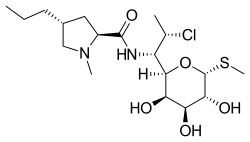
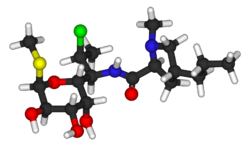
Clindamycin
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /klɪndəˈmaɪsɪn/ |
| Trade names | Cleocin, Clinacin, Dalacin, others |
| Other names | 7-chloro-lincomycin 7-chloro-7-deoxylincomycin, DARE-BV1 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682399 |
| License data |
|
| Pregnancy category |
|
topical, intravenous, intravaginal | |
| Drug class | Lincosamide antibiotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 90% (by mouth) 4–5% (topical) |
| Protein binding | 95% |
| Metabolism | Liver |
| Elimination half-life | 2–3 hour |
| Excretion | Bile duct and kidney (around 20%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Clindamycin is a
Common side effects include nausea and vomiting, diarrhea, rashes, and pain at the site of injection.
Clindamycin was first made in 1966 from
Medical uses
Clindamycin is used primarily to treat
Acne

For the treatment of acne, in the long term, the combined use of topical clindamycin and benzoyl peroxide was similar to salicylic acid plus benzoyl peroxide.[21][22] Topical clindamycin plus topical benzoyl peroxide is more effective than topical clindamycin alone.[21][22]
Susceptible bacteria
It is most effective against infections involving the following types of organisms:
- Aerobic
- Anaerobic, rod-shaped bacteria, including some Bacteroides, Fusobacterium, and Prevotella, although resistance is increasing in Bacteroides fragilis.[24]
Most aerobic Gram-negative bacteria (such as Pseudomonas, Legionella, Haemophilus influenzae and Moraxella) are resistant to clindamycin,[23][25] as are the facultative anaerobic Enterobacteriaceae.[26] A notable exception is Capnocytophaga canimorsus, for which clindamycin is a first-line drug of choice.[27]
The following represents MIC susceptibility data for a few medically significant pathogens.[28]
- Staphylococcus aureus: 0.016 μg/mL – >256 μg/mL
- Streptococcus pneumoniae: 0.002 μg/mL – >256 μg/mL
- Streptococcus pyogenes: <0.015 μg/mL – >64 μg/mL
D-test
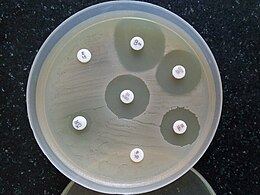
When testing a gram-positive culture for sensitivity to clindamycin, it is common to perform a "D-test" to determine if there is a sub-population of
To perform a D-test, an
If the area of inhibition around the clindamycin disk is circular, the test result is negative and clindamycin can be used.[30]
Malaria
Given with chloroquine or quinine, clindamycin is effective and well tolerated in treating Plasmodium falciparum malaria; the latter combination is particularly useful for children, and is the treatment of choice for pregnant women who become infected in areas where resistance to chloroquine is common.[31][32] Clindamycin should not be used as an antimalarial by itself, although it appears to be very effective as such, because of its slow action.[31][32] Patient-derived isolates of Plasmodium falciparum from the Peruvian Amazon have been reported to be resistant to clindamycin as evidenced by in vitro drug susceptibility testing.[33]
Other
Clindamycin may be useful in skin and soft tissue infections caused by methicillin-resistant Staphylococcus aureus (MRSA).[6] Many strains of MRSA are still susceptible to clindamycin; however, in the United States spreading from the West Coast eastwards, MRSA is becoming increasingly resistant.[medical citation needed]
While it has been used in
Clindamycin is used in cases of suspected
Clindamycin has been proven to decrease the risk of
The combination of clindamycin and quinine is the standard treatment for severe babesiosis.[39]
Clindamycin may also be used to treat toxoplasmosis,[23][40][41] and, in combination with primaquine, is effective in treating mild to moderate Pneumocystis jirovecii pneumonia.[42]
Clindamycin, either applied to skin or taken by mouth, may also be used in hidradenitis suppurativa.[43]
Side effects
Common
Rarely – in less than 0.1% of people – clindamycin therapy has been associated with anaphylaxis, blood dyscrasias, polyarthritis, jaundice, raised liver enzyme levels, renal dysfunction, cardiac arrest, and/or hepatotoxicity.[44]
Clostridioides difficile
Pregnancy and breastfeeding
Use of clindamycin during pregnancy is generally considered safe.[48]
Clindamycin is classified as compatible with breastfeeding by the American Academy of Pediatrics,[49] however, the WHO categorizes it as "avoid if possible".[50] It is classified as L2 probably compatible with breastfeeding according to Medications and Mothers' Milk.[51] A 2009 review found it was likely safe in breastfeeding mothers, but did find one complication (hematochezia) in a breastfed infant which might be attributable to clindamycin.[52] LactMed lists potentially negative gastrointestinal effects in babies whose mothers take it while breastfeeding but did not see that as justification to stop breastfeeding.[53]
Interactions
Clindamycin may prolong the effects of
Chemistry
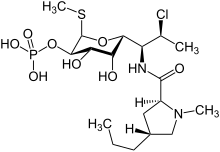
Clindamycin is a
Clindamycin is white or yellow.[61] It is very soluble in water.[61] The topically used clindamycin phosphate is a phosphate-ester prodrug of clindamycin.[57]
Mechanism of action

Clindamycin has a primarily
The X-ray crystal structures of clindamycin bound to ribosomes (or ribosomal subunits) derived from Escherichia coli,[65] Deinococcus radiodurans,[66] and Haloarcura marismortui[67] have been determined; the structure of the closely related antibiotic lincomycin bound to the 50S ribosomal subunit of Staphylococcus aureus has also been reported.[68]
Society and culture
Economics
Clindamycin is available as a
Available forms

Clindamycin preparations that are taken by mouth include
Several combination acne treatments containing clindamycin are also marketed, such as single-product formulations of clindamycin with
Clindamycin is available as a generic drug, for both systemic (oral and intravenous) and topical use.[31] (The exception is the vaginal suppository, which is not available as a generic in the US[72]).
Veterinary use
The
References
- ^ Use During Pregnancy and Breastfeeding
- FDA. Retrieved 22 October 2023.
- ^ a b "Xaciato- clindamycin phosphate gel". DailyMed. Retrieved 24 December 2021.
- ^ a b c d e f g h i j "Clindamycin (Systemic)". The American Society of Health-System Pharmacists. Archived from the original on 12 August 2021. Retrieved 19 December 2021.
- ^ ISBN 9783540331018. Archivedfrom the original on 8 September 2017.
- ^ PMID 17652653.
- ^ "Clindamycin phosphate- clindamycin phosphate gel usp, 1% gel". DailyMed. Retrieved 19 December 2021.
- ^ "Daré Announces FDA Approval of Xaciato (clindamycin phosphate) Vaginal Gel as a Treatment for Bacterial Vaginosis". Daré Bioscience (Press release). 7 December 2021. Retrieved 19 December 2021.
- ^ PMID 12746372.
- PMID 22346533.
- ISBN 9781118819517. Archivedfrom the original on 8 September 2017.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ ISBN 9781284057560.
- ^ "Competitive Generic Therapy Approvals". U.S. Food and Drug Administration (FDA). 29 June 2023. Archived from the original on 29 June 2023. Retrieved 29 June 2023.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Clindamycin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- PMID 16243239.
- ^ a b "Cleocin I.V. Indications & Dosage". RxList.com. 2007. Archived from the original on 27 November 2007. Retrieved 1 December 2007.
- PMID 15117932.
- (PDF) from the original on 27 July 2011.
- ^ PMID 31613567.
- ^ PMID 20488582.
- ^ a b c d "Lincosamides, Oxazolidinones, and Streptogramins". Merck Manual of Diagnosis and Therapy. Merck & Co. May 2020. Archived from the original on 2 December 2007. Retrieved 19 December 2021.
- S2CID 248736289.
- ^ a b Bell EA (January 2005). "Clindamycin: new look at an old drug". Infectious Diseases in Children. Archived from the original on 8 October 2011. Retrieved 1 December 2007.
- Google Book Search.
- PMID 17250994.
- ^ "Clindamycin Phosphate Susceptibility and Minimum Inhibitory Concentration (MIC) Data" (PDF). toku-e.com. 1 June 2020.
- PMID 11797175.
- ^ PMID 19935273.
- ^ PMID 12121898.
- ^ PMID 17519416.
- PMID 20829224.
- S2CID 24494787.
- S2CID 29061418.
- PMID 6662837.
- PMID 7014632.
- ^ S2CID 25572794.
- PMID 10885987.
- S2CID 36696180.
- PMID 9296037.
- PMID 9624465.
- S2CID 5017318.
- ^ a b c Rossi S, ed. (2006). Australian Medicines Handbook. Adelaide: AMH Pty Ltd.
- ^ PMID 17635503.
- PMID 16141157.
- PMID 8043060.
- PMID 12121898.
- PMID 11533352.
- )
- OCLC 959873270.
- S2CID 2594769.
- PMID 30000267. Bookshelf ID: NBK501208.
- PMID 4415332.
- PMID 7553999.
- PMID 11772813.
- ^ a b "Clindamycin Phosphate Topical Solution". RxList. Archived from the original on 2 February 2017. Retrieved 27 January 2017.
- PMID 4916317.
- PMID 4389137.
- PMID 5985307.
- ^ S2CID 7870760.
- ^ Clindamycin University of Michigan. Retrieved 31 July 2009
- S2CID 9264620.
- ^ Beauduy CE, Winston LG. Tetracyclines, Macrolides, Clindamycin, Chloramphenicol, Streptogramins, & Oxazolidinones. In: Katzung BG. eds. Basic & Clinical Pharmacology, 14e New York, NY: McGraw-Hill; .
- PMID 20876128.
- S2CID 205022511.
- S2CID 7086043.
- PMID 28973455.
- ISBN 978-1-4200-9241-7.
- PMID 12182256.
- ^ Waknine Y (1 December 2006). "FDA Approvals: Ziana, Kadian, Polyphenon E". Medscape Medical News. Archived from the original on 6 February 2011. Retrieved 1 December 2007.
- ^ "Generic Cleocin Vaginal Availability". Drugs.com. Retrieved 13 October 2019.
- ISBN 0-911910-50-6. Retrieved 14 December 2007.
- ISBN 0-911910-50-6. Retrieved 14 December 2007.
External links
- Clinical trial number NCT04370548 for "DARE-BV1 in the Treatment of Bacterial Vaginosis (DARE-BVFREE)" at ClinicalTrials.gov
