Connexon
| Connexon | |
|---|---|
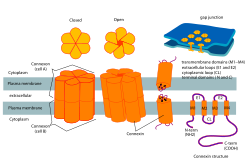 Connexon and connexin structure | |
| Details | |
| Identifiers | |
| Latin | connexona |
| TH | H1.00.01.1.02025 |
| Anatomical terminology | |
In
Connexons made of the same type of connexins are considered
Structure
Assembly
The assembly of connexins destined for gap junction plaques begins with synthesis of connexins within the cell and ends with the formation of gap junction channel plaques on the cell membrane. The connexin subunit proteins that make up connexons are synthesized on the membranes of the cell's endoplasmic reticulum. These subunits are then oligomerized, or combined with other smaller parts, into connexons in the golgi apparatus.[5] The connexons are then delivered to their proper location on the plasma membrane.[6] Connexons then dock with compatible connexons from the neighboring cell to form gap junction channel plaques.[5] A large part of this process is mediated by phosphorylation of different enzymes and proteins, allowing and preventing interaction between certain proteins.[5] The connexons forming channels to the cell exterior or in mitochondria will require a somewhat altered path of assembly.
General
Connexons contribute to the formation of gap junctions, and are an essential component of the electric synapses in neural pathways.
Degradation
Connexon structure is degraded by its removal from the plasma membrane. Connexons will be internalized by the cell itself as a double membrane channel structure (due to the docking of hemi-channels).
Cellular functions
Properties
The properties of individual connexin proteins determine the overall properties of the whole connexon channel. The
Modulation
Communication between gap-junctions can be modulated/regulated in many ways. The main types of modulation are:
- Chemical – one common type of chemical modulation is through the interaction of Ca2+ and certain domains of connexins. It is not completely understood, however, it is suggested that this interaction causes Ca2+ to block the C-terminal domain of connexins which then reduces the channel activity.[2]
- Protein Phosphorylation – protein phosphorylation regulates the communication between channels in multiple ways by controlling: connexin trafficking from the Golgi Apparatus, accumulation of connexons to certain areas, and degradation of unnecessary channels. The process of these actions is very complex but involvement of protein phosphorylation is known.[2]
- Humoral – humoral modulation of gap junction communication is done through many biomolecules such as action potentials down neurons. These types of gap-junctions with this type of modulation are often found in neurons in cardiac tissue and vertebrate retina.[2]
Overall functions
Connexons play an imperative role in behavior and neurophysiology. Many of the details surrounding their pathological functions remain unknown as research has only begun recently. In the central nervous system (CNS), connexons play a major role in conditions such as
Related diseases
Some of the diseases associated with connexons are cardiovascular disease and diabetes, which is the inability of the body to produce insulin for glucose uptake by cells and degradation in the smaller units of connexons, called connexins, possibly leading to the onset of heart disease. Cardiovascular disease and diabetes, type I and II, affects similar locations within cells of the heart and pancreas. This location is the gap junction, where connexons facilitate rapid cell-to-cell interactions via electrical transmissions. Gap junctions are often present at nerve endings such as in cardiac muscle and are important in maintaining homeostasis in the liver and proper function of the kidneys. The gap junction itself is a structure that is a specialized transmembrane protein formed by a connexon hemichannel.[8] Cardiovascular disease and possibly type I and II diabetes, are each associated with a major protein connexin that makes up the gap junction.
In cardiovascular disease, Cx43 (connexin 43), a subunit of a connexon, is a general protein of the gap junction stimulating cardio
Connexons are also associated with both
References
Further reading
- Andrew L Harris and Darren Locke (2009). Connexins, A Guide. New York: Springer. p. 574. ISBN 978-1-934115-46-6.
