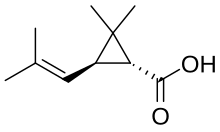
Chrysanthemic acid
| |
| Names | |
|---|---|
| IUPAC name
2,2-Dimethyl-3-(2-methylprop-1-enyl)cyclopropane-1-carboxylic acid
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.022.788 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H16O2 | |
| Molar mass | 168.236 g·mol−1 |
| Melting point | 17 °C (63 °F; 290 K) (1R,3R) or (+)-trans |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chrysanthemic acid is an
allethrins, are esters of all four stereoisomers.[1] Staudinger and Ružička named chrysanthemic acid in 1924.[2]
Biosynthesis
Chrysanthemic acid is derived from its
dimethylallyl diphosphate.[3]
Industrial synthesis
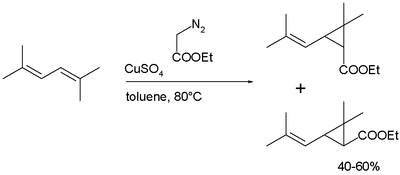
Chrysanthemic acid is produced industrially in a
cis- and trans isomers, followed by hydrolysis of the ester:[4]
Many pyrethroids are accessible by re-esterification of chrysanthemic acid ethylester.