Chytridiomycosis

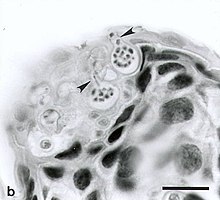
Chytridiomycosis (
History
Whether chytridiomycosis is a new, emergent pathogen or an extant pathogen with recently increased virulence is unclear.
The disease in its epizootic form was first discovered in 1993 in dead and dying frogs in Queensland, Australia. It had been present in the country since at least 1978 and is widespread across Australia. It is also found in Africa, the Americas, Europe, New Zealand, and Oceania. In Australia, Panama, and New Zealand, the fungus seemed to have suddenly 'appeared' and expanded its range at the same time frog numbers declined. In the Americas, it originated in Venezuela in 1987, where it swept up the continent into Central America. It was also found in the lower part of Central America in 1987, where it spread down to meet the upward sweep from South America.[6] However, it may simply be that the fungus occurs naturally and was only identified recently because it has become more virulent or more prevalent in the environment, or because host populations have become less resistant to the disease. The fungus has been detected in four areas of Australia—the east coast, Adelaide, south-west Western Australia and the Kimberley—and is probably present elsewhere.[7] Lately, the genomes of 234 Batrachochytrium dendrobatidis isolates were phylogenetically compared and the results strongly suggest that a lineage found in the Korean peninsula likely seeded the panzootic.[8]
Among frogs, the oldest documented occurrence of Batrachochytrium is from a
Range
The geographic range of chytridiomycosis is difficult to ascertain. If it occurs, the disease is only present where the fungus B. dendrobatidis is present. However, the disease is not always present where the fungus is. Reasons for amphibian declines are often termed ‘enigmatic' because the cause is unknown. Why some areas are affected by the fungus while others are not is not fully understood. Oscillating factors such as climate, habitat suitability, and population density may be factors which cause the fungus to infect amphibians of a given area. Therefore, when considering the geographic range of chytridiomycosis, the range of B. dendrobatidis occurrence must be considered.[6] The geographic range of B. dendrobatidis has recently been mapped, and spans much of the world. B. dendrobatidis has been detected in 56 of 82 countries, and in 516 of 1240 (42%) species using a data set of more than 36,000 individuals. It is widely distributed in the Americas, and detected sporadically in Africa, Asia, and Europe.[2] Asia, for example, has only 2.35% prevalence.[12]
The range suitable for B. dendrobatidis in the New World is vast. Regions with its highest suitability include habitats that contain the world's most diverse amphibian fauna. Areas at risk are the Sierra Madre Pine Oak Occidental Forest, the Sonoran and Sinaloan dry forest, the Veracruz moist forest, Central America east from the Isthmus of Tehuantepec, the Caribbean Islands, the temperate forest in Chile and western Argentina south of 30°S, the Andes above 1000 m above sea level in Venezuela, Colombia, and Ecuador, eastern slopes of the Andes in Peru and Bolivia, the Brazilian Atlantic forest, Uruguay, Paraguay, and northeastern Argentina, as well as the southwestern and Madeira–Tapajós Amazonian rainforests.[13]
Currently, the effects of chytridiomycosis are seen most readily in Central America, eastern Australia, South America, and western North America.[2]
Climate change
A study suggests that changing global temperatures may be responsible for increased proliferation of chytridiomycosis. The rise in temperature has increased evaporation in certain forest environments that as a result has promoted cloud formation.[14] Experts propose that increased cloud cover might actually be decreasing the daytime temperature by blocking the sun, while at night the cloud cover serves as insulation to raise the nighttime temperature from its normal range. The combination of decreased daytime temperature and increased nighttime temperatures may be providing optimal growth and reproduction for Chytrid fungus which has preferred temperature range between 63° and 77 °F (17° and 25 °C).[15] The fungus dies at temperatures at and above 30 °C, which without the cloud cover from increased evaporation is more easily reached by the environment and can, therefore, more easily keep the fungus population in check.[14]
Causative agents
Chytridiomycosis caused by the fungus B. dendrobatidis predominantly affects the outermost layers of skin containing keratin.[6] When most species reach a B. dendrobatidis threshold of 10,000 zoospores, they are not able to breathe, hydrate, osmoregulate, or thermoregulate correctly. This is proven by blood samples that show a lack of certain electrolytes, such as sodium, magnesium, and potassium. B. dendrobatidis is currently known to have two life stages. The first is the asexual zoosporangial stage.[16] When a host first contracts the disease, spores penetrate the skin and attach themselves using microtubule roots.[17] The second stage takes place when the initial asexual zoosporangia produce motile zoospores.[16] To disperse and infect epidermal cells, a wet surface is needed.[16] A second species of Batrachochytrium, B. salamandrivorans, was discovered in 2013 and is known to cause chytridiomycosis in salamanders.[18]
Disease transmission and progression
B. dendrobatidis, a waterborne pathogen, disperses zoospores into the environment.[19] The zoospores use flagella for locomotion through water systems until they reach a new host and enter cutaneously.[17] The B. dendrobatidis' lifecycle continues until new zoospores are produced from the zoosporangium and exit to the environment or reinfect the same host.[17] Once the host is infected with B. dendrobatidis, it can potentially develop chytridiomycosis, but not all infected hosts develop it.[17] Other forms of transmission are currently unknown; however, chytridiomycosis is postulated to be transmitted through direct contact of hosts or through an intermediate host.[17]
Much of how B. dendrobatidis is successfully transmitted from one host to the next is largely unknown.[20] Once released into the aquatic environment, zoospores travel less than 2 cm (0.8 in) within 24 hours before they encyst.[21] The limited range of B. dendrobatidis zoospores suggest some unknown mechanism exists by which they transmit from one host to the next,[21] which can involve the pet trade, and especially the American bullfrog.[22] Abiotic factors such as temperature, pH level, and nutrient levels affect the success of B. dendrobatidis zoospores.[21] The fungus zoospores can survive within a temperature range of 4–25 °C (39–77 °F) and a pH range of 6–7.[21]
Chytridiomycosis is believed to follow this course: zoospores first encounter amphibian skin and quickly give rise to
Besides amphibians Chytridiomycosis also infects crayfish (
Clinical signs
Amphibians infected with B. dendrobatidis have been known to show many different clinical signs. Perhaps the earliest sign of infection is anorexia, occurring as quickly as eight days after being exposed.[20] Individuals infected are also commonly found in a lethargic state, characterized by slow movements, and refuse to move when stimulated. Excessive shedding of skin is seen in most frog species affected by B. dendrobatidis.[6] These pieces of shed skin are described as opaque, gray-white, and tan.[6] Some of these patches of skin are also found adhered to the skin of the amphibians.[6] These signs of infection are often seen 12–15 days following exposure.[20] The most typical symptom of chytridiomycosis is thickening of skin, which promptly leads to the death of the infected individuals because those individuals cannot take in the proper nutrients, release toxins, or, in some cases, breathe.[6] Other common signs are reddening of the skin, convulsions, and a loss of righting reflex.[20] In tadpoles, B. dendrobatidis affects the mouthparts, where keratin is present, leading to abnormal feeding behaviors or discoloration of the mouth.[6]
Research and impact
The amphibian chytrid fungus appears to grow best between 17 and 25 °C (63 and 77 °F),
Although many declines have been credited to the fungus B. dendrobatidis—although likely prematurely so in many cases[4]—some species resist the infection and some populations can survive with a low level of persistence of the disease.[30] In addition, some species that seem to resist the infection may actually harbor a nonpathogenic form of B. dendrobatidis.
Some researchers contend the focus on chytridiomycosis has made amphibian conservation efforts dangerously myopic. A review of the data in the IUCN Red List found the threat of the disease was assumed in most cases, but no evidence shows, in fact, it is a threat.[31] Conservation efforts in New Zealand continue to be focused on curing the critically endangered native Archey's frog, Leiopelma archeyi, of chytridiomycosis, though research has shown clearly that they are immune from infection by B. dendrobatidis and are dying in the wild of other still-to-be identified diseases.[32] In Guatemala, several thousand tadpoles perished from an unidentified pathogen distinct from B. dendrobatidis.[33]
A 2019 It remains unclear how many and which species have been impacted by chytridiomycosis, but there are good data for a limited number of species such as the mountain yellow-legged frog in the Sierra Nevada mountains.
Immunity
Due to the fungus' immense impact on amphibian populations, considerable research has been undertaken to devise methods to combat its proliferation in the wild. Among the most promising is the revelation that amphibians in colonies that survive the passage of the chytrid epidemic tend to carry higher levels of the bacterium
Understanding the interactions of microbial communities present on amphibians' skin with fungal species in the environment can reveal why certain amphibians, such as the frog
One study has postulated that the water flea Daphnia magna eats the spores of the fungus.[44]
Interactions with pesticides
The hypothesis that
Evolution
Hints of emerging evolutionary resistance in a rebounding population of an afflicted frog species were reported from ecological study of an epizootically endangered stream-breeding frog Mixophyes fleayi reported from subtropical Australia.[50] Rebound of frog species in Panama after decline are not associated with pathogen attenuation,[51][52] but rather a host factor - whether an evolved genetic resistance to the fungus infection, or an otherwise acquired trait (such as a hypothetically protective microbial colonization) is yet to be identified.
Treatment options

The use of antifungals and heat-induced therapy has been suggested as a treatment of B. dendrobatidis. However, some of these antifungals may cause adverse skin effects on certain species of frogs, and although they are used to treat species that are infected by chytridiomycosis, the infection is never fully eradicated.[citation needed] A study done by Rollins-Smith and colleagues suggests that itraconazole is the antifungal of choice when it comes to treatment of Bd.[53] This is favored in comparison to amphotericin B and chloramphenicol because of their toxicity—specifically chloramphenicol, as it is correlated with leukemia in toads. This becomes a difficult situation because without treatment, frogs will suffer from limb deformities and even death, but may also suffer skin abnormalities with treatment. "Treatment is not always 100% successful and not all amphibians tolerate treatment very well, therefore chytridiomycosis should always be treated with the advice of a veterinarian."[54]
Individuals infected with B. dendrobatidis are bathed in itraconazole solutions, and within a few weeks, previously infected individuals test negative for B. dendrobatidis using PCR assays.
Bioaugmentation is also considered as a possible treatment against B. dendrobatidis. The amphibian host and even the environment can be augmented with probiotic bacteria that express anti-fungal metabolites that can fight B. dendrobatidis.[60] An example of probiotic application is in the species Rana muscosa in Sierra Nevada; individuals treated with the probiotic J. lividum exhibited greater survival and lower B. dendrobatidis loads compared to untreated controls.[61][62] Similar results were obtained for the Beyşehir frog, an endemic frog species in Turkey (Pelophylax caralitanus).[63]
See also
- Emerging infectious diseases
- Gastric-brooding frog
- Golden toad
- Guajira stubfoot toad
- Holocene extinction
- Rabb's Fringe-limbed Treefrog
- White nose syndrome
References
- . Retrieved 12 November 2021.
- ^ PMID 23463502.

- S2CID 86238651.
- ^ PMID 32193293.
- S2CID 211266075.
- ^ a b c d e f g h Whittaker, Kellie; Vredenburg, Vance. "An Overview of Chytridiomycosis". Amphibiaweb. Retrieved 29 September 2016.
- ^ "Chytridiomycosis (Amphibian Chytrid Fungus Disease)" (PDF). Australian Government Department of Sustainability, Environment, Water, Population and Communities. Archived from the original (PDF) on 11 September 2007. Retrieved 14 October 2013.
- PMID 29748278.
- S2CID 23460986.
- S2CID 25496624.
- ^ a b c d Weldon; du Preez; Hyatt; Muller; and Speare (2004). Origin of the Amphibian Chytrid Fungus. Emerging Infectious Diseases 10(12).
- PMID 21887238.

- S2CID 84272576.
- ^ S2CID 4430672.
- ^ Handwerk, Brian. "Frog Extinctions Linked to Global Warming". National Geographic News. National Geographic. Archived from the original on 14 January 2006. Retrieved 27 May 2016.
- ^ PMID 12102573.
- ^ JSTOR 3761366.
- PMID 24003137.
- PMID 17693553.
- ^ S2CID 17931434.
- ^ PMID 21148822.
- PMID 28562628.
- .
- ^ Padgett-Flohr, G.E. (2007). "Amphibian Chytridiomycosis: An Informational Brochure" (PDF). California Center for Amphibian Disease Control. Archived from the original (PDF) on 13 August 2011. Retrieved 14 October 2013.
- S2CID 205257169.
- ^ Woodhams, D. C., R. A. Alford, et al. (2003). "Emerging disease of amphibians cured by elevated body temperature." Diseases of aquatic organisms 55: 65–67.
- PMID 23519020.
- S2CID 85015019.
- PMID 20202208.
- PMID 15502873.

- PMID 21826233.

- ^ Waldman B (2011) Brief encounters with Archey's Frog. FrogLog 99:39–41.
- S2CID 4421285.
- ^ "Amphibian 'apocalypse' caused by most destructive pathogen ever". Animals. 28 March 2019. Archived from the original on 29 March 2019. Retrieved 6 April 2019.
- ^ Briggs, Helen (29 March 2019). "Killer frog disease extinction toll revealed". Retrieved 29 March 2019.
- S2CID 85565860.
- ^ Black, Richard (6 June 2008). "Bacteria could stop frog killer". BBC News. Retrieved 7 June 2008.
- ^ S2CID 9712168.
- S2CID 27149357.
- S2CID 231788129.
- ^ PMID 19322245.
- PMID 19717627.
- .
- S2CID 13062605.
- ISBN 978-0-691-10251-1.
- .
- PMID 16818245. Archived from the originalon 18 January 2009.
- doi:10.1890/04-1291.
- PMID 17396672.
- PMID 23516509.
- PMID 29599242.
- S2CID 4469435.
- PMID 24771024.
- ^ "Chytrid fungus - causing global amphibian mass extinction". Amphibian Ark (Press release). Archived from the original on 12 June 2019. Retrieved 15 May 2017.
- PMID 22535874.
- PMID 22832723.
- ^ PMID 22422126.
- ^ PMID 21790070.
- PMID 19419002.
- S2CID 47018069.
- PMID 23452227.
- PMID 22259817.
- PMID 25266902.
External links
- Holland, J. (April 2009). "Vanishing". National Geographic Magazine. Sartore, Joel (photograph). Archived from the original on 28 October 2017. Retrieved 16 April 2022.)
We are witnessing a mass extinction. An exotic fungus is delivering the fatal blow to many amphibians already hit by habitat loss, pollution, and climate change.
{{cite news}}: CS1 maint: unfit URL (link - Wildlife Trade and Global Disease Emergence. cdc.gov (Report). U.S. Center for Disease Control.
- "Main preventive management strategies for the Chytrid fungus" (PDF). issg.org (Press release). Invasive Species Specialist Group.
- "Chytridiomycosis". Amphibian Diseases. Australia: James Cook University. Archived from the original on 16 February 2007. Retrieved 16 April 2022.
- "'Amphibian Ark' aims to save frogs from fungus". Associated Press. 15 February 2007. Retrieved 16 April 2022.
Scientists from around the world are meeting to organize a worldwide effort to stem the deaths of frogs by asking zoos, aquariums and botanical gardens to take in threatened frogs until a mystery fungus can be stopped.
