Cobicistat
 | |
| Clinical data | |
|---|---|
| Pronunciation | /koʊˈbɪsɪstæt/ koh-BIS-i-stat |
| Trade names | Tybost |
| Other names | GS-9350 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a616029 |
| License data |
|
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Cobicistat, sold under the brand name Tybost, is a medication for use in the treatment of
Like ritonavir (Norvir), cobicistat is of interest for its ability to inhibit liver enzymes that metabolize other medications used to treat HIV, notably elvitegravir, an HIV integrase inhibitor. By combining cobicistat with elvitegravir, higher concentrations of the latter are achieved in the body with lower dosing, theoretically enhancing elvitegravir's viral suppression while diminishing its adverse side-effects. In contrast with ritonavir, the only other booster approved for use as a part of HAART, cobicistat has no anti-HIV activity of its own.[2]
Cobicistat is a component of three four-drug, fixed-dose combination HIV treatments. The first,
Additionally, in existence are a fixed-dose combination of cobicistat and
Cobicistat is a potent inhibitor of cytochrome P450 3A enzymes, including the important CYP3A4 subtype. It also inhibits intestinal transport proteins, increasing the overall absorption of several HIV medications, including atazanavir, darunavir, and tenofovir alafenamide.[5]
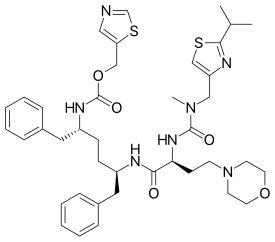
Chemistry
Cobicistat is a drug analogue of ritonavir, in which the valine moiety is exchanged for a 2-morpholinoethyl group, and the backbone hydroxyl group is removed. These changes effectively eliminate the anti-HIV activity of ritonavir while preserving its inhibitory effects on the CYP3A isozyme family of proteins.[6] Cobicistat is therefore able to increase plasma concentration of other coadministered anti-HIV drugs without the risk of causing cobicistat-resistant mutations in the HIV virus.
Synthesis
Cobicistat may be synthesized from any number of commercially available starting materials. The synthesis shown below utilizes
Discovery and development
Cobicistat was developed through
References
- S2CID 29197109.
- ^ a b Highleyman, L. Elvitegravir "Quad" Single-tablet Regimen Shows Continued HIV Suppression at 48 Weeks. HIV and Hepatitis.com
- ^ R Elion, J Gathe, B Rashbaum, and others. The Single-Tablet Regimen of Elvitegravir/Cobicistat/Emtricitabine/Tenofovir Disoproxil Fumarate (EVG/COBI/FTC/TDF; Quad) Maintains a High Rate of Virologic Suppression, and Cobicistat (COBI) is an Effective Pharmacoenhancer Through 48 Weeks. 50th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC 2010). Boston, September 12–15, 2010.
- ^ "Symtuza (cobicistat, darunavir, emtricitabine and tenofovir alafenamide) FDA Approval History". Drugs.com. Retrieved 30 October 2019.
- PMID 22850510.
- PMID 24900196.
- ^ "WO 2016128885" (PDF). Archived from the original (PDF) on 14 August 2019. Retrieved 31 August 2017.
- PMID 24412072.
- PMID 23746300.
- PMID 20937904.
- ^ PDB 3NXU
