Cnidaria
| Cnidaria Temporal range: Ediacaran–Present
| |
|---|---|

| |
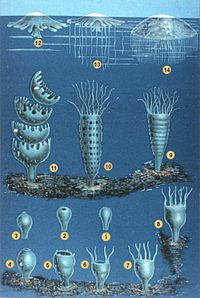
Four examples of cnidaria (clockwise, from top left):
| |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Subkingdom: | Eumetazoa |
| Clade: | ParaHoxozoa |
| Phylum: | Cnidaria Hatschek, 1888 |
| Subphyla and classes[3] | |
| |

Cnidaria (
Cnidarians mostly have two basic body forms: swimming
Cnidarians were formerly grouped with
Most cnidarians prey on
Cnidarians are a very ancient phylum, with fossils having been found in rocks formed about 580 million years ago during the
Etymology
The term cnidaria derives from the Ancient Greek word knídē (κνίδη “nettle”), signifying the coiled thread reminiscent of cnidocytes. The word was first coined in 1766 by the Swedish naturalist Peter Simon Pallas.[10]
Distinguishing features
Cnidarians form a
Like sponges and ctenophores, cnidarians have two main layers of cells that sandwich a middle layer of jelly-like material, which is called the
| Sponges[12]: 76–97 [14] | Cnidarians[11][12] | : 182–195 | Bilateria[11] | |
|---|---|---|---|---|
| Cnidocytes | No | Yes | No | |
| Colloblasts | No | Yes | No | |
organs
|
No | Yes | ||
| Number of main cell layers | Two, with jelly-like layer between them | Two | Two | Three |
| Cells in each layer bound together | cell-adhesion molecules, but no basement membranes except Homoscleromorpha.[15] |
inter-cell connections; basement membranes | ||
Sensory organs
|
No | Yes | ||
| Number of cells in middle "jelly" layer | Many | Few | (Not applicable) | |
| Cells in outer layers can move inwards and change functions | Yes | No | (Not applicable) | |
| Nervous system | No | Yes, simple | Simple to complex | |
| Muscles | None | Mostly epitheliomuscular | Mostly myoepithelial | Mostly myocytes
|
Description
Basic body forms
Most adult cnidarians appear as either free-swimming
Both are
Most have fringes of tentacles equipped with cnidocytes around their edges, and medusae generally have an inner ring of tentacles around the mouth. Some hydroids may consist of colonies of zooids that serve different purposes, such as defense, reproduction and catching prey. The mesoglea of polyps is usually thin and often soft, but that of medusae is usually thick and springy, so that it returns to its original shape after muscles around the edge have contracted to squeeze water out, enabling medusae to swim by a sort of jet propulsion.[12]
Skeletons
In medusae, the only supporting structure is the mesoglea. Hydra and most sea anemones close their mouths when they are not feeding, and the water in the digestive cavity then acts as a hydrostatic skeleton, rather like a water-filled balloon. Other polyps such as Tubularia use columns of water-filled cells for support. Sea pens stiffen the mesoglea with calcium carbonate spicules and tough fibrous proteins, rather like sponges.[12]
In some colonial polyps, a chitinous epidermis gives support and some protection to the connecting sections and to the lower parts of individual polyps. A few polyps collect materials such as sand grains and shell fragments, which they attach to their outsides. Some colonial sea anemones stiffen the mesoglea with sediment particles.[12]
A mineralized exoskeleton made of calcium carbonate is found in subphylum Anthozoa in the order Scleractinia (stony corals; class Hexacorallia) and the class Octocorallia,[16] and in subphylum Medusozoa in three hydrozoan families in order Anthoathecata; Milleporidae, Stylasteridae and Hydractiniidae (the latter with a mix of calcified and uncalcified species).[17]
Main cell layers
Cnidaria are
- Epitheliomuscular cells whose bodies form part of the epithelium but whose bases extend to form muscle fibers in parallel rows.[12]: 103–104 The fibers of the outward-facing cell layer generally run at right angles to the fibers of the inward-facing one. In Anthozoa (anemones, corals, etc.) and Scyphozoa (jellyfish), the mesoglea also contains some muscle cells.[12]
- Cnidocytes, the harpoon-like "nettle cells" that give the phylum Cnidaria its name. These appear between or sometimes on top of the muscle cells.[11]
- Nerve cells. Sensory cells appear between or sometimes on top of the muscle cells,[11] and communicate via synapses (gaps across which chemical signals flow) with motor nerve cells, which lie mostly between the bases of the muscle cells.[12] Some form a simple nerve net.
- Interstitial cells, which are unspecialized and can replace lost or damaged cells by transforming into the appropriate types. These are found between the bases of muscle cells.[11]
In addition to epitheliomuscular, nerve and interstitial cells, the inward-facing gastroderm ("stomach skin") contains gland cells that secrete digestive enzymes. In some species it also contains low concentrations of cnidocytes, which are used to subdue prey that is still struggling.[11][12]
The mesoglea contains small numbers of amoeba-like cells,[12] and muscle cells in some species.[11] However, the number of middle-layer cells and types are much lower than in sponges.[12]
Polymorphism
Cnidocytes
These "nettle cells" function as harpoons, since their payloads remain connected to the bodies of the cells by threads. Three types of cnidocytes are known:[11][12]

Operculum (lid)
"Finger" that turns inside out
/ / / Barbs
Venom
Victim's skin
Victim's tissues
- Spirocysts do not penetrate the victim or inject venom, but entangle it by means of small sticky hairs on the thread.
- Ceriantharia, tube anemones.[12]
The main components of a cnidocyte are:[11][12]

"trigger" cilium[12]
- A cilium (fine hair) which projects above the surface and acts as a trigger. Spirocysts do not have cilia.
- A tough capsule, the )
- A tube-like extension of the wall of the cnida that points into the cnida, like the finger of a rubber glove pushed inwards. When a cnidocyte fires, the finger pops out. If the cell is a venomous nematocyte, the "finger"'s tip reveals a set of barbs that anchor it in the prey.
- The thread, which is an extension of the "finger" and coils round it until the cnidocyte fires. The thread is usually hollow and delivers chemicals from the cnida to the target.
- An operculum (lid) over the end of the cnida. The lid may be a single hinged flap or three flaps arranged like slices of pie.
- The cell body, which produces all the other parts.
It is difficult to study the firing mechanisms of cnidocytes as these structures are small but very complex. At least four hypotheses have been proposed:[11]
- Rapid contraction of fibers round the cnida may increase its internal pressure.
- The thread may be like a coiled spring that extends rapidly when released.
- In the case of Chironex (the "sea wasp"), chemical changes in the cnida's contents may cause them to expand rapidly by polymerization.
- Chemical changes in the liquid in the cnida make it a much more
Cnidocytes can only fire once, and about 25% of a hydra's nematocysts are lost from its tentacles when capturing a brine shrimp. Used cnidocytes have to be replaced, which takes about 48 hours. To minimise wasteful firing, two types of stimulus are generally required to trigger cnidocytes: nearby sensory cells detect chemicals in the water, and their cilia respond to contact. This combination prevents them from firing at distant or non-living objects. Groups of cnidocytes are usually connected by nerves and, if one fires, the rest of the group requires a weaker minimum stimulus than the cells that fire first.[11][12]
Locomotion
Medusae swim by a form of jet propulsion: muscles, especially inside the rim of the bell, squeeze water out of the cavity inside the bell, and the springiness of the mesoglea powers the recovery stroke. Since the tissue layers are very thin, they provide too little power to swim against currents and just enough to control movement within currents.[12]
Hydras and some sea anemones can move slowly over rocks and sea or stream beds by various means: creeping like snails, crawling like inchworms, or by somersaulting. A few can swim clumsily by waggling their bases.[12]
Nervous system and senses
Cnidarians are generally thought to have no brains or even central nervous systems. However, they do have integrative areas of neural tissue that could be considered some form of centralization. Most of their bodies are innervated by decentralized nerve nets that control their swimming musculature and connect with sensory structures, though each clade has slightly different structures.[21] These sensory structures, usually called rhopalia, can generate signals in response to various types of stimuli such as light, pressure, chemical changes, and much more. Medusa usually have several of them around the margin of the bell that work together to control the motor nerve net, that directly innervates the swimming muscles. Most cnidarians also have a parallel system. In scyphozoans, this takes the form of a diffuse nerve net, which has modulatory effects on the nervous system.[22] As well as forming the "signal cables" between sensory neurons and motoneurons, intermediate neurons in the nerve net can also form ganglia that act as local coordination centers. Communication between nerve cells can occur by chemical synapses or gap junctions in hydrozoans, though gap junctions are not present in all groups. Cnidarians have many of the same neurotransmitters as bilaterians, including chemicals such as glutamate, GABA, and acetylcholine.[23] Serotonin, dopamine, noradrenaline, octopamine, histamine, and acetylcholine, on the other hand, are absent.[24]
This structure ensures that the musculature is excited rapidly and simultaneously, and can be directly stimulated from any point on the body, and it also is better able to recover after injury.[21][22]
Medusae and complex swimming colonies such as
Feeding and excretion
Cnidarians feed in several ways:
Predatory species use their
Once the food is in the digestive cavity, gland cells in the gastroderm release enzymes that reduce the prey to slurry, usually within a few hours. This circulates through the digestive cavity and, in colonial cnidarians, through the connecting tunnels, so that gastroderm cells can absorb the nutrients. Absorption may take a few hours, and digestion within the cells may take a few days. The circulation of nutrients is driven by water currents produced by cilia in the gastroderm or by muscular movements or both, so that nutrients reach all parts of the digestive cavity.[12] Nutrients reach the outer cell layer by diffusion or, for animals or zooids such as medusae which have thick mesogleas, are transported by mobile cells in the mesoglea.[11]
Indigestible remains of prey are expelled through the mouth. The main waste product of cells' internal processes is ammonia, which is removed by the external and internal water currents.[12]
Respiration
There are no respiratory organs, and both cell layers absorb oxygen from and expel carbon dioxide into the surrounding water. When the water in the digestive cavity becomes stale it must be replaced, and nutrients that have not been absorbed will be expelled with it. Some Anthozoa have ciliated grooves on their tentacles, allowing them to pump water out of and into the digestive cavity without opening the mouth. This improves respiration after feeding and allows these animals, which use the cavity as a hydrostatic skeleton, to control the water pressure in the cavity without expelling undigested food.[11]
Cnidaria that carry
Regeneration
All cnidarians can regenerate, allowing them to recover from injury and to reproduce asexually. Medusae have limited ability to regenerate, but polyps can do so from small pieces or even collections of separated cells. This enables corals to recover even after apparently being destroyed by predators.[11]
Reproduction
Sexual
Cnidarian
This phenomenon of succession of differently organized generations (one asexually reproducing, sessile polyp, followed by a
Shortened forms of this life cycle are common, for example some oceanic scyphozoans omit the polyp stage completely, and cubozoan polyps produce only one medusa. Hydrozoa have a variety of life cycles. Some have no polyp stages and some (e.g. hydra) have no medusae. In some species, the medusae remain attached to the polyp and are responsible for sexual reproduction; in extreme cases these reproductive zooids may not look much like medusae. Meanwhile, life cycle reversal, in which polyps are formed directly from medusae without the involvement of sexual reproduction process, was observed in both Hydrozoa (Turritopsis dohrnii[27] and Laodicea undulata[28]) and Scyphozoa (Aurelia sp.1[29]). Anthozoa have no medusa stage at all and the polyps are responsible for sexual reproduction.[11]
Spawning is generally driven by environmental factors such as changes in the water temperature, and their release is triggered by lighting conditions such as sunrise, sunset or the phase of the moon. Many species of Cnidaria may spawn simultaneously in the same location, so that there are too many ova and sperm for predators to eat more than a tiny percentage — one famous example is the Great Barrier Reef, where at least 110 corals and a few non-cnidarian invertebrates produce enough gametes to turn the water cloudy. These mass spawnings may produce hybrids, some of which can settle and form polyps, but it is not known how long these can survive. In some species the ova release chemicals that attract sperm of the same species.[11]
The fertilized eggs develop into larvae by dividing until there are enough cells to form a hollow sphere (
Anthozoan larvae either have large
Asexual
All known cnidaria can reproduce asexually by various means, in addition to regenerating after being fragmented. Hydrozoan polyps only bud, while the medusae of some hydrozoans can divide down the middle. Scyphozoan polyps can both bud and split down the middle. In addition to both of these methods, Anthozoa can split horizontally just above the base. Asexual reproduction makes the daughter cnidarian a clone of the adult. The ability of Cnidarians to asexually reproduce ensures a greater number of mature medusa that can mature to reproduce sexually.[11][12]
DNA repair
Two classical DNA repair pathways, nucleotide excision repair and base excision repair, are present in hydra,[30] and these repair pathways facilitate unhindered reproduction. The identification of these pathways in hydra is based, in part, on the presence in the hydra genome of genes homologous to genes in other genetically well studied species that have been demonstrated to play key roles in these DNA repair pathways.[30]
Classification
Cnidarians were for a long time grouped with
| Hydrozoa | Scyphozoa | Cubozoa |
Anthozoa | Myxozoa | |
|---|---|---|---|---|---|
| Number of species[32] | 3,600 | 228 | 42 | 6,100 | 1300 |
| Examples | siphonophores |
Jellyfish | Box jellies |
Sea anemones, corals, sea pens | Myxobolus cerebralis |
| Cells found in mesoglea | No | Yes | Yes | Yes | |
Nematocysts in exodermis
|
No | Yes | Yes | Yes | |
| Medusa phase in life cycle | In some species | Yes | Yes | No | |
| Number of medusae produced per polyp | Many | Many | One | (not applicable) |
Stauromedusae, small
The
Some researchers classify the extinct conulariids as cnidarians, while others propose that they form a completely separate phylum.[38]
Current classification according to the World Register of Marine Species:
- class Anthozoa Ehrenberg, 1834
- subclass CerianthariaPerrier, 1893 — Tube-dwelling anemones
- subclass Hexacorallia Haeckel, 1896 — stony corals
- subclass Octocorallia Haeckel, 1866 — soft corals and sea fans
- subclass
- class CubozoaWerner, 1973 — box jellies
- class Hydrozoa Owen, 1843 — hydrozoans (fire corals, hydroids, hydroid jellyfishes, siphonophores...)
- class Myxozoa Grassé, 1970 — obligate parasites
- class PolypodiozoaRaikova, 1994 — (uncertain status)
- class Scyphozoa Goette, 1887 — "true" jellyfishes
- class Staurozoa Marques & Collins, 2004 — stalked jellyfishes
-
Ceriantharia)
-
Sea anemones (Actinaria, part of Hexacorallia)
-
Box jellyfishCubozoa)
-
SiphonophorePhysalia physalis (Hydrozoa)
-
Polypodiozoa)
-
Jellyfish Phyllorhiza punctata (Scyphozoa)
-
Stalked jelly Haliclystus antarcticus (Staurozoa)
Ecology
Many cnidarians are limited to shallow waters because they depend on
Prey of cnidarians ranges from plankton to animals several times larger than themselves.
Evolutionary history


Fossil record
The earliest widely accepted animal fossils are rather modern-looking cnidarians, possibly from around 580 million years ago, although fossils from the Doushantuo Formation can only be dated approximately.[49] The identification of some of these as embryos of animals has been contested, but other fossils from these rocks strongly resemble tubes and other mineralized structures made by corals.[50] Their presence implies that the cnidarian and bilaterian lineages had already diverged.[51] Although the Ediacaran fossil Charnia used to be classified as a jellyfish or sea pen,[52] more recent study of growth patterns in Charnia and modern cnidarians has cast doubt on this hypothesis,[53][54] leaving the Canadian polyp Haootia and the British Auroralumina as the only recognized cnidarian body fossils from the Ediacaran. Auroralumina is the earliest known animal predator.[55] Few fossils of cnidarians without mineralized skeletons are known from more recent rocks, except in lagerstätten that preserved soft-bodied animals.[56]
A few mineralized fossils that resemble
Family tree
It is difficult to reconstruct the early stages in the

It is now generally thought that the
Within the Cnidaria, the Anthozoa (sea anemones and corals) are regarded as the sister-group of the rest, which suggests that the earliest cnidarians were
However, in 2005 Katja Seipel and Volker Schmid suggested that cnidarians and ctenophores are simplified descendants of
In molecular phylogenetics analyses from 2005 onwards, important groups of developmental genes show the same variety in cnidarians as in chordates.[65] In fact cnidarians, and especially anthozoans (sea anemones and corals), retain some genes that are present in bacteria, protists, plants and fungi but not in bilaterians.[66]
The mitochondrial genome in the medusozoan cnidarians, unlike those in other animals, is linear with fragmented genes.[67] The reason for this difference is unknown.
Interaction with humans

Jellyfish stings killed about 1,500 people in the 20th century,[68] and cubozoans are particularly dangerous. On the other hand, some large jellyfish are considered a delicacy in East and Southeast Asia. Coral reefs have long been economically important as providers of fishing grounds, protectors of shore buildings against currents and tides, and more recently as centers of tourism. However, they are vulnerable to over-fishing, mining for construction materials, pollution, and damage caused by tourism.
Beaches protected from tides and storms by coral reefs are often the best places for housing in tropical countries. Reefs are an important food source for low-technology fishing, both on the reefs themselves and in the adjacent seas.
Some large
The "sea wasp"
A number of the parasitic
Notes
- ^ Classes in Medusozoa based on "ITIS Report – Taxon: Subphylum Medusozoa". Universal Taxonomic Services. Retrieved 2018-03-18.
- ^ S2CID 11108911.
- ^ Subphyla Anthozoa and Medusozoa based on "The Taxonomicon – Taxon: Phylum Cnidaria". Universal Taxonomic Services. Archived from the original on 2007-09-29. Retrieved 2007-07-10.
- ^ "cnidaria". Oxford English Dictionary (Online ed.). Oxford University Press. (Subscription or participating institution membership required.)
- ^ "WoRMS - World Register of Marine Species". www.marinespecies.org. Retrieved 2018-12-17.
- PMID 25840473.
- ^ S2CID 5170702.
- PMID 22040765.
- PMID 26465609.
- ^ "Cnidaria - etymonline.com".
- ^ ISBN 978-0-19-551368-4.
- ^ ISBN 978-0-03-025982-1.
- ^ PMID 15936326.
- ISBN 978-0-19-551368-4.
- S2CID 12376172.
- ^ Comparative Proteomics of Octocoral and Scleractinian Skeletomes and the Evolution of Coral Calcification
- ^ Evolution of Calcium-carbonate Skeletons in the Hydractiniidae
- )
- S2CID 278540.
- ^ Trumble, W.; Brown, L. (2002). "Cnida". Shorter Oxford English Dictionary. Oxford University Press.
- ^ PMID 21430196.
- ^ S2CID 18244609.
- PMID 17101286.
- ^ What is a neuron? (Ctenophores, sponges and placozoans)
- ^ a b "Jellyfish Have Human-Like Eyes". www.livescience.com. April 1, 2007. Retrieved 2012-06-12.
- ISBN 9780412337604.
- ^ Bavestrello; et al. (1992). "Bi-directional conversion in Turritopsis nutricula (Hydrozoa)" (PDF). Scientia Marina. Archived from the original (PDF) on 2014-12-14. Retrieved 2015-12-31.
- S2CID 84325535.
- PMID 26690755.
- ^ PMID 33995496.
- ^ a b c Schuster, Ruth (20 November 2015). "Microscopic parasitic jellyfish defy everything we know, astonish scientists". Haaretz. Retrieved 4 April 2018.
- (PDF) from the original on 2012-01-12.
- PMID 21676805.
- ^ Štolc, A. (1899). "Actinomyxidies, nouveau groupe de Mesozoaires parent des Myxosporidies". Bull. Int. l'Acad. Sci. Bohème. 12: 1–12.
- S2CID 221583517.
- S2CID 5170702.
- PMID 26627241.
- ^ "The Conulariida". University of California Museum of Paleontology. Retrieved 2008-11-27.
- ^ ISBN 978-0470016176.
- ISBN 978-1-4020-3119-9. Retrieved 2008-11-21.
- ^ ISBN 978-0-8050-8318-7. Retrieved 2008-11-21.
- ISBN 978-0-632-04444-3. Retrieved 2008-11-21.
- PMID 29661512.
- PMID 19269306.
- ^ Frick, K (2003). "Predator Suites and Flabellinid Nudibranch Nematocyst Complements in the Gulf of Maine". In: SF Norton (Ed). Diving for Science...2003. Proceedings of the American Academy of Underwater Sciences (22nd Annual Scientific Diving Symposium). Archived from the original on 2011-01-06. Retrieved 2008-07-03.
{{cite journal}}: CS1 maint: unfit URL (link) - ISBN 978-0-12-547665-2.
- ^ ISBN 978-0-632-02983-9. Retrieved 2008-11-26.
- ISBN 978-0-08-037718-6. Retrieved 2008-11-21.
- PMID 10781044.
- PMID 11095754.
- PMID 12142030. Archived from the original(PDF) on 2008-09-11. Retrieved 2008-09-03.
- S2CID 128395097.
- S2CID 130602154.
- S2CID 83486435.
- ^ Amos, Jonathan (25 July 2022). "Ancient fossil is earliest known animal predator". bbc.co.uk. BBC News. Retrieved 25 July 2022.
- ^ a b c d "Cnidaria: Fossil Record". University of California Museum of Paleontology. Retrieved 2008-11-27.
- S2CID 42938715.
- ^ "The Rudists". University of California Museum of Paleontology. Retrieved 2008-11-27.
- doi:10.1146/annurev.ecolsys.35.112202.130124. Archived from the original(PDF) on 2008-10-07. Retrieved 2008-11-27.
- S2CID 25119754.
- PMID 11504944.
- (PDF) from the original on 2018-07-26.
- S2CID 86185156.
- . Retrieved 2008-11-27.
- PMID 16098631.
- PMID 16226338.
- PMID 22117085.
- ISBN 978-0-86840-279-6. Retrieved 2008-10-03.
- ^ ISBN 978-0-632-04955-4. Retrieved 2008-11-28.
Coral Reef productivity.
- ISBN 978-0-412-29270-5. Retrieved 2008-11-28.
- ISSN 1880-8247.
- ^ Omori, M.; Kitamura, M. (2004). "Taxonomic review of three Japanese species of edible jellyfish (Scyphozoa: Rhizostomeae)". Plankton Biol. Ecol. 51 (1): 36–51.
- ^ S2CID 6518460.
- S2CID 20719121.
- ISBN 978-0-7817-4586-4.
- ^ PMID 16687419.
- PMID 14172390.
- PMID 14634600.
- PMID 28993459.
- ^ Irish Times. Retrieved 2022-04-23.
Further reading
Books
- Arai, M.N. (1997). A Functional Biology of Scyphozoa. London: Chapman & Hall [p. 316]. ISBN 0-412-45110-7.
- Ax, P. (1999). Das System der Metazoa I. Ein Lehrbuch der phylogenetischen Systematik. Gustav Fischer, Stuttgart-Jena: Gustav Fischer. ISBN 3-437-30803-3.
- Barnes, R.S.K., P. Calow, P. J. W. Olive, D. W. Golding & J. I. Spicer (2001). The invertebrates—a synthesis. Oxford: Blackwell. 3rd edition [chapter 3.4.2, p. 54]. ISBN 0-632-04761-5.
- Brusca, R.C., G.J. Brusca (2003). Invertebrates. Sunderland, Mass.: Sinauer Associates. 2nd edition [chapter 8, p. 219]. ISBN 0-87893-097-3.
- Dalby, A. (2003). Food in the Ancient World: from A to Z. London: Routledge.
- Moore, J.(2001). An Introduction to the Invertebrates. Cambridge: Cambridge University Press [chapter 4, p. 30]. ISBN 0-521-77914-6.
- Schäfer, W. (1997). Cnidaria, Nesseltiere. In Rieger, W. (ed.) Spezielle Zoologie. Teil 1. Einzeller und Wirbellose Tiere. Stuttgart-Jena: Gustav Fischer. Spektrum Akademischer Verl., Heidelberg, 2004. ISBN 3-8274-1482-2.
- Werner, B. 4. Stamm Cnidaria. In: V. Gruner (ed.) Lehrbuch der speziellen Zoologie. Begr. von Kaestner. 2 Bde. Stuttgart-Jena: Gustav Fischer, Stuttgart-Jena. 1954, 1980, 1984, Spektrum Akad. Verl., Heidelberg-Berlin, 1993. 5th edition. ISBN 3-334-60474-8.
Journal articles
- D. Bridge, B. Schierwater, C. W. Cunningham, R. DeSalle R, L. W. Buss: Mitochondrial DNA structure and the molecular phylogeny of recent cnidaria classes. in: Proceedings of the Academy of Natural Sciences of Philadelphia. Philadelphia USA 89.1992, p. 8750. ISSN 0097-3157
- D. Bridge, C. W. Cunningham, R. DeSalle, L. W. Buss: Class-level relationships in the phylum Cnidaria—Molecular and morphological evidence. in: Molecular biology and evolution. Oxford University Press, Oxford 12.1995, p. 679. ISSN 0737-4038
- D. G. Fautin: Reproduction of CnidariaISSN 0008-4301
- G. O. Mackie: What's new in cnidarian biology? Archived 2019-02-18 at the ISSN 0008-4301
- P. Schuchert: Phylogenetic analysis of the Cnidaria. in: Zeitschrift für zoologische Systematik und Evolutionsforschung. Paray, Hamburg-Berlin 31.1993, p. 161. ISSN 0044-3808
- G. Kass-Simon, A. A. Scappaticci Jr.: The behavioral and developmental physiology of nematocysts.ISSN 0044-3808
- J. Zrzavý (2001). "The interrelationships of metazoan parasites: a review of phylum- and higher-level hypotheses from recent morphological and molecular phylogenetic analyses". Folia Parasitologica. 48 (2): 81–103. PMID 11437135.
External links
- YouTube: Nematocysts Firing
- YouTube:My Anemone Eat Meat Defensive and feeding behaviour of sea anemone
- Cnidaria - Guide to the Marine Zooplankton of south eastern Australia, Tasmanian Aquaculture & Fisheries Institute
- Cnidaria page at Tree of Life
- Fossil Gallery: Cnidarians
- Hexacorallians of the World