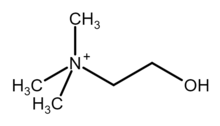
Choline
| |

| |
| Names | |
|---|---|
| IUPAC name
2-Hydroxyethyl(trimethyl)azanium[1]
| |
| Preferred IUPAC name
2-Hydroxy-N,N,N-trimethylethan-1-aminium | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 1736748 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.000.487 |
| EC Number |
|
| 324597 | |
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| [(CH3)3NCH2CH2OH]+ | |
| Molar mass | 104.173 g·mol−1 |
| Appearance | Viscous colorless deliquescent liquid (choline hydroxide)[2] |
| Very soluble (choline hydroxide)[2] | |
| Solubility | soluble in diethylether and chloroform[3] (choline hydroxide)
|
| Structure | |
| Tetrahedral at the nitrogen atom | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Corrosive
|
| GHS labelling: | |

| |
| Danger | |
| H314 | |
| P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
3–6 g/kg (rat, oral)[2] |
| Safety data sheet (SDS) | 4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Choline (
It is a structural part of
Chemistry
Choline is a
Choline chloride can be made by treating TMA with
- (CH3)3N + ClCH2CH2OH → [(CH3)3NCH2CH2OH]+Cl−
The 2-chloroethanol can be generated from ethylene oxide.[how?] Choline has historically been produced from natural sources, such as via hydrolysis of lecithin.[2]
Choline as a nutrient
Choline is widespread in nature in living beings. In most animals, choline phospholipids are necessary components in cell membranes, in the membranes of cell organelles, and in very low-density lipoproteins.[5]
Choline is an
Choline is required to produce
Symptomatic choline deficiency causes
Metabolism
Biosynthesis
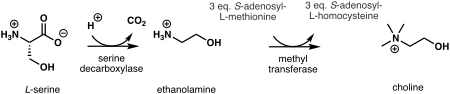
In plants, the first step in

In humans and most other animals, de novo synthesis of choline is via the
Choline can also be released from more complex choline containing molecules. For example,
In humans, certain PEMT-enzyme
Absorption
In humans, choline is absorbed from the
Phosphocholine and
Transport
In humans, choline is transported as a free molecule in blood. Choline–containing
Choline is a water-soluble ion and thus requires transporters to pass through fat-soluble cell membranes. Three types of choline transporters are known:[16]
SLC5A7s are
CTL1s have moderate affinity for choline and transport it in almost all tissues, including the intestines, liver, kidneys,
OCT1s and OCT2s are not associated with the acetylcholine production.[8] They transport choline with low affinity. OCT1s transport choline primarily in the liver and kidneys; OCT2s in kidneys and the brain.[16]
Storage
Choline is stored in the cell membranes and organelles as phospholipids, and inside cells as phosphatidylcholines and glycerophosphocholines.[8]
Excretion
Even at choline doses of 2–8 g, little choline is excreted into urine in humans. Excretion happens via transporters that occur within kidneys (see transport). Trimethylglycine is demethylated in the liver and kidneys to
Function
Choline and its derivatives have many functions in humans and in other organisms. The most notable function is that choline serves as a synthetic precursor for other essential cell components and signalling molecules, such as phospholipids that form cell membranes, the
Phospholipid precursor
Choline is transformed to different phospholipids, like phosphatidylcholines and sphingomyelins. These are found in all cell membranes and the membranes of most cell organelles.[3] Phosphatidylcholines are structurally important part of the cell membranes. In humans 40–50% of their phospholipids are phosphatidylcholines.[8]
Choline phospholipids also form
Phosphatidylcholines are needed for the synthesis of
Choline is also needed for the synthesis of pulmonary surfactant, which is a mixture consisting mostly of phosphatidylcholines. The surfactant is responsible for lung elasticity, that is for lung tissue's ability to contract and expand. For example, deficiency of phosphatidylcholines in the lung tissues has been linked to acute respiratory distress syndrome.[20]
Phosphatidylcholines are excreted into
Acetylcholine synthesis
Choline is needed to produce acetylcholine. This is a neurotransmitter which plays a necessary role in
Source of trimethylglycine
In humans, choline is
Content in foods
Choline occurs in foods as a free molecule and in the form of phospholipids, especially as phosphatidylcholines. Choline is highest in
Trimethylglycine is a functional
| Meats | Vegetables | ||
|---|---|---|---|
| Bacon, cooked | 124.89 | Bean, snap | 13.46 |
| Beef, trim-cut, cooked | 78.15 | Beetroot | 6.01 |
Beef liver , pan fried
|
418.22 | Broccoli | 40.06 |
| Chicken, roasted, with skin | 65.83 | Brussels sprout | 40.61 |
| Chicken, roasted, no skin | 78.74 | Cabbage | 15.45 |
Chicken liver
|
290.03 | Carrot | 8.79 |
| Cod, atlantic | 83.63 | Cauliflower | 39.10 |
| Ground beef, 75–85% lean, broiled | 79.32–82.35 | Sweetcorn, yellow | 21.95 |
| Pork loin cooked | 102.76 | Cucumber | 5.95 |
| Shrimp, canned | 70.60 | Lettuce, iceberg
|
6.70 |
| Dairy products (cow) | Lettuce, romaine | 9.92 | |
| Butter, salted | 18.77 | Pea | 27.51 |
| Cheese | 16.50–27.21 | Sauerkraut | 10.39 |
| Cottage cheese | 18.42 | Spinach | 22.08 |
| Milk, whole/skimmed | 14.29–16.40 | Sweet potato | 13.11 |
| Sour cream | 20.33 | Tomato | 6.74 |
| Yogurt, plain | 15.20 | Zucchini | 9.36 |
| Grains | Fruits | ||
| Oat bran, raw | 58.57 | Apple | 3.44 |
| Oats, plain | 7.42 | Avocado | 14.18 |
| Rice, white | 2.08 | Banana | 9.76 |
| Rice, brown | 9.22 | Blueberry | 6.04 |
| Wheat bran | 74.39 | Cantaloupe | 7.58 |
Wheat germ , toasted
|
152.08 | Grape | 7.53 |
| Others | Grapefruit | 5.63 | |
| Bean, navy | 26.93 | Orange | 8.38 |
Egg, chicken
|
251.00 | Peach | 6.10 |
| Olive oil | 0.29 | Pear | 5.11 |
| Peanut | 52.47 | Prune | 9.66 |
| Soybean, raw | 115.87 | Strawberry | 5.65 |
| Tofu, soft | 27.37 | Watermelon | 4.07 |
- ^ Foods are raw unless noted otherwise. Contents are "total choline" as defined above.
Daily values
This section may require cleanup to meet Wikipedia's quality standards. The specific problem is: Should be merged to above list. The overlaps are quite large to the extent that the values (when converted to 100g) are virtually identical. DV calculation is quite trivial, so this isn't adding anything useful for now. (September 2022) |
The following table contains updated sources of choline to reflect the new Daily Value and the new Nutrition Facts and Supplement Facts Labels.[21] It reflects data from the U.S. Department of Agriculture, Agricultural Research Service. FoodData Central, 2019.[21]
| Food | Milligrams (mg) per serving | Percent DV* |
Beef liver , pan fried, 3 oz (85 g)
|
356 | 65 |
| Egg, hard boiled, 1 large egg | 147 | 27 |
| Beef top round , separable lean only, braised, 3 oz (85 g)
|
117 | 21 |
| Soybeans, roasted, 1⁄2 cup | 107 | 19 |
| Chicken breast, roasted, 3 oz (85 g) | 72 | 13 |
| Beef, ground, 93% lean meat, broiled, 3 oz (85 g) | 72 | 13 |
| Cod, Atlantic, cooked, dry heat, 3 oz (85 g) | 71 | 13 |
| Mushrooms, shiitake, cooked, 1⁄2 cup pieces | 58 | 11 |
Potatoes, red , baked, flesh and skin, 1 large potato
|
57 | 10 |
Wheat germ , toasted, 1 oz (28 g)
|
51 | 9 |
| Beans, kidney, canned, 1⁄2 cup | 45 | 8 |
| Quinoa, cooked, 1 cup | 43 | 8 |
Milk , 1% fat, 1 cup
|
43 | 8 |
| Yogurt, vanilla, nonfat, 1 cup | 38 | 7 |
| Brussels sprouts, boiled, 1⁄2 cup | 32 | 6 |
| Broccoli, chopped, boiled, drained, 1⁄2 cup | 31 | 6 |
| Cottage cheese, nonfat, 1 cup | 26 | 5 |
| Tuna, white, canned in water, drained in solids, 3 oz (85 g) | 25 | 5 |
| Peanuts, dry roasted, 1⁄4 cup | 24 | 4 |
| Cauliflower, 1 in (2.5 cm) pieces, boiled, drained, 1⁄2 cup | 24 | 4 |
Peas, green , boiled, 1⁄2 cup
|
24 | 4 |
Sunflower seeds , oil roasted, 1⁄4 cup
|
19 | 3 |
| Rice, brown, long-grain, cooked, 1 cup | 19 | 3 |
| Bread, pita, whole wheat, 1 large (6+1⁄2 in or 17 cm diameter) | 17 | 3 |
| Cabbage, boiled, 1⁄2 cup | 15 | 3 |
| Tangerine (mandarin orange), sections, 1⁄2 cup | 10 | 2 |
Beans, snap , raw, 1⁄2 cup
|
8 | 1 |
| Kiwifruit, raw, 1⁄2 cup sliced | 7 | 1 |
| Carrots, raw, chopped, 1⁄2 cup | 6 | 1 |
| Apples, raw, with skin, quartered or chopped, 1⁄2 cup | 2 | 0 |
DV = Daily Value. The U.S. Food and Drug Administration (FDA) developed DVs to help consumers compare the nutrient contents of foods and dietary supplements within the context of a total diet. The DV for choline is 550 mg for adults and children age 4 years and older.[25] The FDA does not require food labels to list choline content unless choline has been added to the food. Foods providing 20% or more of the DV are considered to be high sources of a nutrient, but foods providing lower percentages of the DV also contribute to a healthful diet.[21]
The U.S. Department of Agriculture's (USDA's) FoodData Central lists the nutrient content of many foods and provides a comprehensive list of foods containing choline arranged by nutrient content.[21]
Dietary recommendations
Insufficient data is available to establish an estimated average requirement (EAR) for choline, so the Food and Nutrition Board (FNB) established adequate intakes (AIs).[26][27] For adults, the AI for choline was set at 550 mg/day for men and 425 mg/day for women. These values have been shown to prevent hepatic alteration in men. However, the study used to derive these values did not evaluate whether less choline would be effective, as researchers only compared a choline-free diet to a diet containing 550 mg of choline per day. From this, the AIs for children and adolescents were extrapolated.[28][29]
Recommendations are in milligrams per day (mg/day). The
| Age | EFSA adequate intake[8]
|
US NAM adequate intake[21] | US NAM tolerable upper intake levels[21]
|
|---|---|---|---|
| Infants and children | |||
| 0–6 months | Not established | 125 | Not established |
| 7–12 months | 160 | 150 | Not established |
| 1–3 years | 140 | 200 | 1,000 |
| 4–6 years | 170 | 250 | 1,000 |
| 7–8 years | 250 | 250 | 1,000 |
| 9–10 years | 250 | 375 | 1,000 |
| 11–13 years | 340 | 375 | 2,000 |
| Males | |||
| 14 years | 340 | 550 | 3,000 |
| 15–18 years | 400 | 550 | 3,000 |
| 19+ years | 400 | 550 | 3,500 |
| Females | |||
| 14 years | 340 | 400 | 3,000 |
| 15–18 years | 400 | 400 | 3,000 |
| 19+ y | 400 | 425 | 3,500 |
| If pregnant | 480 | 450 | 3,500 (3,000 if ≤18 y) |
| If breastfeeding | 520 | 550 | 3,500 (3,000 if ≤18 y) |
Intake in populations
Twelve surveys undertaken in 9 EU countries between 2000 and 2011 estimated choline intake of adults in these countries to be 269–468 milligrams per day. Intake was 269–444 mg/day in adult women and 332–468 mg/day in adult men. Intake was 75–127 mg/day in infants, 151–210 mg/day in 1- to 3-year-olds, 177–304 mg/day in 3- to 10-year-olds and 244–373 mg/day in 10- to 18-year-olds. The total choline intake mean estimate was 336 mg/day in pregnant adolescents and 356 mg/day in pregnant women.[8]
A study based on the
A 2013–2014 NHANES study of the US population found the choline intake of 2- to 19-year-olds to be 256±3.8 mg/day and 339±3.9 mg/day in adults 20 and over. Intake was 402±6.1 mg/d in men 20 and over and 278 mg/d in women 20 and over.[32]
Deficiency
Signs and symptoms
Symptomatic choline deficiency is rare in humans. Most obtain sufficient amounts of it from the diet and are able to biosynthesize limited amounts of it via
Besides humans, fatty liver is also a typical sign of choline deficiency in other animals. Bleeding in the kidneys can also occur in some species. This is suspected to be due to deficiency of choline derived trimethylglycine, which functions as an osmoregulator.[3]
Causes and mechanisms
In deficiency, availability of phosphatidylcholines in the liver are decreased – these are needed for formation of VLDLs. Thus VLDL-mediated
Excess intake
Excessive doses of choline can have adverse effects. Daily 8–20 g doses of choline, for example, have been found to cause
The liver oxidizes TMA to trimethylamine N-oxide (TMAO). Elevated levels of TMA and TMAO in the body have been linked to increased risk of
Health effects
Neural tube closure
Low maternal intake of choline is associated with an increased risk of neural tube defects. Higher maternal intake of choline is likely associated with better neurocognition/neurodevelopment in children.[36][5] Choline and folate, interacting with vitamin B12, act as methyl donors to homocysteine to form methionine, which can then go on to form SAM (S-adenosylmethionine).[5] SAM is the substrate for almost all methylation reactions in mammals. It has been suggested that disturbed methylation via SAM could be responsible for the relation between folate and NTDs.[37] This may also apply to choline.[citation needed] Certain mutations that disturb choline metabolism increase the prevalence of NTDs in newborns, but the role of dietary choline deficiency remains unclear, as of 2015.[update][5]
Cardiovascular diseases and cancer
Choline deficiency can cause
Cognition
Studies observing the effect between higher choline intake and cognition have been conducted in human adults, with contradictory results.[5][40] Similar studies on human infants and children have been contradictory and also limited.[5]
Perinatal development
This section needs additional citations for verification. (December 2016) |
Both pregnancy and lactation increase demand for choline dramatically. This demand may be met by upregulation of
While maternal stores of choline are depleted during pregnancy and lactation, the placenta accumulates choline by pumping choline against the concentration gradient into the tissue, where it is then stored in various forms, mostly as acetylcholine. Choline concentrations in amniotic fluid can be ten times higher than in maternal blood.[41]
Functions in the fetus
Choline is in high demand during pregnancy as a substrate for building
Choline uptake into the brain is controlled by a low-affinity transporter located at the blood–brain barrier.[49] Transport occurs when arterial blood plasma choline concentrations increase above 14 μmol/L, which can occur during a spike in choline concentration after consuming choline-rich foods. Neurons, conversely, acquire choline by both high- and low-affinity transporters. Choline is stored as membrane-bound phosphatidylcholine, which can then be used for acetylcholine neurotransmitter synthesis later. Acetylcholine is formed as needed, travels across the synapse, and transmits the signal to the following neuron. Afterwards, acetylcholinesterase degrades it, and the free choline is taken up by a high-affinity transporter into the neuron again.[50]
Uses
Choline
Antagonists and inhibitors
Hundreds of choline antagonists and enzyme inhibitors have been developed for research purposes. Aminomethylpropanol is among the first ones used as a research tool. It inhibits choline and trimethylglycine synthesis. It is able to induce choline deficiency that in turn results in fatty liver in rodents. Diethanolamine is another such compound, but also an environmental pollutant. N-cyclohexylcholine inhibits choline uptake primarily in brains. Hemicholinium-3 is a more general inhibitor, but also moderately inhibits choline kinases. More specific choline kinase inhibitors have also been developed. Trimethylglycine synthesis inhibitors also exist: carboxybutylhomocysteine is an example of a specific BHMT inhibitor.[3]
The cholinergic hypothesis of dementia has not only lead to medicinal acetylcholinesterase inhibitors, but also to a variety of acetylcholine inhibitors. Examples of such inhibiting research chemicals include triethylcholine, homocholine and many other N-ethyl derivates of choline, which are false neurotransmitter analogs of acetylcholine. Choline acetyltransferase inhibitors have also been developed.[3]
History
Discovery
In 1849,
In 1865, Oscar Liebreich isolated "neurine" from animal brains.[59][15] The structural formulas of acetylcholine and Liebreich's "neurine" were resolved by Adolf von Baeyer in 1867.[60][55] Later that year "neurine" and sinkaline were shown to be the same substances as Strecker's choline. Thus, Bayer was the first to resolve the structure of choline.[61][62][55] The compound now known as neurine is unrelated to choline.[15]
Discovery as a nutrient
In the early 1930s,
References
- ^ a b "Choline".
- ^ ISBN 9780471484943.
- ^ ISBN 9780849340222.
- ^ "Choline". Lexico Dictionaries. Archived from the original on 24 October 2019. Retrieved 9 November 2019.
- ^ a b c d e f g h i j k l m n "Choline". Micronutrient Information Center, Linus Pauling Institute, Oregon State University. February 2015. Retrieved 11 November 2019.
- ^ a b c Choline. The Metabolomics Innovation Centre, University of Alberta, Edmonton, Canada. 17 August 2016. Retrieved 13 September 2016.
{{cite encyclopedia}}:|website=ignored (help) - ^ Britannica, The Editors of Encyclopaedia. "choline". Encyclopedia Britannica, 11 Dec. 2013, https://www.britannica.com/science/choline. Accessed 17 February 2022.
- ^ .
In this Opinion, the Panel considers dietary choline including choline compounds (e.g. glycerophosphocholine, phosphocholine, phosphatidylcholine, sphingomyelin).
- ^ "Office of Dietary Supplements – Choline".
- PMID 11461929.
- PMID 16653153.
- PMID 10799484.
- PMID 11481443.
- ^ "Superpathway of choline biosynthesis". BioCyc Database Collection: MetaCyc. SRI International.
- ^ PMID 23183298.
- ^ PMID 31547050.
- PMID 23141292.
- PMID 23661599.
- PMID 8333583.
- PMID 29716960.
- ^ a b c d e f g h i "Choline". Office of Dietary Supplements (ODS) at the National Institutes of Health. Retrieved 19 May 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ PMID 12730414.
- doi:10.15482/USDA.ADC/1178141.)
Total choline content was calculated as the sum of Cho, GPC, Pcho, Ptdcho, and SM.
{{cite journal}}: Cite journal requires|journal=(help - ^ "21 CFR 107.100: Infant formula; Nutrient requirements; Nutrient specifications; Choline content". Code of Federal Regulations, Title 21; Food and Drug Administration. 1 April 2019. Retrieved 24 October 2019.
- ^ "Role of choline in human nutrition". Supplements List. 15 March 2024.
- ^ "Office of Dietary Supplements – Choline". ods.od.nih.gov. Retrieved 7 January 2023.
- LCCN 2000028380.)
{{cite report}}: CS1 maint: multiple names: authors list (link - PMID 30332744.
- S2CID 12393618.
- ^ Choline (17 March 2014). "Choline". www.nrv.gov.au. Retrieved 22 October 2019.
- S2CID 24063121.
- ^ "What We Eat in America, NHANES 2013–2014" (PDF). Retrieved 24 October 2019.
- ^ PMID 22134222.
- PMID 30997120.
- PMID 31167879.
- PMID 36041182.
- PMID 24048206.
- PMID 22952174.
- PMID 30825046.
- PMID 30332744.
- ^ PMID 16848706.
- ^ Institute of Medicine, Food and Nutrition Board. Dietary reference intakes for Thiamine, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin and Choline. Washington, DC: National Academies Press. 1998.
- ^ Allen LH (2006). "Pregnancy and lactation". In Bowman BA, Russle RM (eds.). Present Knowledge in Nutrition. Washington DC: ILSI Press. pp. 529–543.
- PMID 10799394.
- S2CID 7051841.
- PMID 12612207.
- PMID 9264309.
- PMID 377313.
- S2CID 34402785.
- PMID 20656095.
- ISBN 9780702069970.
- ISBN 9780471484943.
- PMID 30192819.
- .
- ^ ISBN 9780126337631.
- .
- ^ Gobley T (1874). "Sur la lécithine et la cérébrine". J Pharm Chim (in French). 19 (4): 346–354.
- ^ Sourkes TL (2004). "The discovery of lecithin, the first phospholipid" (PDF). Bull Hist Chem. 29 (1): 9–15. Archived (PDF) from the original on 13 April 2019.
- S2CID 97165871.
- .
- .
- .
- PMID 16994301.
- ISBN 9780309064118.

