Cis–trans isomerism


Cis–trans isomerism, also known as geometric isomerism, describes certain arrangements of atoms within molecules. The prefixes "cis" and "trans" are from Latin: "this side of" and "the other side of", respectively.
According to
Cis–trans or geometric isomerism is classified as one type of configurational isomerism.[3]
Organic chemistry
In general, cis–trans stereoisomers contain
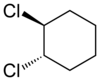 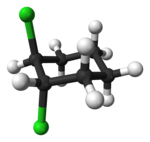 |
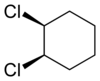 
|
| trans-1,2-dichlorocyclohexane | cis-1,2-dichlorocyclohexane |
Comparison of physical properties
Cis and trans isomers have distinct physical properties. Their differing shapes influences the
 |

|
| cis-2-pentene | trans-2-pentene |
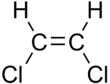 |

|
| cis-1,2-dichloroethene | trans-1,2-dichloroethene |
These differences can be very small, as in the case of the boiling point of straight-chain alkenes, such as
 |
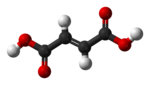
|
| cis-butenedioic acid (maleic acid) |
trans-butenedioic acid (fumaric acid) |
 |

|
| cis-9-octadecenoic acid (oleic acid) |
trans-9-octadecenoic acid (elaidic acid) |
The differing properties of the two isomers of butenedioic acid are often very different.
| maleic acid | fumaric acid | |
|---|---|---|
| color | white | white |
| melting point, °C | 130 | 286 |
| water solubility, g/L | 788 | 7 |
| Acid dissociation constant, pKa1 | 1.90 | 3.03 |
Polarity is key in determining relative boiling point as strong intermolecular forces raise the boiling point. In the same manner, symmetry is key in determining relative melting point as it allows for better packing in the solid state, even if it does not alter the polarity of the molecule. Another example of this is the relationship between oleic acid and elaidic acid; oleic acid, the cis isomer, has a melting point of 13.4 °C, making it a liquid at room temperature, while the trans isomer, elaidic acid, has the much higher melting point of 43 °C, due to the straighter trans isomer being able to pack more tightly, and is solid at room temperature.
Thus, trans alkenes, which are less polar and more symmetrical, have lower boiling points and higher melting points, and cis alkenes, which are generally more polar and less symmetrical, have higher boiling points and lower melting points.
In the case of geometric isomers that are a consequence of double bonds, and, in particular, when both substituents are the same, some general trends usually hold. These trends can be attributed to the fact that the dipoles of the substituents in a cis isomer will add up to give an overall molecular dipole. In a trans isomer, the dipoles of the substituents will cancel out [7] due to being on opposite sides of the molecule. Trans isomers also tend to have lower densities than their cis counterparts.[citation needed]
As a general trend, trans alkenes tend to have higher melting points and lower solubility in inert solvents, as trans alkenes, in general, are more symmetrical than cis alkenes.[8]
Stability
Usually for acyclic systems trans isomers are more stable than cis isomers. This difference is attributed to the unfavorable
E–Z notation

In principle, cis–trans notation should not be used for alkenes with two or more different substituents. Instead the E–Z notation is used based on the priority of the substituents using the Cahn–Ingold–Prelog (CIP) priority rules for absolute configuration. The IUPAC standard designations E and Z are unambiguous in all cases, and therefore are especially useful for tri- and tetrasubstituted alkenes to avoid any confusion about which groups are being identified as cis or trans to each other.
Z (from the German zusammen) means "together". E (from the German entgegen) means "opposed" in the sense of "opposite". That is, Z has the higher-priority groups cis to each other and E has the higher-priority groups trans to each other. Whether a molecular configuration is designated E or Z is determined by the CIP rules; higher atomic numbers are given higher priority. For each of the two atoms in the double bond, it is necessary to determine the priority of each substituent. If both the higher-priority substituents are on the same side, the arrangement is Z; if on opposite sides, the arrangement is E.
Because the cis–trans and E–Z systems compare different groups on the alkene, it is not strictly true that Z corresponds to cis and E corresponds to trans. For example, trans-2-chlorobut-2-ene (the two methyl groups, C1 and C4, on the
Undefined alkene stereochemistry
Wavy single bonds are the standard way to represent unknown or unspecified stereochemistry or a mixture of isomers (as with tetrahedral stereocenters). A crossed double-bond has been used sometimes; it is no longer considered an acceptable style for general use by
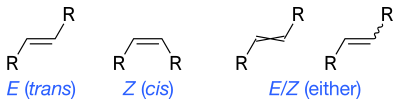
Inorganic chemistry
Cis–trans isomerism can also occur in inorganic compounds.
Diazenes
  |
 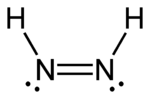
|
| trans-diazene | cis-diazene |
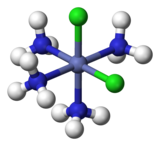
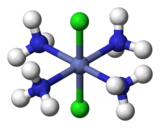
Coordination complexes

For example, there are two isomers of
For
A related type of isomerism in octahedral MX3Y3 complexes is
See also
- Chirality (chemistry)
- Descriptor (chemistry)
- E–Z notation
- Isomer
- Structural isomerism
- Trans fat
References
- ^ Charlton T. Lewis, Charles Short, A Latin Dictionary (Clarendon Press, 1879) Entry for cis
- ^ Hunt, Ian. "Stereochemistry". University of Calgary. Retrieved 3 November 2023.
- ^ Reusch, William (2010). "Stereoisomers Part I". Virtual Textbook of Organic Chemistry. Michigan State University. Retrieved 7 April 2015.
- ^ "Chemicalland values". Chemicalland21.com. Retrieved 2010-06-22.
- ^ CRC Handbook of Chemistry and Physics (60th ed.). 1979–1980. p. C-298.
- ISBN 978-0-12-802444-7.
- ^ ISBN 978-0-471-85472-2.
- ISBN 978-0-07-707212-4.
- .
- .
- S2CID 97528124.