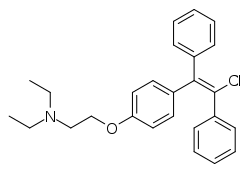
Clomifene
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Clomid, Serophene, others[1] |
| Other names | Clomiphene; Chloramifene; Chloramiphene; MRL-41; MRL/41; NSC-35770 |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Selective estrogen receptor modulator; Progonadotropin |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | High (>90%) |
| Metabolism | Liver CYP2D6 (with enterohepatic circulation)[2] |
| Metabolites | 4-Hydroxyclomiphene (4-OH-CLO), 4-Hydroxy-N-desethylclomiphene (4-OH-DE-CLO) |
| Elimination half-life | 4 – 7 days [2][3][4] active metabolites: |
| Excretion | Mainly feces, some in urine |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Clomifene, also known as clomiphene, is a medication used to treat
Common side effects include
Clomifene was approved for medical use in the United States in 1967.[5] It is on the World Health Organization's List of Essential Medicines, under the category "Ovulation inducers" (Complementary List).[9] Its introduction began the era of assisted reproductive technology.[10]
Clomifene (particularly the purified enclomiphene isomer) has also been found to have a powerful ability to boost or restore testosterone levels in hypogonadal men.[11]
Medical uses
Ovulation induction
Clomifene is one of several alternatives for
Proper timing of the drug is important; it should be taken starting on about the fifth day of the cycle, and there should be frequent intercourse.[7][12][14]
The following procedures may be used to monitor induced cycles:[12]
- Follicular monitoring with luteinizationsuch as loss of clearly defined follicular margins and appearance of internal echoes.
- Serum estradiol levels, starting 4–6 days after the last pill
- Adequacy of LH surge by urine LH surgetests 3 to 4 days after last clomifene pill
- Post-coital test 1–3 days before ovulation to check whether there are at least 5 progressive sperm per HPF
- Mid-luteal progesterone, with at least 10 ng/ml 7–9 days after ovulation being regarded as adequate.
Repeat dosing: This 5-day treatment course can be repeated every 30 days. The dosage may be increased by 50-mg increments in subsequent cycles until ovulation is achieved.[12] It is not recommended by the manufacturer to use clomifene for more than 6 cycles.[7][15]
It is no longer recommended to perform an ultrasound examination to exclude any significant residual ovarian enlargement before each new treatment cycle.[12]
Other uses
Clomifene has also been used with other assisted reproductive technology to increase success rates of these other modalities.[16]
Testosterone replacement therapy
Clomifene is sometimes used in the treatment of male
Clomifene consists of two
Gynecomastia
Clomifene has been used in the treatment of gynecomastia.[21] It has been found to be useful in the treatment of some cases of gynecomastia but it is not as effective as tamoxifen or raloxifene for this indication.[22] It has shown variable results for gynecomastia (probably because the zuclomifene isomer is estrogenic), and hence is not recommended for treatment of the condition.[23] Pure enclomifene isomer is likely to be more effective than clomifene at treating gynecomastia, because of the lack of the zuclomifene isomer (as noted above).
Due to its long half-life, zuclomifene can be detected in urine for at least 261 days after discontinuation[24] (261 days after discontinuation with a half-life of 30 days, there is still 0.24% of the peak level of zuclomifene being excreted, whereas with a half-life of 10 hours, enclomifene reaches the same 0.24% level in less than 4 days).
Because of its potential for boosting testosterone, clomifene is listed as banned for use by competitive sportsmen, both in and out of competition, by the
Contraindications
Contraindications include an allergy to the medication, pregnancy, prior liver problems, abnormal vaginal bleeding of unclear cause, ovarian cysts other than those due to polycystic ovarian syndrome, unmanaged adrenal or thyroid problems, and
Side effects
The most common adverse drug reaction associated with the use of clomifene (>10% of people) is reversible ovarian enlargement.[7]
Less common effects (1–10% of people) include visual symptoms (blurred vision,
Rare adverse events (<1% of people) include:
Clomifene can lead to multiple ovulation, hence increasing the chance of twins (10% of births instead of ~1% in the general population) and triplets.
Rates of birth defects and miscarriages do not appear to change with the use of clomifene for fertility.
Cancer risk
Some studies have suggested that clomifene if used for more than a year may increase the risk of ovarian cancer.[13] This may only be the case in those who have never been and do not become pregnant.[15] Subsequent studies have failed to support those findings.[12][26]
Clomifene has been shown to be associated with an increased risk of malignant melanomas and thyroid cancer.[3] Thyroid cancer risk was not associated with the number of pregnancies carried to viability.[27]
Pharmacology
Pharmacodynamics
Selective estrogen receptor modulator activity
Clomifene is a
Clomifene is a long-acting ER
Even though clomifene has some
In normal physiologic female hormonal cycling, at 7 days past ovulation, high levels of estrogen and progesterone produced from the corpus luteum inhibit GnRH, FSH, and LH at the hypothalamus and anterior pituitary. If fertilization does not occur in the post-ovulation period the corpus luteum disintegrates due to a lack of human chorionic gonadotropin (hCG). This would normally be produced by the embryo in the effort of maintaining progesterone and estrogen levels during pregnancy.
Therapeutically, clomifene is given early in the menstrual cycle. It is typically prescribed beginning on day 3 and continuing for five days. By that time, FSH levels are rising steadily, causing the development of a few follicles. Follicles, in turn, produce the estrogen, which circulates in serum. In the presence of clomifene, the body perceives a low level of estrogen, similar to day 22 in the previous cycle. Since estrogen can no longer effectively exert negative feedback on the hypothalamus, GnRH secretion becomes more rapidly pulsatile, which results in increased pituitary gonadotropin release. (More rapid, lower amplitude pulses of GnRH lead to increased LH and FSH secretion, while more irregular, larger amplitude pulses of GnRH leads to a decrease in the ratio of LH to FSH.) Increased FSH levels cause the growth of more ovarian follicles, and subsequently rupture of follicles resulting in ovulation. Ovulation occurs most often 6 to 7 days after a course of clomifene.
In normal men, 50 mg/day clomifene for 8 months has been found to increase
| Medication | Breast | Bone | Liver | Uterus | Vagina | Brain | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Lipids |
Coagulation | SHBG | IGF-1 | Hot flashes | Gonadotropins | |||||||||
| Estradiol | + | + | + | + | + | + | + | + | + | + | ||||
| "Ideal SERM" | – | + | + | ± | ± | ± | – | + | + | ± | ||||
| Bazedoxifene | – | + | + | + | + | ? | – | ± | – | ? | ||||
| Clomifene | – | + | + | ? | + | + | – | ? | – | ± | ||||
| Lasofoxifene | – | + | + | + | ? | ? | ± | ± | – | ? | ||||
| Ospemifene | – | + | + | + | + | + | ± | ± | – | ± | ||||
| Raloxifene | – | + | + | + | + | + | ± | – | – | ± | ||||
| Tamoxifen | – | + | + | + | + | + | + | – | – | ± | ||||
| Toremifene | – | + | + | + | + | + | + | – | – | ± | ||||
| Effect: + = Estrogenic / agonistic. ± = Mixed or neutral. – = Antiestrogenic / antagonistic. Note: SERMs generally increase gonadotropin levels in hypogonadal and eugonadal men as well as premenopausal women (antiestrogenic) but decrease gonadotropin levels in postmenopausal women (estrogenic). Sources: See template. | ||||||||||||||
Other activities
Clomifene is an
Pharmacokinetics
Clomifene produces N-desmethylclomifene,
Clomifene has an
Most clomifene metabolism occurs in the
Chemistry
Clomifene is a
 |
 |
History
A team at
| Antiestrogen | Dosage | Year(s) | Response rate | Adverse effects |
|---|---|---|---|---|
| Ethamoxytriphetol | 500–4,500 mg/day | 1960 | 25% | Acute psychotic episodes
|
| Clomifene | 100–300 mg/day | 1964–1974 | 34% | Risks of cataracts |
| Nafoxidine | 180–240 mg/day | 1976 | 31% | Cataracts, ichthyosis, photophobia |
| Tamoxifen | 20–40 mg/day | 1971–1973 | 31% | Transient thrombocytopeniaa |
| Footnotes: a = "The particular advantage of this drug is the low incidence of troublesome side effects (25)." "Side effects were usually trivial (26)." Sources: [48][50] | ||||
Clinical studies were conducted under an
The drug is widely considered to have been a revolution in the treatment of female infertility, the beginning of the modern era of
The company was acquired by
Society and culture
Brand names
Clomifene is marketed under many brand names worldwide, including Beclom, Bemot, Biogen, Blesifen, Chloramiphene, Clofert, Clomene, ClomHEXAL, Clomi, Clomid, Clomidac, Clomifen, Clomifencitrat, Clomifene, Clomifène, Clomifene citrate, Clomifeni citras, Clomifeno, Clomifert, Clomihexal, Clomiphen, Clomiphene, Clomiphene Citrate, Cloninn, Clostil, Clostilbegyt, Clovertil, Clovul, Dipthen, Dufine, Duinum, Fensipros, Fertab, Fertec, Fertex, Ferticlo, Fertil, Fertilan, Fertilphen, Fertin, Fertomid, Ferton, Fertotab, Fertyl, Fetrop, Folistim, Genoclom, Genozym, Hete, I-Clom, Ikaclomin, Klofit, Klomen, Klomifen, Lomifen, MER 41, Milophene, Ofertil, Omifin, Ova-mit, Ovamit, Ovinum, Ovipreg, Ovofar, Ovuclon, Ovulet, Pergotime, Pinfetil, Profertil, Prolifen, Provula, Reomen, Serofene, Serophene, Serpafar, Serpafar, Surole, Tocofeno, and Zimaquin.[1]
Regulation
Clomifene is included on the World Anti-Doping Agency list of illegal doping agents in sport.[60] It is listed because it is an "anti-estrogenic substance".
Research
Clomifene has been used almost exclusively for ovulation induction in
Clomifene was studied for treatment and prevention of
References
- ^ a b "International brands of clomifene -". Drugs.com. Archived from the original on 20 September 2016. Retrieved 11 September 2016.
- ^ S2CID 4034257.
- ^ PMID 29159661.
- ^ PMID 3091405.
- ^ a b c d e f g "Clomiphene Citrate". The American Society of Health-System Pharmacists. Archived from the original on 14 September 2017. Retrieved 8 December 2016.
- ^ ISBN 9789241547659.
- ^ a b c d e f g h i "Clomiphene citrate tablets label" (PDF). FDA. Archived (PDF) from the original on September 27, 2016. Retrieved September 11, 2016.
- ISBN 9788132216865. Archivedfrom the original on 2016-12-27.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ PMID 9111183.
- ^ PMID 27337642.
- ^ PMID 23809505.
- ^ PMID 20091498.
- ^ "Clomifene 50mg Tablets - Summary of Product Characteristics (SPC) - (eMC)". UK Electronic Medicines Compendium. Archived from the original on August 22, 2016. Retrieved September 11, 2016.
- ^ PMID 24011610.
- ^ a b Seli E, Arici A. "Ovulation induction with clomiphene citrate". UpToDate. Retrieved 2019-07-30.
- ^ S2CID 79220716.
- ^ S2CID 20304701.
- PMID 28078215.
- S2CID 5538782.
- ISBN 978-0-7817-1750-2.
- ISBN 978-981-10-3694-1.
- S2CID 205834072.
- PMID 30295816.
- ISBN 978-3-642-61013-4.
- S2CID 1240526.
- PMID 29862285.
- ^ a b "ClomiPHENE (Professional Patient Advice)". Drugs.com. Retrieved 2019-07-30.
- ^ PMID 10874566.
- ^ S2CID 40607634.
- S2CID 10136018.
- ISBN 978-3-662-07635-4.
- ^ ISBN 978-1-4963-8587-1.
- ^ S2CID 720243.
- ISBN 9780080548005.
- PMID 10746941.
- PMID 11258977.
- ^ PMID 9586957.
- PMID 3778464.
- ^ PMID 27601965.)
{{cite journal}}: CS1 maint: DOI inactive as of January 2024 (link - ^ ISBN 978-3-319-99350-8.
- ^ ISBN 978-3-0348-0664-0.
- ^ ISBN 978-3-0348-7065-8.
- ISBN 978-0-12-416676-9.
- ISBN 978-0-08-086120-3. Archivedfrom the original on 5 November 2017.
- PMID 15136073.
- ^ Allen, R.E., Palopoli, F.P., Schumann, E.L. and Van Campen, M.G. Jr. (1959) U.S. Patent No. 2,914,563 Archived 2016-10-26 at the Wayback Machine, Nov. 24, 1959.
- ^ PMID 12796359.
- ISBN 978-1-84816-959-3.
- ISBN 978-1-84816-959-3.
- S2CID 1448466.
- )
- ^ Adashi EY (Fall 2014). "Iatrogenic Birth Plurality: The Challenge and Its Possible Solution" (PDF). Harvard Health Policy Review. 14 (1): 9–10. Archived from the original (PDF) on 2016-10-06. Retrieved 2016-09-12.
- ^ a b Lee P (18 July 1989). "Dow Chemical to Get Control of Marion Labs : $5-Billion-Plus Deal Is an Effort to Diversify". Los Angeles Times. Archived from the original on 29 June 2016.
- from the original on 6 October 2016.
- ^ "Hoechst AG to Buy Marion Merrell Dow / Acquisition worth over $7 billion". San Francisco Chronicle. Reuters. May 5, 1995. Archived from the original on October 6, 2016.
- ^ Arturo Bris and Christos Cabolis, Corporate Governance Convergence Through Cross-Border Mergers The Case of Aventis Archived 2014-04-21 at the Wayback Machine, Chapter 4 in Corporate Governance and Regulatory Impact on Mergers and Acquisitions: Research and Analysis on Activity Worldwide Since 1990. Eds Greg N. Gregoriou, Luc Renneboog. Academic Press, 26 July 2007
- ^ Timmons H, Bennhold K (27 April 2004). "France Helped Broker the Aventis-Sanofi Deal". The New York Times. Archived from the original on 5 November 2017.
- ISBN 978-1-4557-2758-2. Archivedfrom the original on 5 November 2017.
- ^ The WADA Prohibited List 2016 (listed as clomiphene) Archived 2016-03-06 at the Wayback Machine
- S2CID 29053109.
- PMID 23062036.
- ISBN 978-0-12-416676-9. Archivedfrom the original on 5 November 2017.
- ISSN 2296-6064.
External links
- "Clomifene". Drug Information Portal. U.S. National Library of Medicine.
