Chronic myelogenous leukemia
| Chronic myelogenous leukemia | |
|---|---|
| Other names | Chronic myeloid leukemia, chronic granulocytic leukemia (CGL) |
Haematology and oncology | |
| Frequency | 298,000 (2015)[1] |
| Deaths | 32,200 (2015)[2] |
Chronic myelogenous leukemia (CML), also known as chronic myeloid leukemia, is a cancer of the
CML is largely treated with targeted drugs called
Signs and symptoms
The way CML presents depends on the stage of the disease at diagnosis as it has been known to skip stages in some cases.[4]
Most patients (~90%) are diagnosed during the chronic stage which is most often asymptomatic. In these cases, it may be diagnosed incidentally with an elevated white blood cell count on a routine laboratory test. It can also present with symptoms indicative of hepatosplenomegaly and the resulting left upper quadrant pain this causes. The enlarged spleen may put pressure on the stomach causing a loss of appetite and resulting weight loss. It may also present with mild fever and night sweats due to an elevated basal level of metabolism.[4]
Some (<10%) are diagnosed during the accelerated stage which most often presents bleeding,
Some patients are initially diagnosed in the blast phase in which the symptoms are most likely fever, bone pain and an increase in bone marrow fibrosis.[4]
Cause
In most cases, no obvious cause for CML can be isolated.[5]
Risk factors
CML is more common in males than in females (male to female ratio of 1.4:1) and appears more commonly in the elderly with a median age at diagnosis of 65 years.[5] Exposure to ionising radiation appears to be a risk factor, based on a 50 fold higher incidence of CML in Hiroshima and Nagasaki nuclear bombing survivors.[5] The rate of CML in these individuals seems to peak about 10 years after the exposure.[5]
Pathophysiology

CML was the first cancer to be linked to a clear genetic abnormality, the
In this translocation, parts of two chromosomes (the 9th and 22nd) switch places. As a result, part of the

The fused BCR-ABL protein interacts with the interleukin 3beta(c) receptor subunit. The BCR-ABL transcript is continuously active and does not require activation by other cellular messaging proteins. In turn, BCR-ABL activates a cascade of proteins that control the cell cycle, speeding up cell division. Moreover, the BCR-ABL protein inhibits DNA repair, causing genomic instability and making the cell more susceptible to developing further genetic abnormalities. The action of the BCR-ABL protein is the pathophysiologic cause of chronic myelogenous leukemia. With improved understanding of the nature of the BCR-ABL protein and its action as a tyrosine kinase, targeted therapies (the first of which was imatinib) that specifically inhibit the activity of the BCR-ABL protein have been developed. These tyrosine kinase inhibitors can induce complete remissions in CML, confirming the central importance of bcr-abl as the cause of CML.[8]
Diagnosis

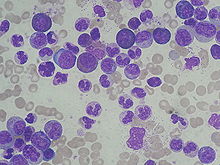

CML is often suspected on the basis of a
Controversy exists over so-called Ph-negative CML, or cases of suspected CML in which the Philadelphia chromosome cannot be detected. Many such patients in fact have complex chromosomal abnormalities that mask the (9;22) translocation, or have evidence of the translocation by
CML must be distinguished from a leukemoid reaction, which can have a similar appearance on a
Classification
CML is often divided into three phases based on clinical characteristics and laboratory findings. In the absence of intervention, CML typically begins in the chronic phase, and over the course of several years progresses to an accelerated phase and ultimately to a blast crisis. Blast crisis is the terminal phase of CML and clinically behaves like an acute leukemia. Drug treatment will usually stop this progression if early. One of the drivers of the progression from chronic phase through acceleration and blast crisis is the acquisition of new chromosomal abnormalities (in addition to the Philadelphia chromosome).[7] Some patients may already be in the accelerated phase or blast crisis by the time they are diagnosed.[10]
Chronic phase
Approximately 85% of patients with CML are in the chronic phase at the time of diagnosis. During this phase, patients are usually asymptomatic or have only mild symptoms of fatigue, left side pain, joint and/or hip pain, or abdominal fullness. The duration of chronic phase is variable and depends on how early the disease was diagnosed as well as the therapies used. In the absence of treatment, the disease progresses to an accelerated phase.[10] Precise patient staging based on clinical markers and personal genomic profile will likely prove beneficial in the assessment of disease history with respect to progression risk.[13]
Accelerated phase
Criteria for diagnosing transition into the accelerated phase are somewhat variable; the most widely used criteria are those put forward by investigators at
- Haematological/cytogenetic criteria
- Persistent or increasing high white blood cell count (> 10 × 109/L), unresponsive to therapy
- Persistent or increasing splenomegaly, unresponsive to therapy
- Persistent thrombocytosis (> 1000 × 109/L), unresponsive to therapy
- Persistent thrombocytopenia (< 100 × 109/L), unrelated to therapy
- ≥ 20% basophils in the peripheral blood
- 10–19% blasts in the peripheral blood and/or bone marrow
- Additional clonal chromosomal abnormalities in Philadelphia (Ph) chromosome-positive (Ph+) cells at diagnosis, including so-called major route abnormalities (a second Ph chromosome, trisomy 8, isochromosome 17q, trisomy 19), complex karyotype, and abnormalities of 3q26.2
- Any new clonal chromosomal abnormality in Ph+ cells that occurs during therapy
- Provisional response-to-TKI criteria
- Haematological resistance (or failure to achieve a complete haematological response d) to the first TKI
- Any haematological, cytogenetic, or molecular indications of resistance to two sequential TKIs
- Occurrence of two or more mutations in the BCR-ABL1 fusion gene during TKI therapy
The patient is considered to be in the accelerated phase if any of the above are present. The accelerated phase is significant because it signals that the disease is progressing and transformation to blast crisis is imminent. Drug treatment often becomes less effective in the advanced stages.[12]
Blast crisis
Blast crisis is the final phase in the evolution of CML, and behaves like an acute leukemia, with rapid progression and short survival.[10] Blast crisis is diagnosed if any of the following are present in a patient with CML:[18]
- >20% blasts in the blood or bone marrow
- The presence of an extramedullary proliferation of blasts
Treatment
The only curative treatment for CML is a bone marrow transplant or an allogeneic stem cell transplant.
Chronic phase
In the past, antimetabolites (e.g.,
Imatinib
The first of this new class of drugs was
Dasatinib, nilotinib, radotinib, bosutinib, and asciminib
To overcome imatinib resistance and to increase responsiveness to TK inhibitors, four novel agents were later developed. The first, dasatinib, blocks several further oncogenic proteins, in addition to more potent inhibition of the BCR-ABL protein, and was approved in 2007, by the U.S. Food and Drug Administration (FDA) to treat CML in people who were either resistant to or intolerant of imatinib. A second TK inhibitor, nilotinib, was approved by the FDA for the same indication. In 2010, nilotinib and dasatinib were also approved for first-line therapy, making three drugs in this class available for treatment of newly diagnosed CML. In 2012, radotinib joined the class of novel agents in the inhibition of the BCR-ABL protein and was approved in South Korea for people resistant to or intolerant of imatinib. Bosutinib received US FDA and EU European Medicines Agency approval on 4 September 2012, and 27 March 2013, respectively for the treatment of adults with Philadelphia chromosome-positive (Ph+) chronic myelogenous leukemia (CML) with resistance, or intolerance to prior therapy.[citation needed]
Asciminib (Scemblix) was approved for medical use in the United States in October 2021.[25]
Treatment-resistant CML
While capable of producing significantly improved responses compared with the action of imatinib, neither dasatinib nor nilotinib could overcome drug resistance caused by one particular mutation found to occur in the structure of BCR-ABL1 known as the T315I mutation (in other words, where the 315th amino acid is mutated from a threonine residue to an isoleucine residue).[citation needed] Two approaches were developed to the treatment of CML as a result:
In 2007,
Independently, ARIAD pharmaceuticals, adapting the chemical structures from first and second-generation TK inhibitors, arrived at a new pan-BCR-ABL1 inhibitor which showed (for the first time) efficacy against T315I, as well as all other known mutations of the oncoprotein. The drug, ponatinib, gained FDA approval in December 2012 for treatment of patients with resistant or intolerant CML. Just as with second-generation TK inhibitors, early approval is being sought to extend the use of ponatinib to newly diagnosed CML also.[citation needed]
Vaccination
In 2005, encouraging but mixed results of
Prognosis
Before the advent of tyrosine kinase inhibitors, the median survival time for CML patients had been about 3–5 years from time of diagnosis.[3]
With the use of tyrosine kinase inhibitors, survival rates have improved dramatically. A 2006 follow-up of 553 patients using imatinib (Gleevec) found an overall survival rate of 89% after five years.[32]
A 2011 followup of 832 patients using imatinib who achieved a stable cytogenetic response found an overall survival rate of 95.2% after 8 years, which is similar to the rate in the general population. Fewer than 1% of patients died because of leukemia progression.[24]
Epidemiology
United Kingdom
CML accounts for 8% of all leukaemias in the UK, and around 680 people were diagnosed with the disease in 2011.[33]
United States
The American Cancer Society estimates that in 2014, about 5,980 new cases of chronic myeloid leukemia were diagnosed, and about 810 people died of the disease. This means that a little over 10% of all newly diagnosed leukemia cases will be chronic myeloid leukemia. The average risk of a person getting this disease is 1 in 588. The disease is more common in men than women, and more common in whites than African-Americans. The average age at diagnosis is 64 years, and this disease is rarely seen in children.[34]
References
- PMID 27733282.
- PMID 27733281.
- ^ a b Besa EC, Buehler B, Markman M, Sacher RA (27 December 2013). Krishnan K (ed.). "Chronic Myelogenous Leukemia". Medscape Reference. WebMD. Retrieved 3 January 2014.
- ^ a b c d e Besa EC, Buehler B, Markman M, Sacher RA (27 December 2013). Krishnan K (ed.). "Chronic Myelogenous Leukemia Clinical Presentation". Medscape Reference. WebMD. Retrieved 3 January 2014.
- ^ ISBN 9781444318548.
- PMID 17671636.
- ^ S2CID 46260906.
- ^ S2CID 1420863.
- S2CID 206550237.
- ^ PMID 17124067.
- S2CID 41243342.
- ^ S2CID 7086172.
- PMID 27048866.
- PMID 3162181.
- PMID 3279515.
- S2CID 9413654.
- S2CID 18338178.
- S2CID 21092684.
- ^ a b Besa EC, Buehler B, Markman M, Sacher RA (27 December 2013). Krishnan K (ed.). "Chronic Myelogenous Leukemia Treatment & Management". Medscape Reference. WebMD. Retrieved 4 January 2014.
- PMID 18472198.
- ISBN 978-1-55009-213-4. Retrieved October 27, 2012.
- ^ Besa EC, Buehler B, Markman M, Sacher RA (27 December 2013). Krishnan K (ed.). "Chronic Myelogenous Leukemia". Medscape Reference. WebMD. Retrieved 3 January 2014.
- S2CID 1761631.
- ^ PMID 21422402.
- ^ "FDA approves Novartis Scemblix (asciminib), with novel mechanism of action for the treatment of chronic myeloid leukemia". Novartis (Press release). Retrieved 29 October 2021.
- S2CID 46509746.
- PMID 17076652.
- ^ "Homoharringtonine (Omacetaxine Mepesuccinate) in Treating Patients With Chronic Myeloid Leukemia (CML) With the T315I BCR-ABL Gene Mutation". ClinicalTrial.gov (database record). Retrieved October 27, 2012.
- ^ "FDA approves Synribo for chronic myelogenous leukemia" (Press release). US Food and Drug Administration. October 26, 2012. Retrieved October 27, 2012.
- ^ "FDA approves new orphan drug for chronic myelogenous leukemia" (Press release). US Food and Drug Administration. September 4, 2012. Retrieved October 27, 2012.
- S2CID 26816784.
- S2CID 21772851.
- ^ "Chronic myeloid leukaemia (CML) statistics". Cancer Research UK. Retrieved 28 October 2014.
- ^ "What are the key statistics about chronic myeloid leukemia?". cancer.org. American Cancer Society. Archived from the original on 9 February 2015. Retrieved 6 January 2015.
External links
- Chronic Myelogenous Leukemia Treatment at the US National Cancer Institute
- Chronic Myelocytic Leukemia (CML) at Merck Manual of Diagnosis and Therapy Professional Edition
