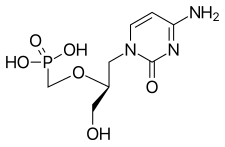
Cidofovir
 | |
| Clinical data | |
|---|---|
| Trade names | Vistide |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
Intravenous | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | complete |
| Protein binding | <6% |
| Elimination half-life | 2.6 hours (active metabolites: 15–65 hours) |
| Excretion | renal The above pharmacokinetic parameters are measured for cidofovir used in conjunction with probenecid.[3] |
| Identifiers | |
| |
JSmol) | |
| Specific rotation | -97.3 |
| Melting point | 260 °C (500 °F) |
| |
| |
| (verify) | |
Cidofovir, brand name Vistide, is a
Cidofovir was approved for medical use in 1996.[6]
Medical use
DNA virus
Its only indication that has received regulatory approval worldwide is
It first received FDA approval on 26 June 1996,[16] TGA approval on 30 April 1998[5] and EMA approval on 23 April 1997.[17]
It has been used topically to treat
Other
It has been suggested as an antitumour agent, due to its suppression of
Administration
Cidofovir is only available as an intravenous formulation. Cidofovir is to be administered with probenecid which decreases side effects to the kidney.[22] Probenecid mitigates nephrotoxicity by inhibiting organic anion transport of the proximal tubule epithelial cells of the kidney.[23] In addition, hydration must be administered to patients receiving cidofovir. 1 liter of normal saline is recommended in conjunction with each dose of cidofovir.[22]
Side effects
The major dose-limiting side effect of cidofovir is nephrotoxicity (i.e., kidney damage).[24] Other common side effects (occurring in >1% of people treated with the drug) include:[4][24]
- Nausea
- Vomiting
- Neutropenia
- Hair loss
- Weakness
- Headache
- Chills
- Decreased intraocular pressure
- Uveitis
- Iritis
Whereas uncommon side effects include:
Contraindications
Hypersensitivity to cidofovir or probenecid (as probenecid needs to be given concurrently to avoid nephrotoxicity).[4]
Interactions
It is known to interact with nephrotoxic agents (e.g.
Mechanism of action
Its active metabolite, cidofovir diphosphate, inhibits viral replication by selectively inhibiting viral
It possesses in vitro activity against the following viruses:[26]
- Human herpesviruses
- Adenoviruses
- Human poxviruses (including the smallpoxvirus)
- Human papillomavirus
History
Cidofovir was discovered at the Institute of Organic Chemistry and Biochemistry, Prague, by Antonín Holý, and developed by Gilead Sciences[27] and is marketed with the brand name Vistide by Gilead in the US, and by Pfizer elsewhere.
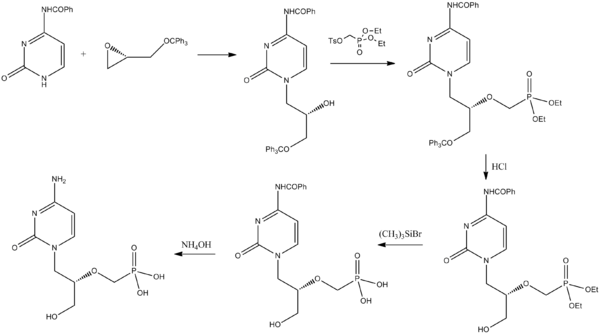
Synthesis
Cidofovir can be synthesized from a pyrimidone derivative and a protected derivative of glycidol.[28]
See also
- Brincidofovir, a prodrug of cidofovir
References
- FDA. Retrieved 22 Oct 2023.
- ^ "Drug and medical device highlights 2018: Helping you maintain and improve your health". Health Canada. 14 October 2020. Retrieved 17 April 2024.
- ^ Cundy, Kenneth C. "Clinical Pharmacokinetics of the Antiviral Nucleotide Analogues Cidofovir and Adefovir." Clinical Pharmacokinetics 36.2 (1999): 127–143.
- ^ a b c d e "Vistide (cidofovir) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 4 February 2014.
- ^ a b c d e f g h "Product Information VISTIDE®". TGA eBusiness Services. Gilead Sciences Pty Ltd. 3 September 2013. Retrieved 5 February 2014.
- ISBN 978-1437727029.
- PMID 12757254.
- S2CID 32026770.[permanent dead link]
- PMID 23737811.
- PMID 12076747.
- S2CID 22903225.
- PMID 16048917.
- S2CID 24131709.
- PMID 18458927.
- S2CID 22895067.
- ^ "Cidofovir Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 5 February 2014.
- ^ "Vistide : EPAR – Product Information" (PDF). European Medicines Agency. Gilead Sciences International Ltd. 7 November 2013. Retrieved 5 February 2014.
- S2CID 32295082.
- S2CID 23136929.
- PMID 21994641.
- ^ "Interim Clinical Guidance for the Treatment of Monkeypox". U.S. Centers for Disease Control and Prevention. 26 May 2022.
- ^ a b "Details" (PDF). www.gilead.com. Retrieved 2019-06-05.
- PMID 9742650.
- ^ ISBN 978-0-9805790-9-3.
- package insert). Gilead Sciences. September 2010. Dosage and Administration: Dosage.
- S2CID 32366514.
- ^ "Press Releases: Gilead". Archived from the original on 2013-02-08. Retrieved 2007-12-05.
- .