Composition of the human body
Body composition may be analyzed in various ways. This can be done in terms of the
Elements
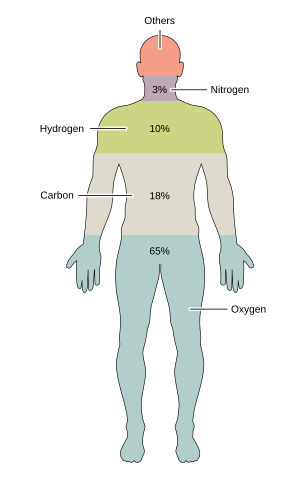
|
Element | Symbol | percent mass |
percent atoms |
|---|---|---|---|---|
| Oxygen | O | 65.0 | 24.0 | |
| Carbon | C | 18.5 | 12.0 | |
| Hydrogen | H | 9.5 | 62.0 | |
| Nitrogen | N | 2.6 | 1.1 | |
| Calcium | Ca | 1.3 | 0.22 | |
| Phosphorus | P | 0.6 | 0.22 | |
| Potassium | K | 0.2 | 0.03 | |
| Sulfur | S | 0.3 | 0.038 | |
| Sodium | Na | 0.2 | 0.037 | |
| Chlorine | Cl | 0.2 | 0.024 | |
| Magnesium | Mg | 0.1 | 0.015 | |
| All others | < 0.1 | < 0.3 |
About 99% of the mass of the human body is made up of six elements: oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorus. Only about 0.85% is composed of another five elements: potassium, sulfur, sodium, chlorine, and magnesium. All 11 are necessary for life. The remaining elements are trace elements, of which more than a dozen are thought on the basis of good evidence to be necessary for life.[1] All of the mass of the trace elements put together (less than 10 grams for a human body) do not add up to the body mass of magnesium, the least common of the 11 non-trace elements.
Other elements
Not all elements which are found in the human body in trace quantities play a role in life. Some of these elements are thought to be simple common contaminants without function (examples: caesium, titanium), while many others are thought to be active toxins, depending on amount (cadmium, mercury, lead, radioactives). In humans, arsenic is toxic, and its levels in foods and dietary supplements are closely monitored to reduce or eliminate its intake.[2]
Some elements (silicon, boron, nickel, vanadium) are probably needed by mammals also, but in far smaller doses. Bromine is used by some (though not all) bacteria, fungi,
Elemental composition list
The average 70 kg (150 lb) adult human body contains approximately 7×1027 atoms and contains at least detectable traces of 60 chemical elements.[5] About 29 of these elements are thought to play an active positive role in life and health in humans.[6]
The relative amounts of each element vary by individual, mainly due to differences in the proportion of fat, muscle and bone in their body. Persons with more fat will have a higher proportion of carbon and a lower proportion of most other elements (the proportion of hydrogen will be about the same). The numbers in the table are averages of different numbers reported by different references.
The adult human body averages ~53% water.
The elements listed below as "Essential in humans" are those listed by the US
| Atomic number | Element | Fraction of mass [11][12][13][14][15][16] |
Mass (kg)[17] | Atomic percent |
Essential in humans[18] | Negative effects of excess | Group
|
|---|---|---|---|---|---|---|---|
| 8 | Oxygen | 0.65 | 45 | 24 | Yes (e.g. water, electron acceptor)[19] | Reactive oxygen species | 16 |
| 6 | Carbon | 0.18 | 13 | 12 | Yes[19] (organic compounds) | 14 | |
| 1 | Hydrogen | 0.10 | 7 | 62 | Yes[19] (e.g. water) | Acidosis | 1 |
| 7 | Nitrogen | 0.02–0.03 | 1.8 | 1.1 | Yes[19] (e.g. DNA and amino acids) | 15 | |
| 20 | Calcium | 0.011–0.015 | 1.0 | 0.22 | Hydroxylapatite in bones ) |
Hypercalcaemia | 2 |
| 15 | Phosphorus | 5–7×10−3 [22] | 0.78 | 0.22 | Yes[19][20][21] (e.g. DNA, Phospholipids and Phosphorylation) | Hyperphosphatemia | 15 |
| 19 | Potassium | 1.5–2×10−3[23] | 0.14 | 0.033 | Na+/K+-ATPase ) |
Hyperkalemia | 1 |
| 16 | Sulfur | 2.5×10−3 | 0.14 | 0.038 | Yes[19] (e.g. Cysteine, Methionine, Biotin, Thiamine) | Sulfhemoglobinemia | 16 |
| 11 | Sodium | 1.5×10−3 | 0.10 | 0.037 | Na+/K+-ATPase ) |
Hypernatremia | 1 |
| 17 | Chlorine | 1.5×10−3 | 0.095 | 0.024 | Yes[20][21] (e.g. Cl-transporting ATPase) | Hyperchloremia | 17 |
| 12 | Magnesium | 500×10−6 | 0.019 | 0.0070 | Yes[20][21] (e.g. binding to ATP and other nucleotides) | Hypermagnesemia | 2 |
| 26 | Iron* | 60×10−6 | 0.0042 | 0.00067 | Yes[20][21] (e.g. Hemoglobin, Cytochromes) | Iron overload | 8 |
| 9 | Fluorine | 37×10−6 | 0.0026 | 0.0012 | Yes ( | Fluorine: Highly toxic
Fluoride: Toxic in high amounts |
17 |
| 30 | Zinc | 32×10−6 | 0.0023 | 0.00031 | Zinc finger proteins ) |
Zinc toxicity | 12 |
| 14 | Silicon | 20×10−6 | 0.0010 | 0.0058 | Probably[28] | 14 | |
| 31 | Gallium | 4.9×10−6 | 0.0007 | 0.00093 | No | Gallium halide poisoning[29] | 13 |
| 37 | Rubidium | 4.6×10−6 | 0.00068 | 0.000033 | No | Potassium replacement | 1 |
| 38 | Strontium | 4.6×10−6 | 0.00032 | 0.000033 | No | Calcium replacement | 2 |
| 35 | Bromine | 2.9×10−6 | 0.00026 | 0.000030 | Maybe[30] | Bromism | 17 |
| 82 | Lead | 1.7×10−6 | 0.00012 | 0.0000045 | No | Lead poisoning | 14 |
| 29 | Copper | 1×10−6 | 0.000072 | 0.0000104 | copper proteins ) |
Copper toxicity | 11 |
| 13 | Aluminium | 870×10−9 | 0.000060 | 0.000015 | No | Aluminium poisoning |
13 |
| 48 | Cadmium | 720×10−9 | 0.000050 | 0.0000045 | No | Cadmium poisoning | 12 |
| 58 | Cerium | 570×10−9 | 0.000040 | No | |||
| 56 | Barium | 310×10−9 | 0.000022 | 0.0000012 | No | toxic in higher amounts | 2 |
| 50 | Tin | 240×10−9 | 0.000020 | 6.0×10−7 | Maybe[1] | 14 | |
| 53 | Iodine | 160×10−9 | 0.000020 | 7.5×10−7 | thyroxine, triiodothyronine ) |
Iodine-induced hyperthyroidism | 17 |
| 22 | Titanium | 130×10−9 | 0.000020 | No | 4 | ||
| 5 | Boron | 690×10−9 | 0.000018 | 0.0000030 | Probably[10][31] | 13 | |
| 34 | Selenium | 190×10−9 | 0.000015 | 4.5×10−8 | Yes[20][21] (e.g. selenocysteine) | Selenium toxicity | 16 |
| 28 | Nickel | 140×10−9 | 0.000015 | 0.0000015 | Maybe[1] | Nickel Toxicity | 10 |
| 24 | Chromium | 24×10−9 | 0.000014 | 8.9×10−8 | Maybe[1][20][21] | 6 | |
| 25 | Manganese | 170×10−9 | 0.000012 | 0.0000015 | Mn-SOD ) |
Manganism | 7 |
| 33 | Arsenic | 260×10−9 | 0.000007 | 8.9×10−8 | Maybe[1][2] | Arsenic poisoning | 15 |
| 3 | Lithium | 31×10−9 | 0.000007 | 0.0000015 | hormones and vitamins ) |
Lithium toxicity | 1 |
| 80 | Mercury | 190×10−9 | 0.000006 | 8.9×10−8 | No | Mercury poisoning | 12 |
| 55 | Caesium | 21×10−9 | 0.000006 | 1.0×10−7 | No | 1 | |
| 42 | Molybdenum | 130×10−9 | 0.000005 | 4.5×10−8 | Yes[20][21] (e.g. the molybdenum oxotransferases, Xanthine oxidase and Sulfite oxidase) | 6 | |
| 32 | Germanium | 5×10−6 | No | 14 | |||
| 27 | Cobalt | 21×10−9 | 0.000003 | 3.0×10−7 | Yes (e.g. Cobalamin/Vitamin B12)[32][33] | 9 | |
| 44 | Ruthenium | 22×10−9 | 0.000007 | No [34] | 8 | ||
| 51 | Antimony | 110×10−9 | 0.000002 | No | toxic | 15 | |
| 47 | Silver | 10×10−9 | 0.000002 | No | 11 | ||
| 41 | Niobium | 1600×10−9 | 0.0000015 | No | 5 | ||
| 40 | Zirconium | 6×10−9 | 0.000001 | 3.0×10−7 | No | 4 | |
| 57 | Lanthanum | 1370×10−9 | 8×10−7 | No | |||
| 52 | Tellurium | 120×10−9 | 7×10−7 | No | 16 | ||
| 39 | Yttrium | 6×10−7 | No | 3 | |||
| 83 | Bismuth | 5×10−7 | No | 15 | |||
| 81 | Thallium | 5×10−7 | No | highly toxic | 13 | ||
| 49 | Indium | 4×10−7 | No | 13 | |||
| 79 | Gold | 3×10−9 | 2×10−7 | 3.0×10−7 | No | uncoated nanoparticles possibly | 11 |
| 21 | Scandium | 2×10−7 | No | 3 | |||
| 73 | Tantalum | 2×10−7 | No | 5 | |||
| 23 | Vanadium | 260×10−9 | 0.000020 | 1.2×10−8 | Possibly[10] (suggested osteo-metabolism (bone) growth factor) | 5 | |
| 90 | Thorium | 1×10−7 | No | toxic, radioactive | |||
| 92 | Uranium | 1×10−7 | 3.0×10−9 | No | toxic, radioactive | ||
| 62 | Samarium | 5.0×10−8 | No | ||||
| 74 | Tungsten | 2.0×10−8 | No | 6 | |||
| 4 | Beryllium | 3.6×10−8 | 4.5×10−8 | No | toxic in higher amounts | 2 | |
| 88 | Radium | 3×10−14 | 1×10−17 | No | toxic, radioactive | 2 | |
| 2 | Helium | 20.39×10−21 | 2.4×10−14 | 1×10−17 | No | noble gas | 18 |
| 10 | Neon | 8.5×10−23 | 1×10−14 | 1×10−17 | No | noble gas | 18 |
| 18 | Argon | 4.25×10−23 | 0.5×10−14 | 1×10−17 | No | noble gas | 18 |
| 36 | Krypton | 2.125×10−23 | 0.25×10−14 | 1×10−17 | No | noble gas | 18 |
*Iron = ~3 g in males, ~2.3 g in females
Of the 94 naturally occurring chemical elements, 61 are listed in the table above. Of the remaining 33, it is not known how many occur in the human body.
Most of the elements needed for life are relatively common in the Earth's crust. Aluminium, the third most common element in the Earth's crust (after oxygen and silicon), serves no function in living cells, but is toxic in large amounts, depending on its physical and chemical forms and magnitude, duration, frequency of exposure, and how it was absorbed by the human body.[38] Transferrins can bind aluminium.[39]
Periodic table
| Essential elements for higher organisms (eucarya).[40][41][42][30][43] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H | He | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Legend:
The four basic organic elements
Quantity elements
Essential trace elements
Essentiality or function debated
|
Composition
The composition of the human body can be classified as follows:
- Water
- Proteins
- Fats (or lipids)
- Hydroxyapatite in bones
- DNA and RNA
- Inorganic ions such as sodium, potassium, chloride, bicarbonate, phosphate
- Gases mainly being oxygen, carbon dioxide
- Many cofactors.
The estimated contents of a typical 20-micrometre human cell is as follows:[44]
| Compound type | Percent of mass | Mol. weight (daltons) | Compound | Percent of molecules |
|---|---|---|---|---|
| Water | 65 | 18 | 1.74×1014 | 98.73 |
| Other inorganics |
1.5 | N/A | 1.31×1012 | 0.74 |
| Lipids | 12 | N/A | 8.4×1011 | 0.475 |
| Other organics | 0.4 | N/A | 7.7×1010 | 0.044 |
| Protein | 20 | N/A | 1.9×1010 | 0.011 |
| RNA | 1.0 | N/A | 5×107 | 3×10−5 |
| DNA | 0.1 | 1×1011 | 46 | 3×10−11 |
Tissues

|
Cell type | % mass | % cell count |
|---|---|---|---|
| Erythrocytes (red blood cells) | 4.2 | 85.0 | |
| Muscle cells | 28.6 | 0.001 | |
| Adipocytes (fat cells) | 18.6 | 0.2 | |
| Other cells | 14.3 | 14.8 | |
| Extracellular components | 34.3 | - |
Body composition can also be expressed in terms of various types of material, such as:
- Muscle
- Fat
- Bone and teeth
- Nervous tissue (brain and nerves)
- Hormones
- Connective tissue
- Body fluids (blood, lymph, urine)
- Contents of intestinal gas
- Airin lungs
- Epithelium
Composition by cell type
There are many species of bacteria and other microorganisms that live on or inside the healthy human body. In fact, there are roughly as many microbial as human cells in the human body by number.[45][48][49][50][51] (much less by mass or volume). Some of these
See also
- List of organs of the human body
- Hydrostatic weighing
- Dietary element
- Composition of blood
- List of human blood components
- Body composition
- Abundance of elements in Earth's crust
- Abundance of the chemical elements
References
- ^ .
- ^ a b "Arsenic in Food and Dietary Supplements". US Food and Drug Administration. 22 May 2019. Retrieved 20 August 2019.
- PMID 24906154.
- ISBN 978-1-319-23090-6.
- ^ "Questions and Answers - How many atoms are in the human body?". education.jlab.org. Retrieved 4 April 2023.
- ^ "Ultratrace minerals". Authors: Nielsen, Forrest H. USDA, ARS Source: Modern nutrition in health and disease / editors, Maurice E. Shils ... et al.. Baltimore : Williams & Wilkins, c. 1999, p. 283-303. Issue Date: 1999 URI: [1]
- WP:CALCfor the mean of means for males and females, since the two groups are of about equal size
- ^ See table 1. here
- ^ "Guidance for Industry: A Food Labeling Guide 14. Appendix F". US Food and Drug Administration. 1 January 2013. Archived from the original on 4 April 2017.
- ^ ISBN 978-0-309-15742-1. Retrieved 21 June 2016.
- ISBN 978-0-07-110595-8.
- ^ "Elemental Composition of the Human Body" Archived 2018-12-18 at the Wayback Machine by Ed Uthman, MD Retrieved 17 June 2016
- ISBN 978-0-19-850848-9.
- ISBN 978-0-395-98581-6.)
- PMID 1246210.
- PMID 9042814.
- ISBN 978-0-19-960563-7. Retrieved 17 June 2016.
- ^ Nielsen FH. "Ultratrace minerals". Modern nutrition in health and disease / editors, Maurice E. Shils ... [et al.] Retrieved 4 April 2023 – via PubAg.
- ^ ISBN 978-0-07-773093-2. Retrieved 19 June 2016.
- ^ ISBN 978-0-309-04633-6. Retrieved 18 June 2016.
- ^ a b c d e f g h i j k l "Federal Register :: Request Access". unblock.federalregister.gov. Retrieved 4 April 2023.
- PMID 11833045.
- PMID 32469402.
- ^ Australian National Health and Medical Research Council (NHMRC) and New Zealand Ministry of Health (MoH)
- ^ "Fluoride in Drinking Water: A Review of Fluoridation and Regulation Issues"
- ISSN 1831-4732.
- ^ "WHO/SDE/WSH/03.04/96 "Fluoride in Drinking-water"" (PDF). Retrieved 4 April 2023.
- PMID 26617630.
- PMID 22024274.
- ^ PMID 24906154.
- ISBN 1-904026-11-7
- PMID 24470095.
- ISBN 978-94-007-5561-1. Retrieved 19 June 2016.
- ^ Toeniskoetter S (2020). "Ruthenium". Biochemical Periodic Table.
- S2CID 84837060.
- ISSN 1831-4732.
- PMID 11745751.
- PMID 25233067.
- PMID 16301797.
- ^ Ultratrace minerals. Authors: Nielsen, Forrest H. USDA, ARS Source: Modern nutrition in health and disease / editors, Maurice E. Shils ... et al. Baltimore: Williams & Wilkins, c1999., p. 283-303. Issue Date: 1999 URI: [2]
- PMID 30066063.
- PMID 32824874.
- .
- ISBN 978-1-57059-680-3. Archived from the originalon 2018-04-16. Retrieved 2011-04-06.
- ^ PMID 37722043.
- PMID 27541692.
- PMID 23829164.
- ^ American Academy of Microbiology FAQ: Human Microbiome Archived 31 December 2016 at the Wayback Machine January 2014
- ^ Judah L. Rosner for Microbe Magazine, February 2014. Ten Times More Microbial Cells than Body Cells in Humans?
- PMID 29634682.
- PMID 26824647.
