]
Natural occurrence
Naturally occurring fusion genes are most commonly created when a
Examples include:
- Gag-onc fusion protein
- Bcr-abl fusion protein
- Tpr-met fusion protein
Antibodies are fusion proteins produced by V(D)J recombination.
There are also rare examples of naturally occurring polypeptides that appear to be a fusion of two clearly defined modules, in which each module displays its characteristic activity or function, independent of the other. Two major examples are: double PP2C chimera in The evolutionary origin of such chimera remains unclear.
See also
References
External links
- Mutant+Chimeric+Proteins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- ChiPPI: The Server Protein–Protein Interaction of Chimeric Proteins.
