Coenzyme A

| |

| |

| |
| Names | |
|---|---|
| Systematic IUPAC name
[(2R,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)tetrahydro-2-furanyl]methyl (3R)-3-hydroxy-2,2-dimethyl-4-oxo-4-({3-oxo-3-[(2-sulfanylethyl)amino]propyl}amino)butyl dihydrogen diphosphate | |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.001.472 |
| KEGG | |
| MeSH | Coenzyme+A |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C21H36N7O16P3S | |
| Molar mass | 767.535 |
| UV-vis (λmax) | 259.5 nm[1] |
| Absorbance | ε259 = 16.8 mM−1 cm−1[1]
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Coenzyme A (CoA, SHCoA, CoASH) is a
In
Discovery of structure

Coenzyme A was identified by
Biosynthesis
Coenzyme A is naturally synthesized from pantothenate (vitamin B5), which is found in food such as meat, vegetables, cereal grains, legumes, eggs, and milk.[10] In humans and most living organisms, pantothenate is an essential vitamin that has a variety of functions.[11] In some plants and bacteria, including Escherichia coli, pantothenate can be synthesised de novo and is therefore not considered essential. These bacteria synthesize pantothenate from the amino acid aspartate and a metabolite in valine biosynthesis.[12]
In all living organisms, coenzyme A is synthesized in a five-step process that requires four molecules of ATP, pantothenate and cysteine[13] (see figure):

- Pantothenate (vitamin B5) is phosphorylated to 4′-phosphopantothenate by the enzyme pantothenate kinase (PanK; CoaA; CoaX). This is the committed step in CoA biosynthesis and requires ATP.[12]
- A phosphopantothenoylcysteine synthetase (PPCS; CoaB) to form 4'-phospho-N-pantothenoylcysteine (PPC). This step is coupled with ATP hydrolysis.[12]
- PPC is decarboxylated to 4′-phosphopantetheine by phosphopantothenoylcysteine decarboxylase(PPC-DC; CoaC)
- 4′-phosphopantetheine is adenylated (or more properly, AMPylated) to form dephospho-CoA by the enzyme phosphopantetheine adenylyl transferase(COASY; PPAT; CoaD)
- Finally, dephospho-CoA is phosphorylated to coenzyme A by the enzyme dephosphocoenzyme A kinase (COASY, DPCK; CoaE). This final step requires ATP.[12]
Enzyme nomenclature abbreviations in parentheses represent mammalian, other eukaryotic, and prokaryotic enzymes respectively. In mammals steps 4 and 5 are catalyzed by a bifunctional enzyme called COASY.[14] This pathway is regulated by product inhibition. CoA is a competitive inhibitor for Pantothenate Kinase, which normally binds ATP.[12] Coenzyme A, three ADP, one monophosphate, and one diphosphate are harvested from biosynthesis.[13]
Coenzyme A can be synthesized through alternate routes when intracellular coenzyme A level are reduced and the de novo pathway is impaired.
A 2024 article detailed a plausible chemical synthesis mechanism for the pantetheine component (the main functional part) of coenzyme A in a primordial prebiotic world.
Commercial production
Coenzyme A is produced commercially via extraction from yeast, however this is an inefficient process (yields approximately 25 mg/kg) resulting in an expensive product. Various ways of producing CoA synthetically, or semi-synthetically have been investigated although none are currently operating at an industrial scale.[17]
Function
Fatty acid synthesis
Since coenzyme A is, in chemical terms, a
Coenzyme A is also the source of the phosphopantetheine group that is added as a prosthetic group to proteins such as acyl carrier protein and formyltetrahydrofolate dehydrogenase.[18][19]
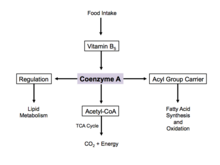
Energy production
Coenzyme A is one of five crucial coenzymes that are necessary in the reaction mechanism of the citric acid cycle. Its acetyl-coenzyme A form is the primary input in the citric acid cycle and is obtained from glycolysis, amino acid metabolism, and fatty acid beta oxidation. This process is the body's primary catabolic pathway and is essential in breaking down the building blocks of the cell such as carbohydrates, amino acids, and lipids.[20]
Regulation
When there is excess glucose, coenzyme A is used in the cytosol for synthesis of fatty acids.
During cell starvation, coenzyme A is synthesized and transports fatty acids in the cytosol to the mitochondria. Here, acetyl-CoA is generated for oxidation and energy production.[21] In the citric acid cycle, coenzyme A works as an allosteric regulator in the stimulation of the enzyme pyruvate dehydrogenase.
Antioxidant function and regulation
Discovery of the novel antioxidant function of coenzyme A highlights its protective role during cellular stress. Mammalian and Bacterial cells subjected to oxidative and metabolic stress show significant increase in the covalent modification of protein cysteine residues by coenzyme A.[23][24] This reversible modification is termed protein CoAlation (Protein-S-SCoA), which plays a similar role to protein S-glutathionylation by preventing the irreversible oxidation of the thiol group of cysteine residues.
Using anti-coenzyme A antibody[25] and liquid chromatography tandem mass spectrometry (LC-MS/MS) methodologies, more than 2,000 CoAlated proteins were identified from stressed mammalian and bacterial cells.[26] The majority of these proteins are involved in cellular metabolism and stress response.[26] Different research studies have focused on deciphering the coenzyme A-mediated regulation of proteins. Upon protein CoAlation, inhibition of the catalytic activity of different proteins (e.g. metastasis suppressor NME1, peroxiredoxin 5, GAPDH, among others) is reported.[27][28][24][29] To restore the protein's activity, antioxidant enzymes that reduce the disulfide bond between coenzyme A and the protein cysteine residue play an important role. This process is termed protein deCoAlation. So far, two bacterial proteins, Thioredoxin A and Thioredoxin-like protein (YtpP), are shown to deCoAlate proteins.[30]
Use in biological research
Coenzyme A is available from various chemical suppliers as the free acid and
Non-exhaustive list of coenzyme A-activated acyl groups
- Acetyl-CoA
- esterification)
- Propionyl-CoA
- Butyryl-CoA
- Myristoyl-CoA
- Crotonyl-CoA
- Acetoacetyl-CoA
- Coumaroyl-CoA (used in flavonoid and stilbenoidbiosynthesis)
- Benzoyl-CoA
- Phenylacetyl-CoA
- Acyl derived from dicarboxylic acids
- fatty acid biosynthesis and polyketidebiosynthesis)
- Succinyl-CoA (used in heme biosynthesis)
- isoprenoidbiosynthesis)
- Pimelyl-CoA (used in biotinbiosynthesis)
References
- ^ ISBN 978-0-19-855299-4.
- PMID 11923312.
- ^ "Coenzyme A: when small is mighty". www.asbmb.org. Archived from the original on 2018-12-20. Retrieved 2018-12-19.
- .
- S2CID 630898.
- ^ ISSN 0021-9258. Archived from the originalon 2019-04-12. Retrieved 2017-10-24.
- PMID 20287921.
- PMID 14778827.
- ^ "Fritz Lipmann – Facts". Nobelprize.org. Nobel Media AB. 2014. Retrieved 8 November 2017.
- ^ "Vitamin B5 (Pantothenic acid)". University of Maryland Medical Center. Archived from the original on 2017-10-18. Retrieved 2017-11-08.
- ^ "Pantothenic Acid (Vitamin B5): MedlinePlus Supplements". medlineplus.gov. Archived from the original on 2017-12-22. Retrieved 2017-12-10.
- ^ PMID 26443589.
- ^ PMID 15893380.
- S2CID 27153945.
- PMID 26379022.
- S2CID 10344527.
- S2CID 92802641.
- PMID 4872726.
- PMID 19933275.
- ^ Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2002). "Chapter 2: How Cells Obtain Energy from Food". Molecular Biology of the Cell (4th ed.). Garland Science.
- ^ PMID 25703630.
- ^ Berg JM, Tymoczko JL, Stryer L (2002). "Acetyl Coenzyme A Carboxylase Plays a Key Role in Controlling Fatty Acid Metabolism". Biochemistry.
- PMID 28341808.
- ^ PMID 29626155.
- ISSN 0233-7657.
- ^ PMID 35883853.
- PMID 33339386.
- PMID 33903070.
- PMID 31375973.
- PMID 37107313.
- ^ "Datasheet for free acid coenzyme A" (PDF). Oriental Yeast Co., LTD.
- ^ "Datasheet for lithium salt coenzyme A" (PDF). Oriental Yeast Co., LTD.
Bibliography
- Nelson DL, Cox MM (2005). Lehninger: Principles of Biochemistry (4th ed.). New York: W .H. Freeman. ISBN 978-0-7167-4339-2.
