Cholangiocarcinoma
| Cholangiocarcinoma | |
|---|---|
| Other names | Bile duct cancer, cancer of the bile duct stenting procedures, liver transplantation[1] |
| Prognosis | Generally poor[5] |
| Frequency | 1–2 people per 100,000 per year (Western world)[6] |
Cholangiocarcinoma, also known as bile duct cancer, is a type of
Risk factors for cholangiocarcinoma include
Cholangiocarcinoma is typically incurable at diagnosis which is why early detection is ideal.
Cholangiocarcinoma is rare in the Western world, with estimates of it occurring in 0.5–2 people per 100,000 per year.[1][6] Rates are higher in Southeast Asia where liver flukes are common.[5] Rates in parts of Thailand are 60 per 100,000 per year.[5] It typically occurs in people in their 70s; however, in those with primary sclerosing cholangitis it often occurs in the 40s.[3] Rates of cholangiocarcinoma within the liver in the Western world have increased.[6]
Signs and symptoms
The most common physical indications of cholangiocarcinoma are abnormal
Risk factors

Although most people present without any known risk factors evident, a number of
Certain parasitic liver diseases may be risk factors as well. Colonization with the liver flukes Opisthorchis viverrini (found in Thailand, Laos PDR, and Vietnam)[17][18][19] or Clonorchis sinensis (found in China, Taiwan, eastern Russia, Korea, and Vietnam)[20][21] has been associated with the development of cholangiocarcinoma. Control programs (Integrated Opisthorchiasis Control Program) aimed at discouraging the consumption of raw and undercooked food have been successful at reducing the incidence of cholangiocarcinoma in some countries.[22] People with chronic liver disease, whether in the form of viral hepatitis (e.g. hepatitis B or hepatitis C),[23][24][25] alcoholic liver disease, or cirrhosis of the liver due to other causes, are at significantly increased risk of cholangiocarcinoma.[26][27] HIV infection was also identified in one study as a potential risk factor for cholangiocarcinoma, although it was unclear whether HIV itself or other correlated and confounding factors (e.g. hepatitis C infection) were responsible for the association.[26]
Infection with the bacteria Helicobacter bilis and Helicobacter hepaticus species can cause biliary cancer.[28]
Pathophysiology

Cholangiocarcinoma can affect any area of the bile ducts, either within or outside the liver. Tumors occurring in the bile ducts within the liver are referred to as intrahepatic, those occurring in the ducts outside the liver are extrahepatic, and tumors occurring at the site where the bile ducts exit the liver may be referred to as perihilar. A cholangiocarcinoma occurring at the junction where the left and right hepatic ducts meet to form the common hepatic duct may be referred to eponymously as a Klatskin tumor.[39]
Although cholangiocarcinoma is known to have the histological and molecular features of an
Diagnosis
Blood tests
There are no specific
Abdominal imaging
Imaging of the biliary tree

While abdominal imaging can be useful in the diagnosis of cholangiocarcinoma, direct imaging of the
Surgery
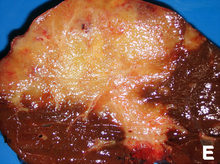
Surgical exploration may be necessary to obtain a suitable biopsy and to accurately stage a person with cholangiocarcinoma. Laparoscopy can be used for staging purposes and may avoid the need for a more invasive surgical procedure, such as laparotomy, in some people.[62][63]
Pathology
Histologically, cholangiocarcinomas are classically well to moderately differentiated adenocarcinomas. Immunohistochemistry is useful in the diagnosis and may be used to help differentiate a cholangiocarcinoma from hepatocellular carcinoma and metastasis of other gastrointestinal tumors.[65] Cytological scrapings are often nondiagnostic,[66] as these tumors typically have a desmoplastic stroma and, therefore, do not release diagnostic tumor cells with scrapings.
Staging
Although there are at least three staging systems for cholangiocarcinoma (e.g. those of Bismuth, Blumgart, and the American Joint Committee on Cancer), none have been shown to be useful in predicting survival.[67] The most important staging issue is whether the tumor can be surgically removed, or whether it is too advanced for surgical treatment to be successful. Often, this determination can only be made at the time of surgery.[12]
General guidelines for operability include:[68][69]
- Absence of lymph node or liver metastases
- Absence of involvement of the portal vein
- Absence of direct invasion of adjacent organs
- Absence of widespread metastatic disease
Treatment
Cholangiocarcinoma is considered to be an incurable and rapidly lethal disease unless all the tumors can be fully resected (cut out surgically). Since the operability of the tumor can only be assessed during surgery in most cases,[70] a majority of people undergo exploratory surgery unless there is already a clear indication that the tumor is inoperable.[12] However, the Mayo Clinic has reported significant success treating early bile duct cancer with liver transplantation using a protocolized approach and strict selection criteria.[71]
Adjuvant therapy followed by liver transplantation may have a role in treatment of certain unresectable cases.[72] Locoregional therapies including transarterial chemoembolization (TACE), transarterial radioembolization (TARE) and ablation therapies have a role in intrahepatic variants of cholangiocarcinoma to provide palliation or potential cure in people who are not surgical candidates.[73]
Adjuvant chemotherapy and radiation therapy
If the tumor can be removed surgically, people may receive adjuvant chemotherapy or radiation therapy after the operation to improve the chances of cure. If the tissue margins are negative (i.e. the tumor has been totally excised), adjuvant therapy is of uncertain benefit. Both positive[74][75] and negative[11][76][77] results have been reported with adjuvant radiation therapy in this setting, and no prospective randomized controlled trials have been conducted as of March 2007. Adjuvant chemotherapy appears to be ineffective in people with completely resected tumors.[78][79] The role of combined chemoradiotherapy in this setting is unclear. However, if the tumor tissue margins are positive, indicating that the tumor was not completely removed via surgery, then adjuvant therapy with radiation and possibly chemotherapy is generally recommended based on the available data.[80] [81]
Treatment of advanced disease
The majority of cases of cholangiocarcinoma present as inoperable (unresectable) disease
Infigratinib (Truseltiq) is a tyrosine kinase inhibitor of fibroblast growth factor receptor (FGFR) that was approved for medical use in the United States in May 2021.[91] It is indicated for the treatment of people with previously treated locally advanced or metastatic cholangiocarcinoma harboring an FGFR2 fusion or rearrangement.[91]
Pemigatinib (Pemazyre) is a kinase inhibitor of fibroblast growth factor receptor 2 (FGFR2) that was approved for medical use in the United States in April 2020.[92] It is indicated for the treatment of adults with previously treated, unresectable locally advanced or metastatic cholangiocarcinoma with a fibroblast growth factor receptor 2 (FGFR2) fusion or other rearrangement as detected by an FDA-approved test.
Ivodesinib (Tibsovo) is a small molecule inhibitor of isocitrate dehydrogenase 1. The FDA approved ivosidenib in August 2021 for adults with previously treated, locally advanced or metastatic cholangiocarcinoma with an isocitrate dehydrogenase-1 (IDH1) mutation as detected by an FDA-approved test.[93]
Durvalumab, (Imfinzi) is an immune checkpoint inhibitor that blocks the PD-L1 protein on the surface of immune cells, thereby allowing the immune system to recognize and attack tumor cells. In Phase III clinical trials, durvalumab, in combination with standard-of-care chemotherapy, demonstrated a statistically significant and clinically meaningful improvement in overall survival and progression-free survival versus chemotherapy alone as a 1st-line treatment for patients with advanced biliary tract cancer.[94]
Futibatinib (Lytgobi) was approved for medical use in the United States in September 2022.[95]
Prognosis
Surgical resection offers the only potential chance of cure in cholangiocarcinoma. For non-resectable cases, the five-year survival rate is 0% where the disease is inoperable because distal lymph nodes show metastases,[96] and less than 5% in general.[97] Overall mean duration of survival is less than 6 months in people with metastatic disease.[98]
For surgical cases, the odds of cure vary depending on the tumor location and whether the tumor can be completely, or only partially, removed. Distal cholangiocarcinomas (those arising from the
The prognosis may be worse for people with primary sclerosing cholangitis who develop cholangiocarcinoma, likely because the cancer is not detected until it is advanced.[15][102] Some evidence suggests that outcomes may be improving with more aggressive surgical approaches and adjuvant therapy.[103]
Epidemiology
| Country | IC (men/women) | EC (men/women) |
|---|---|---|
| U.S.A. | 0.60/0.43 | 0.70/0.87 |
| Japan | 0.23/0.10 | 5.87/5.20 |
| Australia | 0.70/0.53 | 0.90/1.23 |
| England/Wales | 0.83/0.63 | 0.43/0.60 |
| Scotland | 1.17/1.00 | 0.60/0.73 |
| France | 0.27/0.20 | 1.20/1.37 |
| Italy | 0.13/0.13 | 2.10/2.60 |
Cholangiocarcinoma is a relatively rare form of cancer; each year, approximately 2,000 to 3,000 new cases are diagnosed in the United States, translating into an annual incidence of 1–2 cases per 100,000 people.[105] Autopsy series have reported a prevalence of 0.01% to 0.46%.[82][106] There is a higher prevalence of cholangiocarcinoma in Asia, which has been attributed to endemic chronic parasitic infestation. The incidence of cholangiocarcinoma increases with age, and the disease is slightly more common in men than in women (possibly due to the higher rate of primary sclerosing cholangitis, a major risk factor, in men).[47] The prevalence of cholangiocarcinoma in people with primary sclerosing cholangitis may be as high as 30%, based on autopsy studies.[15]
Multiple studies have documented a steady increase in the incidence of intrahepatic cholangiocarcinoma; increases have been seen in North America, Europe, Asia, and Australia.[107] The reasons for the increasing occurrence of cholangiocarcinoma are unclear; improved diagnostic methods may be partially responsible, but the prevalence of potential risk factors for cholangiocarcinoma, such as HIV infection, has also been increasing during this time frame.[26]

References
- ^ a b c d e f g h i j k "Bile Duct Cancer (Cholangiocarcinoma) Treatment". National Cancer Institute. 23 September 2020. Retrieved 29 May 2021.
- ^ a b "cholangiocarcinoma". National Cancer Institute. 2 February 2011. Retrieved 21 January 2019.
- ^ PMID 24581682.
- ^ a b c d "Bile Duct Cancer (Cholangiocarcinoma)". National Cancer Institute. 5 July 2018. Retrieved 21 January 2019.
- ^ ISBN 978-92-832-0443-5.

- ^ PMID 27249723.
- PMID 26607930.
- PMID 29769113.
- PMID 34888258.
- PMID 8393652.
- ^ PMID 8857851.
- ^ ISBN 978-1-4160-0245-1.
- PMID 10436847.
- ^ Epidemiologic studies which have addressed the incidence of cholangiocarcinoma in people with primary sclerosing cholangitis include the following:
- Bergquist A, Ekbom A, Olsson R, Kornfeldt D, Lööf L, Danielsson A, et al. (March 2002). "Hepatic and extrahepatic malignancies in primary sclerosing cholangitis". Journal of Hepatology. 36 (3): 321–7. PMID 11867174.
- Bergquist A, Glaumann H, Persson B, Broomé U (February 1998). "Risk factors and clinical presentation of hepatobiliary carcinoma in patients with primary sclerosing cholangitis: a case-control study". Hepatology. 27 (2): 311–6. PMID 9462625.
- Burak K, Angulo P, Pasha TM, Egan K, Petz J, Lindor KD (March 2004). "Incidence and risk factors for cholangiocarcinoma in primary sclerosing cholangitis". American Journal of Gastroenterology. 99 (3): 523–6. S2CID 8412954.
- Bergquist A, Ekbom A, Olsson R, Kornfeldt D, Lööf L, Danielsson A, et al. (March 2002). "Hepatic and extrahepatic malignancies in primary sclerosing cholangitis". Journal of Hepatology. 36 (3): 321–7.
- ^ PMID 1845927.
- PMID 30819129.
- S2CID 5606131.
- PMID 17622191.
- )
- S2CID 31590872.
- PMID 21741496.
- PMID 27890720.
- S2CID 22206944.
- PMID 15245596.
- PMID 11776153.
- ^ PMID 15765398.
- S2CID 72842845.
- PMID 20930075.
- PMID 7979612.
- PMID 6295196.
- S2CID 23468163.
- PMID 14770435.
- S2CID 25753564.
- S2CID 29475282.
- S2CID 12117363.
- S2CID 23531547.
- PMID 15619021.
- PMID 12437262.
- PMID 14256720.
- PMID 16799623.
- PMID 15484322.
- PMID 2474256.
- ^ S2CID 10903371.
- PMID 10436802.
- S2CID 5441766.
- PMID 10536130.
- ^ S2CID 10348568.
- ^ Studies of the performance of serum markers for cholangiocarcinoma (such as carcinoembryonic antigen and CA19-9) in patients with and without primary sclerosing cholangitis include the following:
- Nehls O, Gregor M, Klump B (May 2004). "Serum and bile markers for cholangiocarcinoma". Seminars in Liver Disease. 24 (2): 139–54. S2CID 260316851.
- Siqueira E, Schoen RE, Silverman W, Martin J, Rabinovitz M, Weissfeld JL, et al. (July 2002). "Detecting cholangiocarcinoma in patients with primary sclerosing cholangitis". Gastrointestinal Endoscopy. 56 (1): 40–7. PMID 12085033.
- Levy C, Lymp J, Angulo P, Gores GJ, Larusso N, Lindor KD (September 2005). "The value of serum CA 19-9 in predicting cholangiocarcinomas in patients with primary sclerosing cholangitis". Digestive Diseases and Sciences. 50 (9): 1734–40. S2CID 24744509.
- Patel AH, Harnois DM, Klee GG, LaRusso NF, Gores GJ (January 2000). "The utility of CA 19-9 in the diagnoses of cholangiocarcinoma in patients without primary sclerosing cholangitis". American Journal of Gastroenterology. 95 (1): 204–7. S2CID 11325616.
- Nehls O, Gregor M, Klump B (May 2004). "Serum and bile markers for cholangiocarcinoma". Seminars in Liver Disease. 24 (2): 139–54.
- PMID 9197218.
- PMID 10769604.
- PMID 10489176.
- S2CID 12010522.
- PMID 9725291.
- PMID 10524843.
- S2CID 19988847.
- PMID 9609160.
- PMID 10601064.
- S2CID 25092608.
- S2CID 25350361.
- PMID 14668691.
- PMID 12607576.
- PMID 11882761.
- PMID 9208958.
- ^ Image by
Mikael Häggström, MD. Source for caption:
- Nat Pernick, M.D. "Cytokeratin 19 (CK19, K19)". Pathology Outlines. Last author update: 1 October 2013 - S2CID 7571236.
- ^ Darwin PE, Kennedy A. Cholangiocarcinoma at eMedicine
- PMID 16226963.
- PMID 10903592.
- PMID 15255172.
- PMID 8633917.
- PMID 18773052.
- S2CID 25466829.
- S2CID 37403999.
- PMID 10701737.
- PMID 8138448.
- PMID 10436826.
- PMID 7794082.
- PMID 34515993.
- PMID 12365016.
- ^ "National Comprehensive Cancer Network (NCCN) guidelines on evaluation and treatment of hepatobiliary malignancies" (PDF). Retrieved 13 March 2007.
- ^ a b "NCCN Guidelines for Patients: Gallbladder and Bile Duct Cancers; Hepatobiliary Cancers" (PDF). National Comprehensive Cancer Network. 2021. Retrieved 10 February 2021.
- ^ S2CID 37111064.
- PMID 8879373.
- PMID 10955877.
- PMID 15709089.
- PMID 16760299.
- PMID 14569844.
- PMID 15800324.
- PMID 16809731.
- PMID 21652148.
- ^ a b "BridgeBio Pharma's Affiliate QED Therapeutics and Partner Helsinn Group Announce FDA Approval of Truseltiq (infigratinib) for Patients with Cholangiocarcinoma" (Press release). BridgeBio Pharma. 28 May 2021. Retrieved 28 May 2021 – via GlobeNewswire.
- ^ "Pemazyre Prescribing Information" (PDF). Food and Drug Administration. 2020. Archived (PDF) from the original on 20 July 2020. Retrieved 5 March 2022.
- ^ Research, Center for Drug Evaluation and (1 February 2022). "FDA approves ivosidenib for advanced or metastatic cholangiocarcinoma". FDA.
- ^ "Imfinzi plus chemotherapy reduced risk of death by 20% in 1st-line advanced biliary tract cancer". www.astrazeneca.com. Retrieved 5 March 2022.
- ^ "FDA Approves Taiho's Lytgobi (futibatinib) Tablets for Previously Treated, Unresectable, Locally Advanced or Metastatic Intrahepatic Cholangiocarcinoma" (Press release). Taiho Oncology. 30 September 2022. Retrieved 4 October 2022 – via PR Newswire.
- PMID 10225697.
- PMID 7537346.
- PMID 1709795.
- ^ Studies of surgical outcomes in distal cholangiocarcinoma include:
- Nakeeb A, Pitt HA, Sohn TA, Coleman J, Abrams RA, Piantadosi S, et al. (October 1996). "Cholangiocarcinoma. A spectrum of intrahepatic, perihilar, and distal tumors". Annals of Surgery. 224 (4): 463–73, discussion 473–5. PMID 8857851.
- Jang JY, Kim SW, Park DJ, Ahn YJ, Yoon YS, Choi MG, et al. (January 2005). "Actual long-term outcome of extrahepatic bile duct cancer after surgical resection". Annals of Surgery. 241 (1): 77–84. PMID 15621994.
- Bortolasi L, Burgart LJ, Tsiotos GG, Luque-De León E, Sarr MG (2000). "Adenocarcinoma of the distal bile duct. A clinicopathologic outcome analysis after curative resection". Digestive Surgery. 17 (1): 36–41. S2CID 23190342.
- Fong Y, Blumgart LH, Lin E, Fortner JG, Brennan MF (December 1996). "Outcome of treatment for distal bile duct cancer". British Journal of Surgery. 83 (12): 1712–5. S2CID 30172073.
- Nakeeb A, Pitt HA, Sohn TA, Coleman J, Abrams RA, Piantadosi S, et al. (October 1996). "Cholangiocarcinoma. A spectrum of intrahepatic, perihilar, and distal tumors". Annals of Surgery. 224 (4): 463–73, discussion 473–5.
- ^ Studies of outcome in intrahepatic cholangiocarcinoma include:
- Nakeeb A, Pitt HA, Sohn TA, Coleman J, Abrams RA, Piantadosi S, et al. (October 1996). "Cholangiocarcinoma. A spectrum of intrahepatic, perihilar, and distal tumors". Annals of Surgery. 224 (4): 463–73, discussion 473–5. PMID 8857851.
- Lieser MJ, Barry MK, Rowland C, Ilstrup DM, Nagorney DM (1998). "Surgical management of intrahepatic cholangiocarcinoma: a 31-year experience". Journal of Hepato-Biliary-Pancreatic Surgery. 5 (1): 41–7. PMID 9683753.
- Valverde A, Bonhomme N, Farges O, Sauvanet A, Flejou JF, Belghiti J (1999). "Resection of intrahepatic cholangiocarcinoma: a Western experience". Journal of Hepato-Biliary-Pancreatic Surgery. 6 (2): 122–7. PMID 10398898.
- Nakagohri T, Asano T, Kinoshita H, Kenmochi T, Urashima T, Miura F, et al. (March 2003). "Aggressive surgical resection for hilar-invasive and peripheral intrahepatic cholangiocarcinoma". World Journal of Surgery. 27 (3): 289–93. S2CID 25358444.
- Weber SM, Jarnagin WR, Klimstra D, DeMatteo RP, Fong Y, Blumgart LH (October 2001). "Intrahepatic cholangiocarcinoma: resectability, recurrence pattern, and outcomes". Journal of the American College of Surgeons. 193 (4): 384–91. PMID 11584966.
- Nakeeb A, Pitt HA, Sohn TA, Coleman J, Abrams RA, Piantadosi S, et al. (October 1996). "Cholangiocarcinoma. A spectrum of intrahepatic, perihilar, and distal tumors". Annals of Surgery. 224 (4): 463–73, discussion 473–5.
- ^ Estimates of survival after surgery for perihilar cholangiocarcinoma include:
- Burke EC, Jarnagin WR, Hochwald SN, Pisters PW, Fong Y, Blumgart LH (September 1998). "Hilar Cholangiocarcinoma: patterns of spread, the importance of hepatic resection for curative operation, and a presurgical clinical staging system". Annals of Surgery. 228 (3): 385–94. PMID 9742921.
- Tsao JI, Nimura Y, Kamiya J, Hayakawa N, Kondo S, Nagino M, et al. (August 2000). "Management of hilar cholangiocarcinoma: comparison of an American and a Japanese experience". Annals of Surgery. 232 (2): 166–74. PMID 10903592.
- Chamberlain RS, Blumgart LH (2000). "Hilar cholangiocarcinoma: a review and commentary". Annals of Surgical Oncology. 7 (1): 55–66. S2CID 19569428.
- Washburn WK, Lewis WD, Jenkins RL (March 1995). "Aggressive surgical resection for cholangiocarcinoma". Archives of Surgery. 130 (3): 270–6. PMID 7534059.
- Nagino M, Nimura Y, Kamiya J, Kanai M, Uesaka K, Hayakawa N, et al. (1998). "Segmental liver resections for hilar cholangiocarcinoma". Hepato-Gastroenterology. 45 (19): 7–13. PMID 9496478.
- Rea DJ, Munoz-Juarez M, Farnell MB, Donohue JH, Que FG, Crownhart B, et al. (May 2004). "Major hepatic resection for hilar cholangiocarcinoma: analysis of 46 patients". Archives of Surgery. 139 (5): 514–23, discussion 523–5. PMID 15136352.
- Launois B, Reding R, Lebeau G, Buard JL (2000). "Surgery for hilar cholangiocarcinoma: French experience in a collective survey of 552 extrahepatic bile duct cancers". Journal of Hepato-Biliary-Pancreatic Surgery. 7 (2): 128–34. PMID 10982604.
- Burke EC, Jarnagin WR, Hochwald SN, Pisters PW, Fong Y, Blumgart LH (September 1998). "Hilar Cholangiocarcinoma: patterns of spread, the importance of hepatic resection for curative operation, and a presurgical clinical staging system". Annals of Surgery. 228 (3): 385–94.
- S2CID 295347.
- PMID 12407338.
- PMID 12445422.
- PMID 9449931.
- ^ Cancer Statistics Home Page — National Cancer Institute
- ^ Multiple independent studies have documented a steady increase in the worldwide incidence of cholangiocarcinoma. Some relevant journal articles include:
- Patel T (May 2002). "Worldwide trends in mortality from biliary tract malignancies". BMC Cancer. 2: 10. PMID 11991810.
- Patel T (June 2001). "Increasing incidence and mortality of primary intrahepatic cholangiocarcinoma in the United States". Hepatology. 33 (6): 1353–7. S2CID 23115927.
- Shaib YH, Davila JA, McGlynn K, El-Serag HB (March 2004). "Rising incidence of intrahepatic cholangiocarcinoma in the United States: a true increase?". Journal of Hepatology. 40 (3): 472–7. PMID 15123362.
- West J, Wood H, Logan RF, Quinn M, Aithal GP (June 2006). "Trends in the incidence of primary liver and biliary tract cancers in England and Wales 1971-2001". British Journal of Cancer. 94 (11): 1751–8. PMID 16736026.
- Khan SA, Taylor-Robinson SD, Toledano MB, Beck A, Elliott P, Thomas HC (December 2002). "Changing international trends in mortality rates for liver, biliary and pancreatic tumours". Journal of Hepatology. 37 (6): 806–13. PMID 12445422.
- Welzel TM, McGlynn KA, Hsing AW, O'Brien TR, Pfeiffer RM (June 2006). "Impact of classification of hilar cholangiocarcinomas (Klatskin tumors) on the incidence of intra- and extrahepatic cholangiocarcinoma in the United States". Journal of the National Cancer Institute. 98 (12): 873–5. PMID 16788161.
- Patel T (May 2002). "Worldwide trends in mortality from biliary tract malignancies". BMC Cancer. 2: 10.
- OCLC 953861627.
