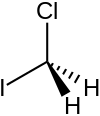
Chloroiodomethane
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Chloro(iodo)methane | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 1730802 | |
| ChemSpider | |
ECHA InfoCard
|
100.008.915 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CH2ClI | |
| Molar mass | 176.38 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 2.422 g mL−1 |
| Boiling point | 108 to 109 °C (226 to 228 °F; 381 to 382 K) |
Henry's law
constant (kH) |
8.9 μmol Pa−1 kg−1 |
Refractive index (nD)
|
1.582 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related alkanes
|
|
Related compounds
|
2-Chloroethanol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chloroiodomethane is the halomethane with the formula is CH
2ClI. It is a colorless liquid of use in organic synthesis.[1] Together with other iodomethanes, chloroiodomethane is produced by some microorganisms.[2]
Applications
Chloroiodomethane is used in
epoxidation, ring opening and addition to terminal alkenes. It is a precursor to agent Ph3P=CHCl that can add a chloromethylene group (=CHCl).[1] It reacts with organolithium compounds to give chloromethyl lithium (ClCH2Li).[3]
Crystallography
It crystallizes
lattice constants: a = 6.383, b = 6.706, c = 8.867 (.10−1 nm).[4]
References
- ^ ISBN 978-0471936237.
- PMID 14680698.
- ISBN 0471936235.
- .
External links

