Colloid

| Condensed matter physics |
|---|
 |
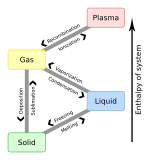
A colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance. Some definitions specify that the particles must be dispersed in a liquid,[1] while others extend the definition to include substances like aerosols and gels. The term colloidal suspension refers unambiguously to the overall mixture (although a narrower sense of the word suspension is distinguished from colloids by larger particle size). A colloid has a dispersed phase (the suspended particles) and a continuous phase (the medium of suspension). The dispersed phase particles have a diameter of approximately 1 nanometre to 1 micrometre.[2][3]
Some colloids are
Colloidal suspensions are the subject of interface and colloid science. This field of study began in 1845 by Francesco Selmi,[4][5][6][7] who called them pseudosolutions, and expanded by Michael Faraday[8] and Thomas Graham, who coined the term colloid in 1861.[9]
Colloid: Short synonym for colloidal system.[10][11]
Colloidal: State of subdivision such that the molecules or polymolecular particles dispersed in a medium have at least one dimension between approximately 1 nm and 1 μm, or that in a system discontinuities are found at distances of that order.[10][11][12]
Classification of colloids
Colloids can be classified as follows:
| Medium/phase | Dispersed phase | |||
|---|---|---|---|---|
| Gas | Liquid | Solid | ||
| Dispersion medium |
Gas | No such colloids are known. Helium and xenon are known to be immiscible under certain conditions.[13][14] |
Liquid aerosol Examples: fog, clouds, condensation, mist, steam, hair sprays |
Solid aerosol Examples: atmospheric particulate matter
|
| Liquid | Foam Example: whipped cream, shaving cream |
biological membranes, liquid biomolecular condensate |
precipitates, solid biomolecular condensate
| |
| Solid | Solid foam Examples: styrofoam, pumice |
Gel Examples: agar, gelatin, jelly, gel-like biomolecular condensate |
Solid sol Example: cranberry glass | |
Homogeneous mixtures with a dispersed phase in this size range may be called colloidal aerosols, colloidal emulsions, colloidal suspensions, colloidal foams, colloidal dispersions, or hydrosols.
-
Aerogel
-
Jello cubes
-
Colloidal silica gel with light opalescence
-
Whipped cream
-
A dollop of hair gel
-
Creams are semi-solid emulsions of oil and water. Oil-in-water creams are used for cosmetic purpose while water-in-oil creams for medicinal purpose
-
Tyndall effect in an opalite:
it scatters blue light making it appear blue from the side, but orange light shines through.
opal is a gel in which water is dispersed in silica crystals -
Mist
Hydrocolloids
Hydrocolloids describe certain
The term hydrocolloids also refers to a type of dressing designed to lock moisture in the skin and help the natural healing process of skin to reduce scarring, itching and soreness.
Components
Hydrocolloids contain some type of gel-forming agent, such as sodium carboxymethylcellulose (NaCMC) and gelatin. They are normally combined with some type of sealant, i.e. polyurethane to 'stick' to the skin.
Colloid compared with solution
A colloid has a
Interaction between particles
The following forces play an important role in the interaction of colloid particles:[17][18]
- Excluded volume repulsion: This refers to the impossibility of any overlap between hard particles.
- Electrostatic interaction: Colloidal particles often carry an electrical charge and therefore attract or repel each other. The charge of both the continuous and the dispersed phase, as well as the mobility of the phases are factors affecting this interaction.
- van der Waals forces: This is due to interaction between two dipoles that are either permanent or induced. Even if the particles do not have a permanent dipole, fluctuations of the electron density gives rise to a temporary dipole in a particle. This temporary dipole induces a dipole in particles nearby. The temporary dipole and the induced dipoles are then attracted to each other. This is known as van der Waals force, and is always present (unless the refractive indexes of the dispersed and continuous phases are matched), is short-range, and is attractive.
- Steric forces between polymer-covered surfaces or in solutions containing non-adsorbing polymer can modulate interparticle forces, producing an additional steric repulsive force (which is predominantly entropic in origin) or an attractive depletion force between them.
Sedimentation velocity

The Earth’s gravitational field acts upon colloidal particles. Therefore, if the colloidal particles are denser than the medium of suspension, they will sediment (fall to the bottom), or if they are less dense, they will cream (float to the top). Larger particles also have a greater tendency to sediment because they have smaller Brownian motion to counteract this movement.
The sedimentation or creaming velocity is found by equating the Stokes drag force with the gravitational force:
where
- is the Archimedean weight of the colloidal particles,
- is the viscosity of the suspension medium,
- is the radius of the colloidal particle,
and is the sedimentation or creaming velocity.
The mass of the colloidal particle is found using:
where
- is the volume of the colloidal particle, calculated using the volume of a sphere ,
and is the difference in mass density between the colloidal particle and the suspension medium.
By rearranging, the sedimentation or creaming velocity is:
There is an upper size-limit for the diameter of colloidal particles because particles larger than 1 μm tend to sediment, and thus the substance would no longer be considered a colloidal suspension.[19]
The colloidal particles are said to be in sedimentation equilibrium if the rate of sedimentation is equal to the rate of movement from Brownian motion.
Preparation
There are two principal ways to prepare colloids:[20]
- spraying, or application of shear (e.g., shaking, mixing, or high shear mixing).
- Condensation of small dissolved molecules into larger colloidal particles by precipitation, condensation, or redox reactions. Such processes are used in the preparation of colloidal silica or gold.
Stabilization
The stability of a colloidal system is defined by particles remaining suspended in solution and depends on the interaction forces between the particles. These include electrostatic interactions and van der Waals forces, because they both contribute to the overall free energy of the system.[21]
A colloid is stable if the interaction energy due to attractive forces between the colloidal particles is less than
If the interaction energy is greater than kT, the attractive forces will prevail, and the colloidal particles will begin to clump together. This process is referred to generally as aggregation, but is also referred to as flocculation, coagulation or precipitation.[22] While these terms are often used interchangeably, for some definitions they have slightly different meanings. For example, coagulation can be used to describe irreversible, permanent aggregation where the forces holding the particles together are stronger than any external forces caused by stirring or mixing. Flocculation can be used to describe reversible aggregation involving weaker attractive forces, and the aggregate is usually called a floc. The term precipitation is normally reserved for describing a phase change from a colloid dispersion to a solid (precipitate) when it is subjected to a perturbation.[19] Aggregation causes sedimentation or creaming, therefore the colloid is unstable: if either of these processes occur the colloid will no longer be a suspension.

Electrostatic stabilization and steric stabilization are the two main mechanisms for stabilization against aggregation.
- Electrostatic stabilization is based on the mutual repulsion of like electrical charges. The charge of colloidal particles is structured in an electrical double layer, where the particles are charged on the surface, but then attract counterions (ions of opposite charge) which surround the particle. The electrostatic repulsion between suspended colloidal particles is most readily quantified in terms of the zeta potential. The combined effect of van der Waals attraction and electrostatic repulsion on aggregation is described quantitatively by the DLVO theory.[23] A common method of stabilising a colloid (converting it from a precipitate) is peptization, a process where it is shaken with an electrolyte.
- Steric stabilization consists absorbing a layer of a polymer or surfactant on the particles to prevent them from getting close in the range of attractive forces.[19] The polymer consists of chains that are attached to the particle surface, and the part of the chain that extends out is soluble in the suspension medium.[24] This technique is used to stabilize colloidal particles in all types of solvents, including organic solvents.[25]
A combination of the two mechanisms is also possible (electrosteric stabilization).

A method called gel network stabilization represents the principal way to produce colloids stable to both aggregation and sedimentation. The method consists in adding to the colloidal suspension a polymer able to form a gel network. Particle settling is hindered by the stiffness of the polymeric matrix where particles are trapped,[26] and the long polymeric chains can provide a steric or electrosteric stabilization to dispersed particles. Examples of such substances are xanthan and guar gum.
Destabilization
Destabilization can be accomplished by different methods:
- Removal of the electrostatic barrier that prevents aggregation of the particles. This can be accomplished by the addition of salt to a suspension to reduce the Debye screening length (the width of the electrical double layer) of the particles. It is also accomplished by changing the pH of a suspension to effectively neutralise the surface charge of the particles in suspension.[1] This removes the repulsive forces that keep colloidal particles separate and allows for aggregation due to van der Waals forces. Minor changes in pH can manifest in significant alteration to the zeta potential. When the magnitude of the zeta potential lies below a certain threshold, typically around ± 5mV, rapid coagulation or aggregation tends to occur.[27]
- Addition of a charged polymer flocculant. Polymer flocculants can bridge individual colloidal particles by attractive electrostatic interactions. For example, negatively charged colloidal silica or clay particles can be flocculated by the addition of a positively charged polymer.
- Addition of non-adsorbed polymers called depletants that cause aggregation due to entropic effects.
Unstable colloidal suspensions of low-volume fraction form clustered liquid suspensions, wherein individual clusters of particles sediment if they are more dense than the suspension medium, or cream if they are less dense. However, colloidal suspensions of higher-volume fraction form colloidal gels with
Monitoring stability

The most widely used technique to monitor the dispersion state of a product, and to identify and quantify destabilization phenomena, is multiple
Dynamic light scattering can be used to detect the size of a colloidal particle by measuring how fast they diffuse. This method involves directing laser light towards a colloid. The scattered light will form an interference pattern, and the fluctuation in light intensity in this pattern is caused by the Brownian motion of the particles. If the apparent size of the particles increases due to them clumping together via aggregation, it will result in slower Brownian motion. This technique can confirm that aggregation has occurred if the apparent particle size is determined to be beyond the typical size range for colloidal particles.[21]
Accelerating methods for shelf life prediction
The kinetic process of destabilisation can be rather long (up to several months or years for some products). Thus, it is often required for the formulator to use further accelerating methods to reach reasonable development time for new product design. Thermal methods are the most commonly used and consist of increasing temperature to accelerate destabilisation (below critical temperatures of phase inversion or chemical degradation). Temperature affects not only viscosity, but also interfacial tension in the case of non-ionic surfactants or more generally interactions forces inside the system. Storing a dispersion at high temperatures enables to simulate real life conditions for a product (e.g. tube of sunscreen cream in a car in the summer), but also to accelerate destabilisation processes up to 200 times. Mechanical acceleration including vibration, centrifugation and agitation are sometimes used. They subject the product to different forces that pushes the particles / droplets against one another, hence helping in the film drainage. Some emulsions would never coalesce in normal gravity, while they do under artificial gravity.[33] Segregation of different populations of particles have been highlighted when using centrifugation and vibration.[34]
As a model system for atoms
In physics, colloids are an interesting model system for atoms.[35] Micrometre-scale colloidal particles are large enough to be observed by optical techniques such as confocal microscopy. Many of the forces that govern the structure and behavior of matter, such as excluded volume interactions or electrostatic forces, govern the structure and behavior of colloidal suspensions. For example, the same techniques used to model ideal gases can be applied to model the behavior of a hard sphere colloidal suspension. Phase transitions in colloidal suspensions can be studied in real time using optical techniques,[36] and are analogous to phase transitions in liquids. In many interesting cases optical fluidity is used to control colloid suspensions.[36][37]
Crystals
A colloidal crystal is a highly
Thus, it has been known for many years that, due to
The large number of experiments exploring the physics and chemistry of these so-called "colloidal crystals" has emerged as a result of the relatively simple methods that have evolved in the last 20 years for preparing synthetic monodisperse colloids (both polymer and mineral) and, through various mechanisms, implementing and preserving their long-range order formation.[43]
In biology
Colloidal
In the environment
Colloidal particles can also serve as transport vector[44] of diverse contaminants in the surface water (sea water, lakes, rivers, fresh water bodies) and in underground water circulating in fissured rocks[45] (e.g.
In soil science, the colloidal fraction in soils consists of tiny clay and humus particles that are less than 1μm in diameter and carry either positive and/or negative electrostatic charges that vary depending on the chemical conditions of the soil sample, i.e. soil pH.[49]
Intravenous therapy
Colloid solutions used in
References
- ^ OCLC 706803091.
- OCLC 406528399.
- S2CID 95122531.
- ^ Selmi, Francesco "Studi sulla dimulsione di cloruro d'argento". Nuovi Annali delle Scienze Naturali di Bologna, 1845.
- ^ Selmi, Francesco, Studio intorno alle pseudo-soluzioni degli azzurri di Prussia ed alla influenza dei sali nel guastarle, Bologna: Tipi Sassi, 1847
- ^ Hatschek, Emil, The Foundations of Colloid Chemistry, A selection of early papers bearing on the subject, The British Association Committee on Colloid Chemistry, London, 1925
- ^ Selmi, Francesco - Sur le soufre pseudosoluble, sa pseudosolution e le soufre mou, Journal de Pharmacie et de Chimie, tome 21, 1852, Paris
- S2CID 55882753.
- S2CID 186208563.. Page 183: "As gelatine appears to be its type, it is proposed to designate substances of the class as colloids, and to speak of their peculiar form of aggregation as the colloidal condition of matter."
- ^ ISBN 978-0-85404-491-7.
- ^ (PDF) from the original on 9 October 2022.
- (PDF) from the original on 9 October 2022.
- .
- .
- PMID 23572691.
- S2CID 221770585.
- ISBN 9789400712225. Archived from the originalon 14 April 2019. Retrieved 5 September 2018.
- PMID 25344532.
- ^ ISBN 9781444320183.
- ^ Kopeliovich, Dmitri. Preparation of colloids. substech.com
- ^ OCLC 232632488.
- S2CID 96812603.
- ISBN 9780123750495.
- OCLC 701308697.
- .
- PMID 19577785.
- JSTOR 41264141.
- PMID 12954183.
- S2CID 24157992.
- .
- ^ Bru, P.; et al. (2004). T. Provder; J. Texter (eds.). Particle sizing and characterisation.
- .
- ISBN 978-0-8247-0304-2.
- PMID 18986182.
- S2CID 5727282.
- ^ PMID 24104290.
- .
- .
- S2CID 4191566.
- .
- .
- .
- PMID 23836297.
- ISBN 978-3-540-71338-8.
- .
- ISSN 1015-2636. Archived from the original(PDF) on 9 March 2009. Retrieved 22 February 2009.
- ^ "Diffusion of colloids in compacted bentonite". Archived from the original on 4 March 2009. Retrieved 12 February 2009.
- .
- OCLC 1035317420.)
{{cite book}}: CS1 maint: location missing publisher (link - ^ a b c Martin, Gregory S. (19 April 2005). "An Update on Intravenous Fluids". Medscape. Retrieved 6 July 2016.
- ^ Blake, Heidi (3 March 2011). "Millions of surgery patients at risk in drug research fraud scandal". The Telegraph. UK. Archived from the original on 4 November 2011. Retrieved 4 November 2011.